Advanced Green Synthesis of N(2)-L-Alanyl-L-Glutamine for Commercial Scale-Up
Advanced Green Synthesis of N(2)-L-Alanyl-L-Glutamine for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust, scalable, and environmentally benign pathways for producing critical parenteral nutrition intermediates. Patent CN103265616A introduces a transformative synthetic methodology for N(2)-L-alanyl-L-glutamine, a stable dipeptide essential for clinical nutrition support. Unlike traditional routes that rely on hazardous reagents or complex protection-deprotection sequences, this innovation leverages a concise three-step sequence starting from readily available L-glutamine and pyruvoyl chloride. The process is characterized by its operation in aqueous and alcoholic media, eliminating the need for chlorinated solvents and significantly reducing the environmental footprint. For R&D directors and procurement strategists, this patent represents a pivotal shift towards sustainable manufacturing, offering a pathway to high-purity intermediates with improved cost structures and supply chain reliability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of N(2)-L-alanyl-L-glutamine has been plagued by significant safety and economic bottlenecks. One prevalent method involves the reaction of D-2-halopropionic acid derivatives with L-glutamine, followed by ammonolysis. While chemically feasible, this route suffers from the high cost and limited availability of optically pure D-2-chloropropionic acid. Furthermore, the final ammonolysis step requires a large excess of ammonia, generating substantial wastewater pollution that complicates regulatory compliance and waste treatment costs. Another established route utilizes phosgene to activate L-alanine, posing severe safety risks due to the extreme toxicity of phosgene gas, necessitating specialized containment equipment and rigorous personnel training that drives up capital expenditure.
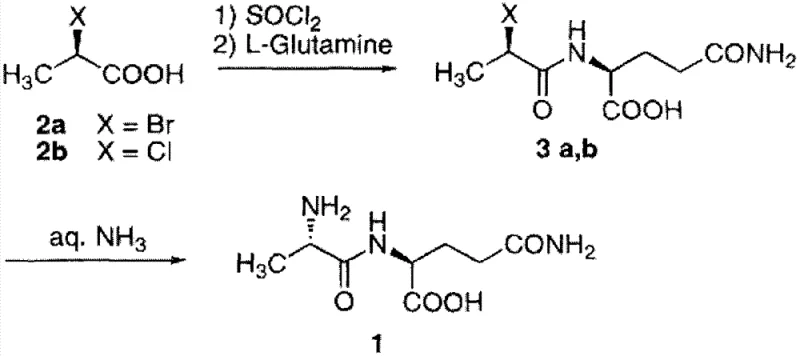
Additionally, protection-group strategies involving BOC or Cbz protection of L-alanine followed by activation with expensive coupling reagents like DCC and N-hydroxysuccinimide add unnecessary complexity. These multi-step sequences not only lower overall yields due to cumulative losses but also introduce difficult-to-remove impurities, requiring extensive purification protocols. The reliance on costly chiral auxiliaries or protected amino acid precursors inflates the raw material bill of materials, making these conventional methods less competitive in a price-sensitive generic pharmaceutical market. Consequently, there is a critical demand for a streamlined process that bypasses these inefficiencies while maintaining strict stereochemical control.
The Novel Approach
The methodology disclosed in CN103265616A offers a paradigm shift by utilizing a direct acylation strategy followed by a clever stereocontrolled reduction. The process initiates with the reaction of L-glutamine and pyruvoyl chloride in an aqueous alkaline solution, forming pyruvoyl-L-glutamine with high efficiency. This step avoids organic solvents entirely, leveraging water as the primary reaction medium, which simplifies downstream processing and solvent recovery. The subsequent conversion to an oxime or oxime ether intermediate sets the stage for the critical stereoselective hydrogenation. By avoiding the use of pre-formed chiral halides or toxic phosgene, this route significantly lowers the barrier to entry for manufacturers and reduces the dependency on scarce starting materials.
