Scalable Metal-Free Synthesis of 4-Arylthiocoumarins for Advanced Pharmaceutical Intermediates
Scalable Metal-Free Synthesis of 4-Arylthiocoumarins for Advanced Pharmaceutical Intermediates
The landscape of heterocyclic chemistry is constantly evolving, driven by the demand for cleaner, more efficient synthetic routes that align with green chemistry principles while maintaining high structural diversity. A significant breakthrough in this domain is documented in patent CN114195818A, which discloses a robust and versatile preparation method for 4-thiocoumarin compounds. These sulfur-containing heterocycles are not merely academic curiosities; they serve as critical scaffolds in medicinal chemistry, offering unique physicochemical properties that can be fine-tuned by introducing various sulfur-containing groups. The patent outlines a general chemical framework where substituents R1, R2, and R3 can be extensively varied, encompassing hydrogen, alkyl, methoxy, halogens, cyano, nitro, aldehyde, and ester groups, thereby enabling the creation of a vast library of bioactive molecules. 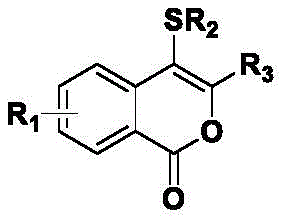 This structural flexibility is paramount for drug discovery teams aiming to optimize lead compounds for potency and metabolic stability. By providing a pathway that avoids harsh conditions and complex catalytic systems, this technology positions itself as a cornerstone for the next generation of reliable pharmaceutical intermediate supplier networks seeking to diversify their portfolios with high-value sulfur heterocycles.
This structural flexibility is paramount for drug discovery teams aiming to optimize lead compounds for potency and metabolic stability. By providing a pathway that avoids harsh conditions and complex catalytic systems, this technology positions itself as a cornerstone for the next generation of reliable pharmaceutical intermediate supplier networks seeking to diversify their portfolios with high-value sulfur heterocycles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-arylthio coumarin compounds has been plagued by reliance on stoichiometric amounts of transition metal catalysts and strong oxidants, which introduce significant bottlenecks in both process development and manufacturing. Traditional protocols often employ ferrous chloride to catalyze the reaction between 2-alkynylbenzoic acid methyl esters and disulfide compounds, or utilize dichloroiodobenzene as a promoter. While these methods achieve the desired transformation, they suffer from inherent drawbacks that are increasingly unacceptable in modern GMP environments. The presence of transition metals necessitates rigorous and costly purification steps to ensure residual metal levels meet stringent regulatory limits for pharmaceutical ingredients. Furthermore, the use of strong oxidants can lead to over-oxidation side reactions, generating complex impurity profiles that are difficult to separate, ultimately reducing the overall yield and purity of the final API intermediate. These factors collectively inflate the cost of goods sold (COGS) and extend the lead time for high-purity pharmaceutical intermediates, creating supply chain vulnerabilities for downstream drug manufacturers who require consistent quality and rapid turnaround.
The Novel Approach
In stark contrast to these legacy techniques, the methodology described in CN114195818A represents a paradigm shift towards metal-free organocatalysis, leveraging the unique solvation properties of fluorinated alcohols to drive the reaction forward. The core innovation lies in the reaction between 2-alkynyl methyl benzoate and N-aryl(alkyl)sulfenyl succinimide in a solvent system comprising hexafluoroisopropanol (HFIP), trifluoroethanol, or acetic acid. This approach completely eliminates the need for transition metal catalysts and external oxidants, fundamentally simplifying the reaction matrix. 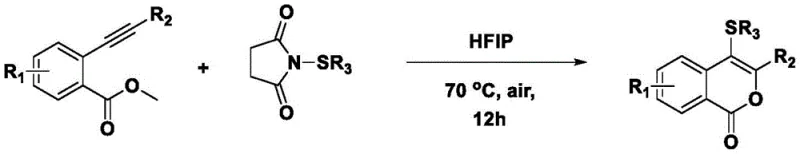 By removing these hazardous and contaminating reagents, the process not only enhances the environmental profile of the synthesis but also drastically reduces the complexity of the post-reaction workup. The absence of metal salts means that expensive scavenging resins or repeated recrystallizations are no longer required, directly translating to cost reduction in fine chemical manufacturing. Moreover, the mild conditions allow for a broader substrate scope, accommodating sensitive functional groups that might decompose under the harsh oxidative conditions of traditional methods, thus expanding the chemical space accessible to R&D teams.
By removing these hazardous and contaminating reagents, the process not only enhances the environmental profile of the synthesis but also drastically reduces the complexity of the post-reaction workup. The absence of metal salts means that expensive scavenging resins or repeated recrystallizations are no longer required, directly translating to cost reduction in fine chemical manufacturing. Moreover, the mild conditions allow for a broader substrate scope, accommodating sensitive functional groups that might decompose under the harsh oxidative conditions of traditional methods, thus expanding the chemical space accessible to R&D teams.
