Advanced NHC-Catalyzed Asymmetric Synthesis of (S)-3-Amino-2-benzyl Methyl Propionate for Commercial Scale-up
The pharmaceutical industry's relentless pursuit of efficient, scalable, and environmentally benign synthetic routes has brought significant attention to the advancements detailed in Chinese Patent CN112209850A. This intellectual property discloses a robust methodology for the preparation of (S)-3-amino-2-benzyl methyl propionate, a critical beta-amino acid derivative serving as a versatile building block in drug synthesis and protein modification. Unlike traditional approaches that rely on wasteful resolution techniques or unstable enzymatic processes, this invention leverages the power of N-Heterocyclic Carbene (NHC) organocatalysis to construct the chiral center with high precision. For R&D directors and procurement strategists alike, this represents a paradigm shift towards more sustainable and cost-effective manufacturing of complex chiral intermediates. The process not only simplifies the operational workflow but also ensures high enantiomeric ratios, addressing the stringent purity requirements of modern active pharmaceutical ingredient (API) supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral beta-amino acids like (S)-3-amino-2-benzyl methyl propionate has been plagued by significant inefficiencies that hinder large-scale commercial viability. Traditional chiral resolution methods, including crystallization and chemical resolution, inherently suffer from a maximum theoretical yield of merely 50%, as they necessitate the discard or recycling of the unwanted enantiomer. Furthermore, biological enzyme resolution, while selective, often lacks the stability required for rigorous industrial conditions and is frequently unsuitable for mass production due to sensitivity to reaction parameters. Chromatographic resolution, though universal, imposes prohibitive equipment costs and is limited to small-scale preparations, making it economically unfeasible for tonnage production. Additionally, asymmetric synthesis induced by chiral prosthetic groups often demands stoichiometric amounts of expensive chiral auxiliaries and harsh reaction conditions involving strong bases like lithium bis(trimethylsilyl)amide (LiHMDS), which pose safety and handling challenges in a plant setting.
The Novel Approach
In stark contrast, the novel approach outlined in the patent utilizes a catalytic asymmetric synthesis strategy that fundamentally alters the economic and operational landscape of producing these intermediates. By employing N-Heterocyclic Carbenes (NHCs) as organocatalysts, the method directly constructs the chiral center from achiral starting materials, thereby bypassing the 50% yield ceiling associated with resolution techniques. This catalytic system operates under mild conditions, avoiding the need for cryogenic temperatures or pyrophoric reagents often seen in metal-mediated asymmetric alkylations. The use of readily available precursors such as p-nitrophenol cyanoacetate and benzyl bromide ensures a stable supply chain, while the subsequent hydrogenation step utilizes Raney nickel, a cost-effective alternative to precious metal catalysts like palladium or platinum. This combination of organocatalysis and heterogeneous hydrogenation creates a streamlined pathway that is both chemically elegant and industrially pragmatic.
Mechanistic Insights into NHC-Catalyzed Asymmetric Alkylation
The core of this technological breakthrough lies in the unique ability of the N-Heterocyclic Carbene catalyst to activate the nucleophile and control the stereochemical outcome of the alkylation reaction. The mechanism involves the generation of a chiral enolate equivalent from the cyanoacetate precursor, which is then intercepted by the electrophilic benzyl bromide within the chiral pocket of the catalyst. The specific structure of the NHC, particularly the substituents on the triazolium ring and the fused bicyclic framework, plays a pivotal role in shielding one face of the reactive intermediate, thus enforcing high facial selectivity. This precise spatial arrangement allows for the formation of the desired (S)-enantiomer with exceptional fidelity, as evidenced by the high e.r. values reported in the experimental data. The catalytic cycle is completed by the release of the product and regeneration of the active carbene species, allowing for turnover numbers that make the process economically viable.
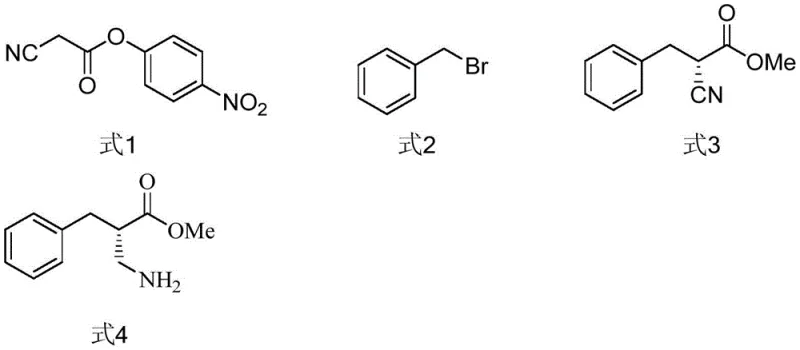
Furthermore, the versatility of this catalytic system is underscored by the tunability of the NHC ligand structure. The patent discloses a library of catalysts where variations in the aryl substituents (R1, R2) and the backbone substitution (X) allow for fine-tuning of the steric and electronic properties to match specific substrate requirements. For instance, catalysts such as NHC-IV and NHC-VII have demonstrated superior performance in this transformation, highlighting the importance of catalyst screening in optimizing reaction outcomes. This modularity ensures that the process can be adapted for various analogues, providing a robust platform for the synthesis of diverse beta-amino acid derivatives. The ability to achieve high stereoselectivity without the use of toxic heavy metals also aligns with the increasing regulatory pressure to minimize metal residues in pharmaceutical products, adding another layer of value to this synthetic strategy.
