High-Performance Oxaspiro Chiral Phosphoric Acid for Asymmetric Synthesis Manufacturing
Introduction to Next-Generation Chiral Catalysis
The landscape of asymmetric synthesis is continually evolving, driven by the demand for higher enantiopurity in active pharmaceutical ingredients (APIs). Patent CN113150031A introduces a breakthrough class of oxaspiro chiral phosphoric acids, characterized by the general structure shown in Formula (A). These catalysts represent a significant leap forward in Bronsted acid catalysis, offering exceptional stereocontrol for the construction of axially chiral biaryl-2-amine scaffolds. Unlike traditional BINOL-derived phosphoric acids, the oxaspiro framework imposes a unique steric environment that minimizes background reactions and maximizes enantioselectivity. Experimental data within the patent demonstrates that these catalysts consistently deliver products with enantiomeric excess (ee) values exceeding 97%, alongside high isolated yields. This level of performance is critical for reducing downstream purification costs in drug manufacturing. For procurement and R&D teams seeking reliable sources of advanced chiral building blocks, understanding the structural nuances and synthetic accessibility of these molecules is paramount.
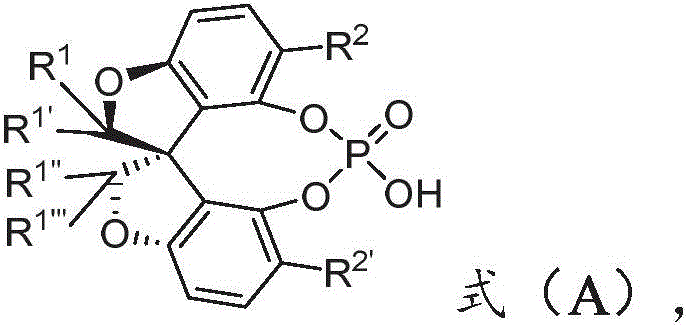
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of axially chiral biaryl compounds has relied heavily on resolution of racemates or the use of less efficient chiral auxiliaries, which often suffer from poor atom economy and limited substrate scope. Traditional chiral phosphoric acids, while effective in many transformations, can struggle with bulky substrates or fail to induce sufficient chirality in challenging C-C bond formations. Furthermore, the preparation of some high-performance chiral catalysts involves multi-step resolutions or expensive starting materials that hinder large-scale adoption. In industrial settings, the inability to consistently achieve >95% ee without extensive chromatographic purification creates significant bottlenecks in supply chains. These limitations necessitate a shift towards catalysts that combine rigid chiral pockets with straightforward synthetic routes, ensuring both high performance and economic viability for the production of complex pharmaceutical intermediates.
The Novel Approach
The invention detailed in CN113150031A addresses these challenges through a rational design of the oxaspiro backbone. By fusing an oxygen-containing spiro-cycle with the biaryl system, the catalyst achieves a locked conformation that precisely directs the approach of substrates during the transition state. This structural rigidity translates directly into superior stereochemical outcomes, as evidenced by the successful synthesis of diverse biaryl-2-amines with excellent yields and selectivity. The novelty also extends to the synthetic methodology, which employs a direct Rh-catalyzed C-H activation strategy to install the necessary aryl groups. This approach bypasses the need for pre-functionalized organometallic reagents, streamlining the catalyst preparation process. For manufacturers, this means a more robust supply of high-purity chiral phosphoric acids, facilitating cost reduction in chiral catalyst manufacturing and enabling the efficient production of high-value drug candidates.
Mechanistic Insights into Rh-Catalyzed Backbone Construction and Phosphorylation
The synthesis of the oxaspiro chiral phosphoric acid backbone is a masterpiece of modern organometallic chemistry, leveraging Rhodium-catalyzed C-H activation to forge the critical biaryl bond. As illustrated in the preparation route, the process begins with an oxaspiro diol precursor (Formula 1) which undergoes coupling with an aryl halide (R2X). This transformation is mediated by a specialized catalyst system comprising (1,5-cyclooctadiene) rhodium chloride dimer and bulky phosphine ligands such as di-tert-butylphosphine chloride. The reaction proceeds in toluene at elevated temperatures ranging from 150°C to 180°C under an inert argon atmosphere. This high-temperature regime is essential for overcoming the activation energy barrier of the C-H bond cleavage, ensuring complete conversion to the biaryl diol intermediate (Formula 2). The choice of base, typically cesium carbonate, plays a crucial role in neutralizing the hydrogen halide byproduct and maintaining the catalytic cycle. This step is fundamental to establishing the axial chirality that defines the catalyst's efficacy.
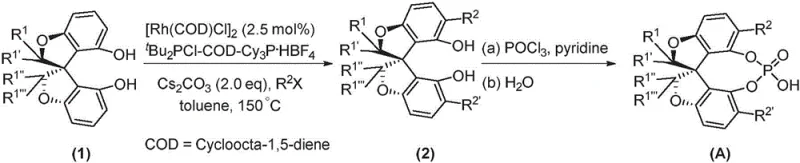
Following the construction of the chiral backbone, the installation of the phosphoric acid moiety is achieved through a classic phosphorylation sequence. The biaryl diol intermediate reacts with phosphorus oxychloride (POCl3) in the presence of pyridine, acting as both solvent and base, at temperatures between 70°C and 110°C. This step forms the cyclic phosphate ester, which is subsequently hydrolyzed under controlled aqueous conditions at 110°C to 120°C to yield the final free acid (Formula A). The mechanistic precision here ensures that the chiral information encoded in the backbone is preserved throughout the functionalization process. For R&D directors, understanding this mechanism highlights the robustness of the process; the use of standard reagents like POCl3 and pyridine suggests that the synthesis can be easily adapted to pilot plant scales without requiring exotic equipment. The resulting catalyst features a highly acidic P-OH proton capable of dual hydrogen-bond activation, which is the key driver for its exceptional performance in asymmetric transformations.