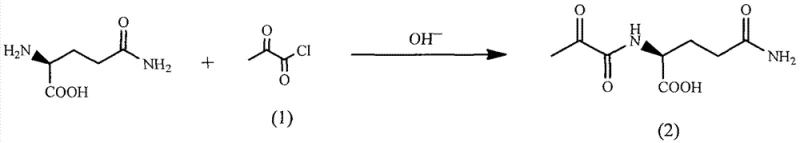
Furthermore, the novel approach integrates a unique hydrogenation step that simultaneously establishes the chiral center and reduces the functional group, effectively merging two synthetic operations into one. The use of commodity chemicals such as hydroxylamine hydrochloride and simple palladium catalysts ensures that the supply chain remains resilient against market fluctuations. This streamlined workflow not only accelerates the production timeline but also minimizes the generation of hazardous byproducts, aligning perfectly with modern green chemistry principles and corporate sustainability goals. The result is a robust, scalable process capable of delivering high-quality intermediates suitable for parenteral applications.
Mechanistic Insights into Stereoselective Catalytic Hydrogenation
The cornerstone of this synthesis lies in the final hydrogenation step, where the prochiral oxime or oxime ether intermediate is converted into the chiral alanine moiety. Mechanistically, this transformation relies on a sophisticated coordination phenomenon rather than expensive chiral ligands. During the reaction, the oxygen atom of the amide carbonyl and the nitrogen atom of the oxime group coordinate with the palladium catalyst surface to form a transient chelated intermediate. This specific spatial arrangement locks the conformation of the substrate, directing the incoming hydrogen atoms to attack from a specific face. The addition of sterically hindered amines, such as cyclohexylamine or tert-butylamine, further enhances this induction effect by modifying the electronic environment around the catalyst active sites.
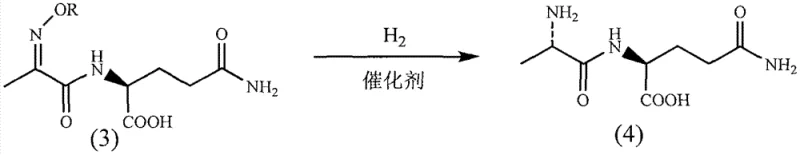
This chelation-controlled mechanism allows for the generation of the desired L-configuration with high diastereomeric excess (d.e.), often exceeding 95% after simple recrystallization. From an impurity control perspective, this mechanism is highly advantageous because it minimizes the formation of the undesired D-isomer, which is difficult to separate and potentially toxic. The ability to tune the stereochemical outcome by simply adjusting the amine additive and catalyst loading provides process chemists with a powerful handle for optimization. Moreover, the heterogeneous nature of the palladium catalyst facilitates easy removal via filtration, ensuring that the final product meets stringent heavy metal specifications required for injectable drugs without the need for complex scavenging resins.
How to Synthesize N(2)-L-Alanyl-L-Glutamine Efficiently
Implementing this synthesis requires precise control over reaction parameters, particularly pH and temperature, to maximize yield and purity. The initial acylation must be maintained at low temperatures (0-10°C) and alkaline pH (9-11) to prevent hydrolysis of the acid chloride and racemization. Following isolation, the oximation step proceeds smoothly in ethanol, where the choice between hydroxylamine or O-alkylhydroxylamine can influence the crystallinity of the intermediate. The final hydrogenation is conducted under mild hydrogen pressure (1-5 atm) in alcohol-water mixtures, balancing solubility with reaction rate. Detailed standard operating procedures for each unit operation are critical for successful technology transfer.
- React L-Glutamine with pyruvoyl chloride in an aqueous alkaline solution (pH 9-11) at 0-10°C to form pyruvoyl-L-glutamine.
- Condense pyruvoyl-L-glutamine with hydroxylamine hydrochloride or O-alkylhydroxylamine in ethanol to generate the oxime or oxime ether intermediate.