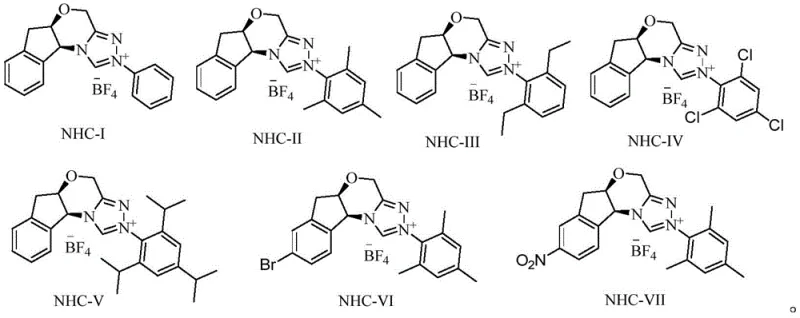
How to Synthesize (S)-3-Amino-2-benzyl Methyl Propionate Efficiently
The synthesis protocol described in the patent offers a clear and reproducible pathway for generating the target chiral intermediate with high purity and yield. The process is designed to be operationally simple, minimizing the need for complex purification steps between the alkylation and reduction stages. By conducting the NHC-catalyzed alkylation in a common solvent like tetrahydrofuran and quenching directly with methanol, the reaction mixture can be seamlessly transitioned into the hydrogenation step. This telescoped approach reduces solvent consumption and processing time, which are critical factors in determining the overall cost of goods. The detailed experimental procedures provided in the patent serve as a reliable blueprint for scaling this chemistry from the laboratory bench to pilot plant operations, ensuring consistency in product quality.
- Perform N-heterocyclic carbene (NHC) catalysis on p-nitrophenol cyanoacetate and benzyl bromide in an organic solvent with a base, followed by methanol quenching to obtain the chiral cyano intermediate.
- Conduct hydrogenation reduction on the chiral cyano intermediate using Raney nickel catalyst in methanol under a hydrogen atmosphere to yield the target amino ester.
- Purify the final product by extraction and convert to the hydrochloride salt using dry hydrogen chloride gas for isolation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this NHC-catalyzed process translates into tangible strategic advantages that extend beyond mere chemical efficiency. The elimination of stoichiometric chiral auxiliaries and the avoidance of expensive noble metal catalysts significantly reduce the raw material cost profile of the intermediate. Moreover, the use of robust and inexpensive reagents like Raney nickel and benzyl bromide ensures a resilient supply chain that is less susceptible to market volatility associated with rare earth or precious metals. The simplicity of the workup procedure, which relies on standard extraction and filtration techniques rather than complex chromatography, facilitates faster batch turnover and higher throughput in manufacturing facilities. These factors collectively contribute to a more predictable and cost-effective supply of high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The process achieves substantial cost savings by replacing expensive chiral resolving agents and precious metal catalysts with organocatalysts and Raney nickel. The high atom economy of the asymmetric synthesis avoids the inherent 50% material loss of resolution methods, effectively doubling the yield potential from the same amount of starting material. Additionally, the mild reaction conditions reduce energy consumption and equipment wear, further lowering the operational expenditure associated with production.
- Enhanced Supply Chain Reliability: By utilizing commodity chemicals such as benzyl bromide and cyanoacetate derivatives as starting materials, the process mitigates the risk of supply disruptions often caused by specialized reagents. The robustness of the NHC catalysts and the tolerance of the reaction conditions allow for flexible manufacturing schedules, ensuring consistent delivery timelines even under fluctuating demand. This reliability is crucial for maintaining uninterrupted production lines for downstream API manufacturing.
- Scalability and Environmental Compliance: The method is explicitly designed for industrial scale-up, featuring simple post-treatment steps that generate minimal hazardous waste compared to traditional resolution or metal-heavy processes. The absence of toxic heavy metals in the final product simplifies regulatory compliance and reduces the burden of wastewater treatment. The ability to scale from grams to tons without compromising enantiomeric purity makes this technology an ideal candidate for long-term commercial partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on the feasibility and benefits of adopting this technology for your specific application needs.
Q: What are the advantages of this NHC-catalyzed method over traditional chiral resolution?
A: Traditional chiral resolution methods often suffer from a maximum theoretical yield of 50% and require multiple crystallization steps. This NHC-catalyzed asymmetric synthesis constructs the chiral center directly, avoiding the loss of half the raw material and eliminating the need for expensive chiral resolving agents or harsh metal reagents like LiHMDS.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly highlights the method's suitability for industrial production. It utilizes readily available starting materials like benzyl bromide and cyanoacetate derivatives, employs inexpensive Raney nickel for hydrogenation instead of precious metals, and features simple post-treatment procedures involving standard extraction and filtration.
Q: What level of enantiomeric purity can be achieved with this route?
A: The disclosed method demonstrates excellent stereocontrol. In the provided examples, the process achieved an enantiomeric ratio (e.r.) of up to 97:3, indicating high optical purity which is critical for pharmaceutical applications where impurity profiles are strictly regulated.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-3-Amino-2-benzyl Methyl Propionate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a stable and high-quality supply of chiral intermediates for your drug development pipelines. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with precision and speed. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. By leveraging advanced catalytic technologies like the NHC-mediated synthesis described in CN112209850A, we provide a competitive edge in the market through superior process efficiency and product consistency.
We invite you to engage with our technical procurement team to discuss how our manufacturing capabilities can align with your project goals. Whether you require a Customized Cost-Saving Analysis for your current supply chain or need to evaluate the feasibility of this specific route for your portfolio, we are ready to assist. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can drive value and innovation in your pharmaceutical projects.