How to Synthesize Oxaspiro Chiral Phosphoric Acid Efficiently
The preparation of these high-value catalysts follows a streamlined two-stage protocol that balances chemical complexity with operational simplicity. The initial stage focuses on the assembly of the sterically demanding biaryl framework using transition metal catalysis, while the second stage involves the introduction of the phosphoric acid functionality. This modular approach allows for the easy variation of substituents (R groups) to tune the catalyst's electronic and steric properties for specific applications. Detailed procedural parameters, including precise stoichiometry, temperature ramps, and workup techniques, are critical for maximizing yield and optical purity. The following guide outlines the standardized synthesis steps derived from the patent embodiments, providing a clear roadmap for laboratory and pilot-scale production.
- Perform Rh-catalyzed C-H activation/arylation of the oxaspiro diol precursor with aryl halides using a Rh/COD catalyst system at 150-180°C.
- React the resulting biaryl diol intermediate with phosphorus oxychloride (POCl3) in pyridine at 70-110°C.
- Hydrolyze the phosphorylated intermediate at 110-120°C to yield the final oxaspiro chiral phosphoric acid.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of this oxaspiro chiral phosphoric acid technology offers distinct advantages over legacy catalytic systems. The synthetic route relies on commodity chemicals such as toluene, pyridine, and cesium carbonate, which are readily available in the global market, thereby mitigating risks associated with raw material scarcity. Furthermore, the reaction conditions, while requiring elevated temperatures, do not necessitate cryogenic cooling or ultra-high pressure equipment, simplifying the engineering requirements for commercial scale-up. This accessibility translates into a more stable supply chain for high-purity pharmaceutical intermediates, ensuring consistent delivery timelines for downstream API manufacturers. By utilizing a catalyst that delivers superior selectivity, companies can significantly reduce the burden on purification units, leading to substantial cost savings in overall manufacturing operations without compromising on product quality.
- Cost Reduction in Manufacturing: The high enantioselectivity (>97% ee) achieved by these catalysts drastically reduces the need for expensive chiral separation processes such as preparative HPLC or recrystallization. In traditional workflows, low selectivity often forces manufacturers to discard significant portions of the unwanted enantiomer, representing a direct loss of raw materials and processing time. By shifting the burden of selectivity to the catalyst, the overall material throughput is optimized. Additionally, the catalyst synthesis itself avoids the use of precious metal scavengers in the final application step, as it functions as an organocatalyst, further lowering the cost of goods sold (COGS) for the final pharmaceutical product.
- Enhanced Supply Chain Reliability: The precursors for the oxaspiro backbone, such as substituted phenols and aryl halides, are produced by established chemical industries with robust capacity. Unlike specialized chiral ligands that may have single-source suppliers, the building blocks for this technology are diversified. This diversity ensures that procurement managers can source materials from multiple vendors, reducing the risk of supply disruptions. Moreover, the stability of the final phosphoric acid catalyst allows for extended storage periods without significant degradation, enabling strategic stockpiling to buffer against market volatility and ensuring continuous production schedules for critical drug substances.
- Scalability and Environmental Compliance: The process utilizes toluene as the primary solvent, which is well-understood in terms of recovery and recycling protocols in modern chemical plants. The absence of heavy metal residues in the final catalytic application (since the Rh is only used in the catalyst prep, not the drug synthesis step where the phosphoric acid is used) simplifies regulatory compliance regarding residual metals in APIs. The reaction generates minimal hazardous waste compared to stoichiometric chiral auxiliary methods. This aligns with green chemistry principles, making it easier for facilities to meet increasingly stringent environmental regulations while scaling production from 100 kgs to 100 MT annual volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of oxaspiro chiral phosphoric acids in industrial processes. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on performance metrics and operational parameters. Understanding these details is essential for technical teams evaluating the feasibility of integrating this technology into existing manufacturing lines.
Q: What is the primary advantage of this oxaspiro chiral phosphoric acid over traditional catalysts?
A: The oxaspiro scaffold provides a rigid chiral environment that significantly enhances stereoselectivity, achieving enantiomeric excess (ee) values greater than 97% in the synthesis of axially chiral biaryl-2-amines, surpassing many conventional Bronsted acid catalysts.
Q: What are the typical reaction conditions for synthesizing the catalyst backbone?
A: The backbone is constructed via a Rhodium-catalyzed coupling reaction using (1,5-cyclooctadiene) rhodium chloride dimer and specific phosphine ligands in toluene at elevated temperatures (150-180°C) under an inert argon atmosphere.
Q: Can this catalyst be scaled for industrial production of pharmaceutical intermediates?
A: Yes, the synthesis utilizes robust conditions and commercially available solvents like toluene and pyridine. The process avoids cryogenic temperatures and uses standard workup procedures, making it highly suitable for commercial scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Oxaspiro Chiral Phosphoric Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced chiral catalysts like the oxaspiro phosphoric acids described in CN113150031A. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to full-scale manufacturing is seamless. Our facility is equipped with rigorous QC labs and stringent purity specifications capable of handling sensitive organocatalysts and complex intermediates. We understand that consistency is key in asymmetric synthesis, and our quality management systems are designed to guarantee batch-to-batch reproducibility, delivering the high enantiomeric excess required for modern drug development.
We invite you to collaborate with our technical procurement team to explore how this technology can optimize your specific synthesis routes. Whether you require a Customized Cost-Saving Analysis for your current process or need to evaluate the feasibility of new chiral scaffolds, our experts are ready to assist. Please contact us to request specific COA data and route feasibility assessments tailored to your project needs, ensuring you stay ahead in the competitive landscape of pharmaceutical innovation.