Mechanistic Insights into HFIP-Promoted Cyclization
The efficacy of this metal-free transformation is deeply rooted in the unique physicochemical properties of hexafluoroisopropanol (HFIP), which acts as more than just a passive solvent. HFIP is known for its high ionizing power and ability to stabilize cationic intermediates through strong hydrogen bonding networks, which is crucial for activating the electrophilic species in the reaction mixture. In this specific cyclization, the N-sulfenyl succinimide serves as an electrophilic sulfur source, attacking the electron-rich alkyne moiety of the 2-alkynyl benzoate. The HFIP solvent likely facilitates the formation of a sulfonium ion intermediate, which subsequently undergoes an intramolecular nucleophilic attack by the carbonyl oxygen of the ester group. This cascade leads to the formation of the coumarin lactone ring with the simultaneous expulsion of the succinimide leaving group. 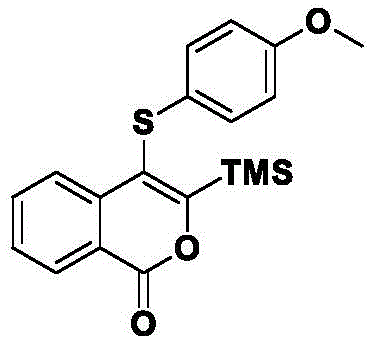 The precision of this mechanism ensures high regioselectivity, favoring the formation of the 4-substituted coumarin over other potential isomers. For R&D directors focused on impurity control, this mechanistic clarity is invaluable; it predicts a cleaner reaction profile with fewer byproducts, simplifying the analytical burden during process validation. The ability to tune the electronic nature of the sulfur source (via the N-sulfenyl succinimide) and the alkyne substrate allows for fine control over reaction kinetics, ensuring that even sterically hindered substrates can be converted efficiently.
The precision of this mechanism ensures high regioselectivity, favoring the formation of the 4-substituted coumarin over other potential isomers. For R&D directors focused on impurity control, this mechanistic clarity is invaluable; it predicts a cleaner reaction profile with fewer byproducts, simplifying the analytical burden during process validation. The ability to tune the electronic nature of the sulfur source (via the N-sulfenyl succinimide) and the alkyne substrate allows for fine control over reaction kinetics, ensuring that even sterically hindered substrates can be converted efficiently.
Furthermore, the tolerance of this system towards diverse functional groups is a testament to its mildness. The patent data demonstrates successful synthesis with substrates containing electron-donating groups like methoxy and methyl, as well as electron-withdrawing groups such as halogens (fluoro, chloro, bromo) and trimethylsilyl groups. This versatility implies that the reaction mechanism is robust against electronic perturbations, likely due to the powerful activation provided by the HFIP solvent environment. For process chemists, this means that a single set of optimized conditions—specifically heating at 70°C for 12 hours—can be applied to a wide array of derivatives without the need for extensive re-optimization. This standardization is key to accelerating the timeline from bench-scale discovery to pilot plant production, allowing for the rapid generation of analog libraries for structure-activity relationship (SAR) studies. The resulting products, such as the 4-(4-methoxyphenylthio)-3-trimethylsilylcoumarin shown, exhibit high structural integrity, ready for further downstream functionalization into sulfones or sulfoxides, thereby serving as versatile building blocks for complex molecule assembly.
How to Synthesize 4-Arylthiocoumarin Efficiently
The practical implementation of this synthesis route is designed for ease of execution, requiring standard laboratory equipment and readily available reagents. The protocol begins with the dissolution of the alkyne and sulfenyl succinimide precursors in HFIP, followed by a controlled heating phase that drives the cyclization to completion. This streamlined workflow minimizes operator intervention and reduces the risk of human error, which is critical for maintaining batch-to-batch consistency in a commercial setting. The simplicity of the procedure underscores its potential for immediate adoption in both academic research laboratories and industrial process development units seeking to upgrade their synthetic capabilities.
- Dissolve 2-alkynyl methyl benzoate and N-aryl(alkyl)sulfenyl succinimide in hexafluoroisopropanol (HFIP) with a molar ratio of approximately 1: 1.2.
- Heat the reaction mixture to 70°C and stir under air atmosphere for 12 hours to facilitate the cyclization.
- Remove the solvent, extract the residue with ethyl acetate and water, dry the organic phase, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this metal-free methodology offers tangible strategic advantages that extend beyond mere chemical elegance. The elimination of transition metal catalysts removes a significant cost center associated with both the purchase of expensive metal salts and the subsequent disposal or recovery of metal-laden waste streams. Additionally, the use of commercially available starting materials like methyl 2-iodobenzoate derivatives and thiophenols ensures a stable and resilient supply chain, mitigating the risk of raw material shortages that often plague specialized synthetic routes. The robustness of the reaction conditions, operating effectively at moderate temperatures and under air atmosphere, further reduces energy consumption and infrastructure requirements, contributing to substantial cost savings in pharmaceutical intermediates manufacturing. This combination of low operational complexity and high output reliability makes the technology an ideal candidate for long-term supply agreements.