- Perform stereoselective catalytic hydrogenation using a Palladium catalyst and a sterically hindered amine base to yield high-purity N(2)-L-alanyl-L-glutamine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic route offers tangible strategic benefits beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the raw material portfolio. By replacing expensive protected amino acids and toxic phosgene with bulk commodities like pyruvic acid derivatives and L-glutamine, manufacturers can achieve substantial cost reductions in API manufacturing. The elimination of hazardous reagents also translates to lower insurance premiums and reduced costs associated with hazardous waste disposal, directly improving the bottom line. Furthermore, the use of water and ethanol as primary solvents mitigates the volatility risks associated with petrochemical-derived solvents, ensuring more stable long-term pricing.
- Cost Reduction in Manufacturing: The economic model of this process is fundamentally superior due to the avoidance of stoichiometric coupling reagents like DCC and expensive chiral pool starting materials. By utilizing a catalytic hydrogenation step to establish chirality, the process eliminates the need for costly resolution steps or chiral chromatography, which are often the most expensive parts of peptide synthesis. Additionally, the ability to recover and recycle the palladium catalyst after filtration further decreases the variable cost per kilogram. This lean manufacturing approach allows for competitive pricing in the global market for parenteral nutrition ingredients.
- Enhanced Supply Chain Reliability: Sourcing stability is significantly improved as the key starting materials, L-glutamine and pyruvoyl chloride precursors, are produced by multiple suppliers globally, reducing the risk of single-source bottlenecks. The robustness of the aqueous reaction conditions means that the process is less sensitive to minor variations in raw material quality compared to sensitive organometallic couplings. This resilience ensures consistent production schedules and reliable delivery timelines, which is critical for maintaining inventory levels of life-saving nutrition products. The simplified logistics of handling non-hazardous solvents also streamline transportation and storage requirements.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing unit operations such as aqueous precipitation and filtration that are easily transferred from pilot plant to multi-ton production reactors. The absence of chlorinated solvents and toxic gases like phosgene simplifies environmental permitting and reduces the burden on scrubber systems and effluent treatment plants. This "green" profile aligns with increasingly strict global environmental regulations, future-proofing the manufacturing site against potential regulatory crackdowns. The high atom economy of the hydrogenation step further minimizes waste generation, supporting corporate sustainability initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. Understanding these nuances is essential for evaluating the feasibility of adopting this technology within existing manufacturing frameworks. The answers are derived directly from the experimental data and mechanistic explanations provided in the patent literature, ensuring accuracy and relevance for technical decision-makers.
Q: How does this synthesis method improve safety compared to traditional phosgene routes?
A: This method eliminates the use of highly toxic phosgene and hazardous organic solvents, utilizing water and ethanol instead, which drastically reduces operational risks and environmental compliance costs.
Q: What mechanism ensures the stereochemical purity of the final product?
A: Stereocontrol is achieved during the hydrogenation step through a chelation mechanism where the oxime nitrogen and amide oxygen coordinate with the palladium catalyst, induced by sterically hindered amine bases.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process uses commodity-grade raw materials like pyruvic acid derivatives and L-glutamine, operates under mild conditions, and allows for catalyst recycling, making it highly scalable and cost-effective.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N(2)-L-Alanyl-L-Glutamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of N(2)-L-alanyl-L-glutamine in modern parenteral nutrition therapy and are committed to delivering this intermediate with the highest standards of quality and consistency. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements regardless of market demand fluctuations. We operate stringent purity specifications and utilize rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch meets the exacting standards required for pharmaceutical applications, including residual solvent and heavy metal limits.
We invite you to collaborate with us to leverage this innovative synthesis technology for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume needs. We are prepared to provide specific COA data from our pilot batches and comprehensive route feasibility assessments to demonstrate how our optimized process can enhance your product competitiveness. Let us be your partner in securing a sustainable and cost-effective supply of this vital pharmaceutical intermediate.
