Advanced Synthesis of 2-Fluoroalkyl-3-Alkynyl Naphthofuran Compounds for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to access complex heterocyclic scaffolds, particularly those incorporating fluorine motifs which are critical for metabolic stability and bioavailability. Patent CN111777582B discloses a groundbreaking preparation method for 2-fluoroalkyl-3-alkynyl substituted naphthofuran compounds, addressing the longstanding challenge of selective C(sp3)-F bond activation. This technology represents a significant leap forward in organic synthesis, offering a streamlined pathway to construct valuable naphthofuran cores that possess potential antibacterial, anti-insect, and plant antiviral activities. By leveraging a palladium-catalyzed tandem defluorination and alkynylation strategy, this invention overcomes the limitations of previous multi-step transformations. The general structure of these novel compounds, characterized by a fused naphthalene-furan system with specific fluoroalkyl and alkynyl substitutions, is depicted below, highlighting the structural diversity achievable through this versatile platform.
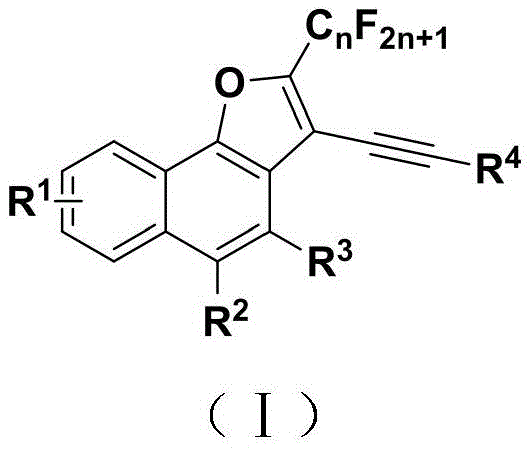
For R&D directors evaluating new synthetic routes, the ability to introduce both fluorine and alkyne functionalities simultaneously is a major asset. The disclosed method utilizes alpha-perfluoroalkyl tetralone compounds as key starting materials, which react with terminal alkynes under mild conditions. This approach not only simplifies the synthetic tree but also opens up new chemical space for drug discovery teams looking for novel scaffolds. The high regioselectivity reported in the patent ensures that the desired isomer is produced predominantly, reducing the burden on downstream purification processes. As a reliable pharmaceutical intermediate supplier, understanding such mechanistic nuances is essential for guaranteeing the quality and consistency of the final active pharmaceutical ingredients (APIs) derived from these precursors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the functionalization of fluorinated compounds has been fraught with difficulties due to the exceptional strength of the carbon-fluorine bond, which is significantly higher than that of carbon-hydrogen or carbon-carbon bonds. Conventional Sonogashira-type reactions involving aryl fluorides often necessitate harsh reaction conditions, including high temperatures and the use of sensitive organometallic reagents that are difficult to handle on a large scale. Furthermore, existing defluoroalkynylation reactions have historically been limited to the activation of single C-F bonds, typically in trifluoromethyl-substituted conjugated systems. This limitation restricts the structural complexity that can be achieved, as activating multiple C-F bonds sequentially without losing control over the reaction outcome is extremely challenging. The reliance on expensive metal catalysts and complicated multi-step procedures in prior art further exacerbates the cost and environmental footprint of producing such specialized intermediates, making them less attractive for commercial manufacturing.
The Novel Approach
In stark contrast to these legacy methods, the technology described in CN111777582B introduces a highly efficient intermolecular defluorination tandem alkynylation reaction. This novel approach allows for the cleavage of multiple C-F bonds (specifically more than three) in a targeted manner while retaining a portion of the fluorine-containing group, a feat rarely achieved in previous literature. The reaction proceeds via a one-pot protocol where alpha-perfluoroalkyl tetralones are mixed with simple alkynes in the presence of a palladium catalyst and a base promoter. As illustrated in the reaction scheme below, this process seamlessly integrates alkynylation, aromatization, and cyclization into a single operational step. The mild reaction conditions, ranging from 25°C to 90°C, and the use of accessible solvents like DMSO make this method exceptionally practical for industrial application. This represents a substantial improvement in cost reduction in pharmaceutical intermediate manufacturing by eliminating the need for protecting groups and intermediate isolation steps.