- Cost Reduction in Manufacturing: The most immediate financial impact stems from the complete removal of transition metal catalysts and stoichiometric oxidants from the bill of materials. In traditional processes, the cost of palladium, iron, or iodine reagents, coupled with the specialized ligands often required, can be prohibitive at scale. By replacing these with inexpensive N-sulfenyl succinimides and recyclable fluorinated solvents, the direct material costs are significantly lowered. Furthermore, the downstream processing is vastly simplified; without heavy metals to remove, the need for expensive scavenging resins, activated carbon treatments, or multiple recrystallization steps is eliminated. This reduction in unit operations translates directly to lower labor costs, reduced solvent consumption, and shorter cycle times, driving down the overall cost per kilogram of the final intermediate. The simplified impurity profile also means higher first-pass yields, minimizing the loss of valuable material during purification and maximizing the return on investment for every batch produced.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of commodity-grade starting materials that are widely sourced from multiple global vendors. Unlike proprietary catalysts that may be subject to single-source supply risks or long lead times, the precursors for this reaction—such as alkynyl benzoates and thiophenols—are established chemicals with mature manufacturing infrastructures. This abundance ensures that procurement teams can negotiate better pricing and secure volume commitments with greater confidence. Moreover, the reaction's tolerance to air and moisture (operating under air atmosphere) reduces the need for specialized inert gas handling equipment and strictly anhydrous conditions, which simplifies logistics and storage requirements. This operational flexibility allows for more agile production scheduling, enabling suppliers to respond rapidly to fluctuating market demands without the bottleneck of complex setup procedures, thereby ensuring a continuous and reliable flow of high-purity intermediates to downstream customers.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this metal-free process aligns perfectly with the industry's push towards greener chemistry. The absence of toxic heavy metals like iron or iodine in the waste stream significantly reduces the environmental footprint of the manufacturing process, easing the burden on wastewater treatment facilities and lowering disposal costs. The use of HFIP, while a specialized solvent, can be managed through established recovery and recycling protocols, further enhancing the sustainability profile. Scalability is inherently supported by the simple thermal nature of the reaction; heating a large reactor to 70°C is a trivial engineering challenge compared to managing exothermic oxidations or handling pyrophoric catalysts on a multi-ton scale. This ease of scale-up de-risks the technology transfer from lab to plant, ensuring that commercial scale-up of complex pharmaceutical intermediates can be achieved with minimal technical hurdles and maximum safety, meeting the rigorous EHS standards expected by top-tier multinational corporations.
Frequently Asked Questions (FAQ)
To address common technical inquiries regarding the implementation and scope of this technology, we have compiled a set of answers based on the detailed experimental data provided in the patent literature. These insights are intended to clarify the operational parameters and potential applications of the 4-arylthiocoumarin synthesis, assisting technical teams in evaluating its fit for their specific project requirements.
Q: Does this synthesis method require transition metal catalysts?
A: No, unlike conventional methods utilizing ferrous chloride or dichloroiodobenzene, this novel protocol operates without transition metal catalysts or external oxidants, significantly simplifying downstream purification and eliminating heavy metal contamination risks.
Q: What is the optimal reaction temperature and time for maximum yield?
A: Experimental data indicates that heating the reaction system at 70°C for 12 hours provides the optimal balance between reaction kinetics and product stability, achieving yields up to 83% for specific substrates like 4-(4-methoxyphenylthio)-3-trimethylsilylcoumarin.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the method features simple operational steps, mild reaction conditions, and a straightforward workup procedure involving standard extraction and chromatography, making it highly amenable to scale-up from laboratory grams to multi-ton industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Arylthiocoumarin Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free cyclization technology for the production of high-value sulfur heterocycles. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from bench-scale discovery to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped to handle fluorinated solvents and sensitive intermediates with the utmost care, adhering to stringent purity specifications and rigorous QC labs to guarantee that every batch meets the highest international standards. We understand that in the competitive landscape of pharmaceutical development, time and quality are paramount, and our dedicated process chemistry team is ready to optimize this route specifically for your target molecules.
We invite you to leverage our expertise to accelerate your drug development timelines. By partnering with us, you gain access to a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this metal-free protocol for your specific portfolio. We encourage you to contact our technical procurement team today to request specific COA data for our reference standards and to discuss detailed route feasibility assessments tailored to your project's unique constraints. Let us help you secure a sustainable, cost-effective, and high-quality supply of 4-arylthiocoumarin intermediates for your next breakthrough therapy.
