Advanced Green Synthesis of Allyl Thio and Seleno Phosphates for Commercial Scale-up
Advanced Green Synthesis of Allyl Thio and Seleno Phosphates for Commercial Scale-up
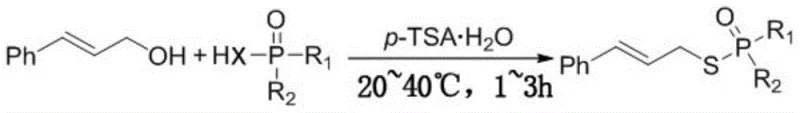
The pharmaceutical and agrochemical industries are constantly seeking more sustainable and efficient pathways for constructing complex molecular scaffolds, particularly those containing phosphorus-sulfur or phosphorus-selenium bonds which are critical for biological activity. Patent CN108129512B introduces a groundbreaking methodology for the preparation of allyl thio or selenophosphate and phosphonate derivatives that fundamentally shifts the paradigm from traditional metal-dependent catalysis to a greener, organocatalytic approach. This innovation leverages p-toluenesulfonic acid monohydrate (p-TSA·H2O) to facilitate the coupling of cinnamyl alcohol with various thiophosphoric or selenophosphoric acid reagents under remarkably mild thermal conditions. By operating effectively at temperatures between 20°C and 40°C, this process not only minimizes energy consumption but also preserves the integrity of sensitive functional groups that might otherwise degrade under harsher regimes. For R&D directors and process chemists, this represents a significant opportunity to streamline the synthesis of high-value intermediates used in the development of novel bactericidal, insecticidal, and medicinal agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of allyl thio and seleno phosphates has often relied upon transition metal catalysts or aggressive reaction conditions that pose substantial challenges for large-scale manufacturing. Traditional routes frequently necessitate the use of heavy metals such as palladium, copper, or nickel, which introduce severe complications regarding residual metal contamination in the final active pharmaceutical ingredient (API). Removing these trace metals to meet stringent regulatory standards requires additional purification steps, such as specialized scavenging resins or repeated recrystallizations, which inevitably drive up production costs and extend lead times. Furthermore, many conventional protocols demand the use of volatile organic solvents in large quantities, creating significant environmental burdens related to solvent recovery, waste treatment, and operator safety. The combination of high energy inputs, toxic reagents, and complex downstream processing renders these older methodologies increasingly obsolete in an era where green chemistry principles are becoming mandatory rather than optional for reliable agrochemical intermediate suppliers.
The Novel Approach
In stark contrast to these legacy techniques, the methodology disclosed in CN108129512B offers a streamlined, metal-free alternative that aligns perfectly with modern sustainability goals. By utilizing p-TSA·H2O as a benign organocatalyst, the reaction proceeds efficiently without the need for any transition metals, thereby eliminating the risk of heavy metal contamination at the source. The process is characterized by its exceptional substrate adaptability, accommodating a wide range of aryl, aryloxy, and alkoxy substituents on the phosphorus center without compromising yield or selectivity. Perhaps most importantly for supply chain optimization, the reaction can be conducted under solvent-free conditions or with minimal solvent usage, drastically reducing the volume of chemical waste generated. This simplification of the reaction matrix allows for faster turnaround times and lower operational expenditures, making it an ideal candidate for the cost reduction in pharmaceutical intermediates manufacturing where margin pressure is always a critical concern.
Mechanistic Insights into p-TSA Catalyzed Substitution
The mechanistic pathway proposed for this transformation involves a sophisticated yet efficient sequence of acid-catalyzed activation and nucleophilic substitution events. Initially, the p-TSA·H2O catalyst serves to protonate the hydroxyl group of the cinnamyl alcohol, increasing its leaving group ability and facilitating the elimination of a water molecule. This dehydration step generates a reactive allylic cation or a transient ether intermediate, which is highly susceptible to nucleophilic attack. The sulfur or selenium species from the thiophosphoric or selenophosphoric acid reagent then acts as a potent nucleophile, attacking the activated allylic position to form the new carbon-sulfur or carbon-selenium bond. This mechanism avoids the formation of unstable radical intermediates often seen in metal-catalyzed cross-couplings, resulting in a cleaner reaction profile with fewer side products. The mild acidity of p-TSA ensures that the reaction environment remains controlled, preventing polymerization or degradation of the sensitive allylic double bond while driving the equilibrium towards the desired product.
From an impurity control perspective, this mechanism offers distinct advantages for ensuring high-purity OLED material or pharmaceutical intermediate standards. Because the reaction does not involve redox-active metal centers, there is no risk of generating metal-induced oxidative byproducts that are notoriously difficult to separate. The primary byproduct of the reaction is water, which is easily removed during the workup or concentration phases, further simplifying the purification strategy. The selectivity of the nucleophilic attack is governed by the electronic properties of the phosphorus reagent and the stability of the allylic intermediate, allowing for precise tuning of the reaction outcome. For quality assurance teams, this predictable mechanistic behavior translates into a robust and reproducible process where the impurity profile is well-understood and easily managed through standard chromatographic techniques, ensuring consistent batch-to-batch quality essential for commercial scale-up of complex polymer additives or fine chemicals.