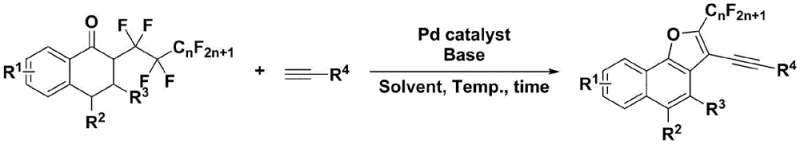
Mechanistic Insights into Pd-Catalyzed Tandem Defluoroalkynylation
The core of this innovation lies in the sophisticated palladium-catalyzed mechanism that facilitates the simultaneous breaking of C-F bonds and formation of C-C bonds. The reaction initiates with the oxidative addition of the palladium catalyst to the C-F bond of the alpha-perfluoroalkyl tetralone substrate. Unlike standard cross-couplings, this system is tuned to promote a cascade sequence where the initial activation triggers a series of elimination and cyclization events. The base promoter, optimally identified as cesium carbonate (Cs2CO3), plays a critical role in deprotonating the terminal alkyne and facilitating the defluorination steps. The choice of ligand on the palladium center, such as triphenylphosphine in Pd(PPh3)2Cl2, is crucial for stabilizing the catalytic species and ensuring high turnover numbers. This precise control over the catalytic cycle prevents the complete defluorination of the alkyl chain, allowing for the retention of the fluoroalkyl group at the 2-position of the naphthofuran ring, which is vital for the biological activity of the final molecule.
From an impurity control perspective, the high chemoselectivity of this tandem reaction is a significant advantage for commercial production. The specific reaction conditions minimize side reactions such as homocoupling of the alkyne or over-reaction of the fluorinated chain. The patent data indicates that by optimizing the molar ratios of reactants—specifically maintaining a ratio of tetralone to alkyne around 1:1.5—and selecting the appropriate solvent system, the formation of by-products is drastically reduced. This inherent selectivity means that the crude reaction mixture is cleaner, simplifying the subsequent workup and purification stages. For supply chain managers, this translates to higher overall yields and reduced waste generation, aligning with green chemistry principles. The ability to tolerate various functional groups on both the tetralone and alkyne substrates further demonstrates the robustness of this catalytic system, making it a versatile tool for synthesizing a wide library of analogs for structure-activity relationship (SAR) studies.
How to Synthesize 2-Fluoroalkyl-3-Alkynyl Naphthofuran Efficiently
The synthesis of these high-value intermediates is designed to be operationally simple, requiring standard laboratory equipment and commercially available reagents. The process begins with the preparation of the reaction mixture under an inert atmosphere to prevent catalyst deactivation. Detailed optimization studies within the patent reveal that dimethyl sulfoxide (DMSO) serves as the superior solvent, providing the necessary polarity to dissolve the ionic intermediates while stabilizing the transition states. The reaction temperature is a critical parameter, with 70°C identified as the optimal balance between reaction rate and selectivity, although the process remains effective across a broad range from 25°C to 90°C. Following the reaction period, typically 12 hours, the product is isolated through a straightforward aqueous workup and column chromatography. For a comprehensive guide on executing this synthesis with maximum efficiency and safety, please refer to the standardized protocol outlined below.
- Mix alpha-perfluoroalkyl tetralone compound with terminal alkyne compound in a reaction vessel under inert atmosphere.
- Add palladium catalyst (e.g., Pd(PPh3)2Cl2), base promoter (e.g., Cs2CO3), and solvent (e.g., DMSO) to the mixture.
