Advanced DCC-Mediated Synthesis of Chlorogenic Acid for Scalable Pharmaceutical Manufacturing
The global demand for bioactive phenolic compounds continues to surge, driven by their extensive applications in pharmaceuticals, nutraceuticals, and functional foods. Among these, chlorogenic acid stands out as a critical molecule due to its potent antioxidant, anti-inflammatory, and hepatoprotective properties. However, relying solely on plant extraction limits supply consistency and purity. Addressing this challenge, the technical disclosure in patent CN101293832A presents a robust synthetic methodology that bypasses the limitations of natural extraction. This report analyzes the strategic value of this DCC-mediated esterification route, highlighting its potential to secure a reliable chlorogenic acid supplier channel for high-volume manufacturing. By shifting from extraction to controlled chemical synthesis, manufacturers can achieve superior batch-to-batch reproducibility and access to diverse derivatives that are otherwise scarce in nature.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the chemical synthesis of chlorogenic acid and its analogues has been plagued by inefficiency and complexity. Early attempts, such as the seminal work by Panizzi in 1955, utilized quinic acid directly but suffered from catastrophic yields of less than 5% due to the necessity of multiple protection and deprotection steps. The traditional acid chloride method, while conceptually straightforward, introduces severe operational hazards and purity issues. Converting caffeic acid derivatives into acid chlorides typically requires aggressive reagents like thionyl chloride or oxalyl chloride, which can degrade the sensitive catechol moiety. Furthermore, the subsequent coupling often necessitates alkaline conditions for deprotection, under which the electron-rich phenolic rings are highly susceptible to rapid oxidation, leading to dark-colored impurities and difficult purification scenarios. These factors collectively render conventional routes economically unviable for cost reduction in pharmaceutical intermediates manufacturing.
The Novel Approach
The methodology outlined in the patent introduces a paradigm shift by employing a direct condensation strategy mediated by carbodiimides. Instead of pre-activating the acid as a chloride, the process utilizes dicyclohexylcarbodiimide (DCC) or its analogues to activate the carboxylic acid in situ. This approach allows the reaction to proceed under significantly milder conditions, typically between 0°C and 100°C, often merely requiring room temperature. Crucially, the deprotection step is conducted in a diluted acid solution rather than a base, effectively preserving the integrity of the phenolic structure and preventing oxidative degradation. This streamlined two-step sequence—condensation followed by acidic hydrolysis—drastically reduces the number of unit operations, minimizes solvent consumption, and delivers moderate to good yields, exemplified by the 58.7% yield achieved in the primary embodiment.
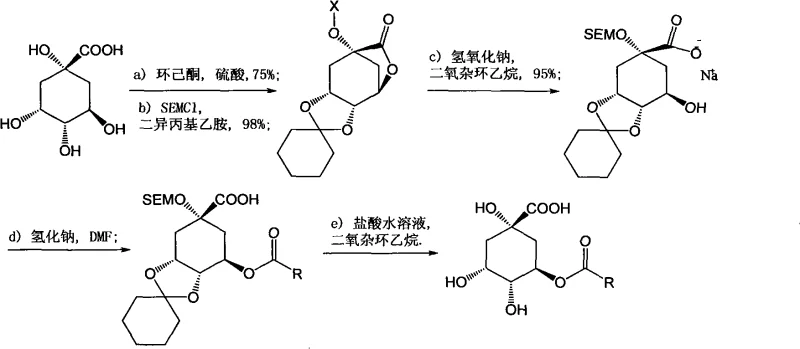
Mechanistic Insights into DCC-Catalyzed Esterification
The core of this synthetic innovation lies in the efficient activation of the sterically hindered carboxylic acid group of the cinnamic acid derivative. In the presence of a nucleophilic catalyst such as 4-dimethylaminopyridine (DMAP), DCC reacts with the carboxylic acid to form a highly reactive O-acylisourea intermediate. This species is far more electrophilic than the parent acid but avoids the extreme reactivity and moisture sensitivity associated with acid chlorides. The protected quinic acid, acting as the nucleophile, attacks this activated carbonyl carbon to form the ester bond. The use of DMAP is critical here; it accelerates the acylation by forming an even more reactive acylpyridinium intermediate, ensuring that the coupling occurs selectively at the desired hydroxyl position on the quinic acid scaffold without significant racemization or side reactions.
Impurity control is inherently built into this mechanism through the choice of reagents and workup conditions. A distinct advantage of using DCC is the formation of dicyclohexylurea (DCU) as the stoichiometric byproduct. Unlike soluble salts generated in other coupling methods, DCU is largely insoluble in common organic solvents like dichloromethane or tetrahydrofuran at room temperature. This physical property allows for the bulk of the chemical waste to be removed via simple mechanical filtration prior to any chromatographic purification. Furthermore, the final acidic hydrolysis step ensures that any acid-sensitive protecting groups (such as acetals or ketals) are cleaved cleanly without exposing the sensitive catechol system to oxidative basic environments. This mechanistic elegance translates directly into high-purity chlorogenic acid with minimal colored impurities.