How to Synthesize Allyl Thio Phosphate Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to stoichiometry and thermal management to maximize the benefits of the patented protocol. The procedure begins with the precise weighing of cinnamyl alcohol and the chosen thiophosphoric or selenophosphoric acid reagent, typically in a molar ratio ranging from 1:1 to 2:1 depending on the specific substrate reactivity. The addition of the p-TSA·H2O catalyst is critical, with optimal loading generally found between equimolar amounts relative to the alcohol to ensure complete conversion without excessive acid waste. Once the components are mixed, the reaction vessel is sealed and maintained at a controlled temperature, ideally between 25°C and 35°C, for a duration of 1 to 3 hours. Detailed standardized synthesis steps for scaling this process from gram to kilogram quantities are provided in the guide below.
- Mix cinnamyl alcohol with the selected sulfur or selenium reagent (thiophosphoric or selenophosphoric acid) in a reaction vessel.
- Add p-TSA·H2O as the catalyst and maintain the reaction mixture at a mild temperature between 20°C and 40°C for 1 to 3 hours.
- Purify the resulting crude product by washing with saturated brine and performing column chromatography to isolate the high-purity allyl thio phosphate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology offers tangible strategic benefits that extend far beyond simple chemical yield. The elimination of transition metal catalysts represents a direct removal of a high-cost input, as precious metals like palladium are subject to significant market volatility and supply constraints. By switching to an organocatalytic system based on p-TSA, manufacturers can stabilize their raw material costs and reduce dependency on scarce resources. Furthermore, the absence of heavy metals simplifies the regulatory compliance landscape, reducing the administrative burden and testing costs associated with proving low metal residuals in the final product. This streamlined compliance pathway accelerates the time-to-market for new drug candidates or agrochemical formulations, providing a competitive edge in fast-moving sectors.
- Cost Reduction in Manufacturing: The economic impact of this solvent-free, metal-free process is profound, as it removes the need for expensive solvent recovery infrastructure and hazardous waste disposal services. Without the requirement for large volumes of organic solvents during the reaction phase, the energy load for heating, cooling, and distilling solvents is drastically reduced, leading to substantial operational savings. Additionally, the simplified workup procedure, which often involves basic aqueous washes and crystallization rather than complex extractions, lowers labor costs and increases equipment throughput. These cumulative efficiencies result in a significantly lower cost of goods sold (COGS), allowing companies to offer more competitive pricing for high-purity pharmaceutical intermediates while maintaining healthy profit margins.
- Enhanced Supply Chain Reliability: The reagents required for this synthesis, such as cinnamyl alcohol and various thiophosphoric acids, are commercially available from multiple global suppliers, mitigating the risk of single-source bottlenecks. The mild reaction conditions mean that the process can be run in standard glass-lined or stainless steel reactors without the need for specialized high-pressure or cryogenic equipment, increasing the number of qualified contract manufacturing organizations (CMOs) capable of producing the material. This flexibility enhances supply chain resilience, ensuring that production schedules are not disrupted by equipment availability or extreme weather events that might impact energy-intensive processes. Consequently, buyers can secure reducing lead time for high-purity pharmaceutical intermediates with greater confidence in delivery continuity.
- Scalability and Environmental Compliance: Scaling this reaction from the bench to multi-ton production is straightforward due to the exothermic nature of the acid-catalyzed substitution being manageable at low temperatures. The lack of volatile solvents minimizes the risk of fire and explosion, improving plant safety profiles and lowering insurance premiums. From an environmental standpoint, the high atom economy and minimal waste generation align with strict international environmental regulations, such as REACH and TSCA, facilitating easier permitting for new manufacturing lines. This sustainability advantage is increasingly valued by end-users who are under pressure to reduce their Scope 3 emissions, making this green synthesis route a preferred choice for responsible sourcing initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear understanding of the process capabilities. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the primary advantages of this synthesis method over traditional metal-catalyzed routes?
A: This method eliminates the need for expensive and toxic transition metal catalysts, thereby reducing heavy metal impurities in the final API intermediate and simplifying the purification workflow significantly.
Q: Does this process require the use of volatile organic solvents during the reaction phase?
A: No, the patented procedure operates under solvent-free or near-solvent-free conditions during the reaction step, which drastically lowers VOC emissions and reduces the cost associated with solvent recovery and waste disposal.
Q: What is the typical yield range for these allyl thio phosphate derivatives?
A: Experimental data from the patent indicates robust yields ranging from 78% to 89%, demonstrating high atom economy and process efficiency suitable for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Allyl Thio Phosphate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of green chemistry innovations like the one described in CN108129512B for the future of fine chemical manufacturing. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to industrial supply is seamless and efficient. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, including the detection of trace metal impurities down to ppm levels, guaranteeing that every batch meets the highest industry standards. We are committed to leveraging our technical expertise to optimize this metal-free route for your specific application needs, whether for pharmaceutical APIs or advanced agrochemical formulations.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis technology can be tailored to your project requirements. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the potential economic benefits of switching to this greener methodology for your supply chain. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive both innovation and profitability in your organization.