- Stir the reaction at 25-90°C for 1-24 hours, then wash, extract, dry, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers tangible benefits in terms of cost efficiency and supply reliability. The primary driver for cost reduction is the simplicity of the raw materials; alpha-perfluoroalkyl tetralones and terminal alkynes are readily accessible from bulk chemical suppliers, avoiding the need for custom-synthesized, high-cost precursors. Furthermore, the one-pot nature of the reaction eliminates the logistical complexities and material losses associated with multi-step syntheses. By consolidating alkynylation, aromatization, and cyclization into a single vessel, the process significantly reduces solvent consumption, energy usage, and labor hours. This streamlined workflow directly contributes to lower manufacturing costs, allowing for more competitive pricing of the final pharmaceutical intermediates without compromising on quality or purity standards.
- Cost Reduction in Manufacturing: The elimination of expensive and sensitive organometallic reagents, which are often required in traditional fluorination strategies, leads to substantial cost savings. The use of a robust palladium catalyst system that operates efficiently at low loading levels (e.g., 10 mol%) further optimizes the cost structure. Additionally, the high yield and selectivity reported in the patent mean that less raw material is wasted on by-products, maximizing the atom economy of the process. This efficiency is critical for scaling up production, as it ensures that the cost per kilogram of the intermediate decreases significantly as volume increases, providing a clear economic advantage over legacy methods.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals and standard reaction conditions enhances the resilience of the supply chain. Since the process does not depend on exotic catalysts or cryogenic conditions, it can be easily transferred between different manufacturing sites or scaled up rapidly to meet surging demand. The mild reaction temperatures (25-90°C) reduce the risk of thermal runaways and equipment failure, ensuring consistent batch-to-batch quality. This reliability is paramount for long-term supply agreements with pharmaceutical companies, where continuity of supply is often as important as price. The ability to produce a diverse range of derivatives from a common platform also allows for agile responses to changing market needs.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work effectively with various substrates and scales in the patent examples. The use of DMSO, a high-boiling polar aprotic solvent, facilitates heat transfer and mixing in large reactors, which is essential for safe commercial scale-up of complex pharmaceutical intermediates. Moreover, the reduced number of steps and the high selectivity of the reaction result in less chemical waste, simplifying effluent treatment and helping manufacturers meet stringent environmental regulations. This alignment with sustainability goals adds value for clients who are increasingly focused on the environmental footprint of their supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 2-fluoroalkyl-3-alkynyl naphthofuran compounds. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details is crucial for making informed decisions about integrating this technology into your development pipeline or procurement strategy.
Q: What are the key advantages of this defluoroalkynylation method over traditional Sonogashira couplings?
A: Unlike traditional methods that often require harsh conditions and activate only single C-F bonds, this novel approach enables continuous activation of multiple C(sp3)-F bonds in a tandem process. It achieves high regioselectivity and chemoselectivity under mild temperatures (25-90°C), constructing the naphthofuran skeleton directly while introducing the alkynyl group.
Q: Is this synthesis route scalable for industrial production of pharmaceutical intermediates?
A: Yes, the process is highly scalable. It utilizes readily available raw materials like alpha-perfluoroalkyl tetralones and simple terminal alkynes. The reaction uses common solvents like DMSO and standard bases like cesium carbonate, avoiding exotic reagents. The one-pot nature simplifies operation, making it suitable for commercial scale-up from kilograms to tons.
Q: What represents the primary challenge in C-F bond activation that this patent addresses?
A: The primary challenge is the high bond energy of C-F single bonds and the difficulty in controlling regioselectivity when multiple similar C-F bonds exist. This patent solves this by using a specific palladium catalytic system that selectively activates multiple C-F bonds in the perfluoroalkyl chain while retaining specific fluorine content, achieving a balance between defluorination and functionalization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Fluoroalkyl-3-Alkynyl Naphthofuran Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this Pd-catalyzed defluoroalkynylation technology for the development of next-generation therapeutics and functional materials. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to full-scale manufacturing is seamless. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Whether you require custom synthesis of specific analogs or bulk supply of the core scaffold, our infrastructure is designed to support your most demanding projects with speed and precision.
We invite you to collaborate with us to leverage this advanced synthetic methodology for your upcoming programs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact us today to request specific COA data for our available inventory or to discuss route feasibility assessments for your proprietary molecules. Let us be your partner in turning innovative chemistry into commercial success.