How to Synthesize Chlorogenic Acid Efficiently
Implementing this synthesis requires precise control over stoichiometry and reaction monitoring to maximize the yield of the target ester. The process begins with the activation of the phenylacrylic acid component, followed by the addition of the protected quinic acid scaffold. Reaction progress is typically monitored via thin-layer chromatography (TLC) to ensure complete consumption of the starting materials before proceeding to the hydrolysis phase. The detailed standardized synthesis steps below outline the specific molar ratios, solvent choices, and workup procedures validated in the patent examples to ensure reproducible results.
- Activate the phenylacrylic acid derivative (e.g., caffeic acid protected form) using DCC and a catalytic amount of DMAP in an organic solvent like dichloromethane at room temperature.
- Add the gem-dioxolane protected quinic acid compound to the activated mixture and stir for 1 to 72 hours to form the protected ester intermediate.
- Perform acidic hydrolysis using dilute hydrochloric acid in tetrahydrofuran to remove protecting groups, followed by extraction and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this DCC-mediated synthesis offers tangible logistical and financial benefits beyond mere chemical yield. The elimination of the acid chloride preparation step removes the need for handling corrosive gases and specialized corrosion-resistant equipment, thereby lowering capital expenditure and safety compliance costs. Additionally, the ability to filter off the urea byproduct significantly reduces the load on downstream purification systems, such as preparative HPLC or silica columns, leading to substantial cost savings in stationary phases and solvents. This efficiency makes the process highly attractive for reducing lead time for high-purity chlorogenic acid derivatives in a commercial setting.
- Cost Reduction in Manufacturing: The economic model of this synthesis is strengthened by the avoidance of expensive coupling reagents like CDI (carbonyldiimidazole) in favor of the more cost-effective DCC. Moreover, the simplified workup procedure, which relies on filtration rather than complex aqueous extractions to remove soluble salts, reduces labor hours and wastewater treatment volumes. The mild reaction temperatures also imply lower energy consumption for heating or cooling large-scale reactors, contributing to a leaner overall production cost structure without compromising product quality.
- Enhanced Supply Chain Reliability: Dependence on plant extraction subjects the supply chain to seasonal variations, weather patterns, and geopolitical instability in sourcing regions. By adopting this synthetic route, manufacturers can decouple production from agricultural cycles, ensuring a consistent year-round supply of high-purity chlorogenic acid. The starting materials, including substituted phenylacrylic acids and quinic acid derivatives, are commodity chemicals with stable global availability, mitigating the risk of raw material shortages that often plague natural extract markets.
- Scalability and Environmental Compliance: The commercial scale-up of complex phenolic esters is often hindered by exothermic risks and hazardous waste generation. This protocol operates safely at or near room temperature, minimizing thermal runaway risks in large vessels. Furthermore, the generation of solid urea waste, which can be potentially recycled or disposed of more easily than liquid acidic or basic waste streams, aligns better with modern environmental regulations. The reduced solvent usage per kilogram of product also supports sustainability goals, making it easier to obtain necessary environmental permits for expanded production capacity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic pathway. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, aiming to clarify the operational feasibility and quality advantages of this method for potential partners and technical stakeholders evaluating the technology.
Q: How does the DCC method compare to traditional acid chloride synthesis for chlorogenic acid?
A: The DCC method described in CN101293832A avoids the use of harsh acid chloride reagents and alkaline hydrolysis steps. This results in significantly higher yields (up to 58.7% in examples) compared to traditional methods which often suffer from oxidation and low yields (<5% to 32%).
Q: What are the purification advantages of using dicyclohexylcarbodiimide (DCC)?
A: A major advantage is the formation of dicyclohexylurea (DCU) as a byproduct, which is insoluble in many organic solvents. This allows for simple removal via filtration before the final purification step, drastically simplifying the workup process compared to methods generating soluble salt byproducts.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the reaction operates under mild conditions (0-100°C, often room temperature) and uses common organic solvents. The elimination of sensitive acid chloride intermediates and the simplicity of the filtration workup make it highly amenable to commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chlorogenic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the theoretical advantages of a patent must be translated into practical, GMP-compliant manufacturing realities. Our R&D team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the DCC-mediated synthesis of chlorogenic acid can be adapted to meet your specific volume requirements. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to verify the identity and potency of every batch, guaranteeing that the final product meets the demanding standards of the global pharmaceutical and nutraceutical industries.
We invite you to collaborate with us to leverage this advanced synthetic technology for your product pipeline. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume needs, demonstrating how this route can optimize your bill of materials. Please contact us today to request specific COA data for our chlorogenic acid intermediates and to discuss route feasibility assessments for your next project, ensuring a secure and efficient supply chain for your critical active ingredients.
