Advanced Aqueous Synthesis of 3-Ethoxy-4-Methoxybenzaldehyde for Commercial Scale-Up
Advanced Aqueous Synthesis of 3-Ethoxy-4-Methoxybenzaldehyde for Commercial Scale-Up
The pharmaceutical industry is constantly seeking robust, scalable, and environmentally benign pathways for the production of critical active pharmaceutical ingredient (API) intermediates. A pivotal development in this domain is detailed in patent CN107827722B, which discloses a revolutionary synthetic method for 3-ethoxy-4-methoxybenzaldehyde, a key building block for the anti-psoriasis drug apremilast. This innovation addresses long-standing challenges in fine chemical manufacturing by replacing hazardous organic solvents and toxic alkylating agents with a green, aqueous-based system. By leveraging phase transfer catalysis (PTC) under mild conditions, this technology enables the production of high-purity intermediates with exceptional molar yields, positioning it as a cornerstone for modern, sustainable API manufacturing. For global procurement teams and R&D directors, understanding this shift from legacy chemistry to green engineering is essential for securing reliable supply chains and reducing regulatory risks associated with toxic reagent handling.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-ethoxy-4-methoxybenzaldehyde has relied on methodologies that pose significant operational and environmental hazards. Traditional routes frequently employ diethyl sulfate or dimethyl sulfate as ethylating or methylating agents, respectively. These reagents are not only highly toxic and carcinogenic but also require stringent safety protocols that increase operational overhead and liability. Furthermore, legacy processes often utilize polar aprotic solvents such as dimethylformamide (DMF). While effective for solubility, DMF presents severe downstream processing challenges; its high boiling point makes recovery energy-intensive, and its removal from the final product often requires complex extraction and distillation steps. As illustrated in prior art reaction schemes, these conventional approaches typically achieve yields ranging from 70% to 83%, leaving substantial room for improvement in atom economy and waste generation.
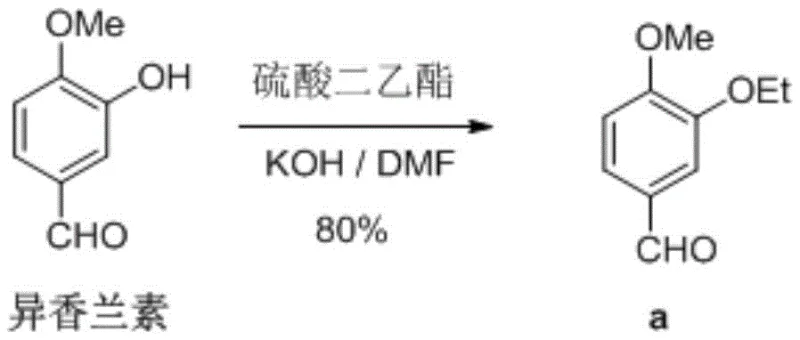
The Novel Approach
In stark contrast to these hazardous legacy methods, the patented technology introduces a streamlined, aqueous-phase alkylation strategy that fundamentally redefines the process safety profile. By utilizing water as the sole reaction medium and bromoethane as the alkylating source, the process eliminates the need for volatile organic compounds (VOCs) and carcinogenic sulfates. The reaction is facilitated by a phase transfer catalyst, which bridges the immiscible organic and aqueous phases, ensuring efficient mass transfer and rapid reaction kinetics at mild temperatures ranging from 0°C to 60°C. This approach not only simplifies the work-up procedure to a mere filtration step but also dramatically enhances the overall molar yield to over 95%. The transition from toxic sulfates to safer haloalkanes, combined with the elimination of difficult-to-remove solvents, represents a paradigm shift towards greener, more cost-effective pharmaceutical intermediate manufacturing.
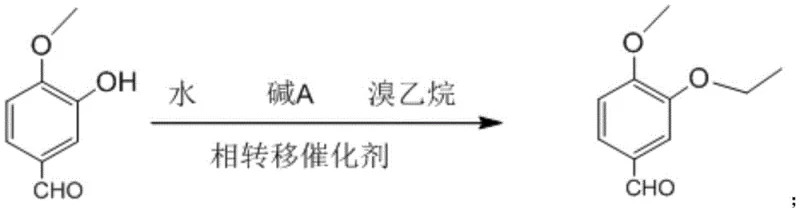
Mechanistic Insights into Aqueous Phase Transfer Catalysis
The core of this technological breakthrough lies in the efficient application of phase transfer catalysis (PTC) within an aqueous alkaline environment. In this system, a strong base such as sodium hydroxide or potassium carbonate deprotonates the phenolic hydroxyl group of isovanillin to form a phenoxide anion. However, since the organic substrate and the alkylating agent (bromoethane) have limited solubility in water, the reaction would normally be kinetically sluggish. The addition of a quaternary ammonium salt, such as tetrabutylammonium bromide, acts as a molecular shuttle. The lipophilic cation of the catalyst transports the reactive phenoxide anion from the aqueous phase into the organic phase where it encounters the bromoethane. This intimate contact facilitates a rapid nucleophilic substitution (SN2) reaction, forming the ethoxy ether bond with high specificity. The mild thermal conditions (25°C in preferred embodiments) prevent thermal degradation of the aldehyde functionality, ensuring the structural integrity of the sensitive intermediate.
From an impurity control perspective, this mechanism offers distinct advantages over high-temperature or strongly acidic alternatives. The use of water as a solvent provides excellent heat capacity, preventing localized hot spots that could lead to polymerization or oxidation byproducts. Furthermore, the stoichiometric control afforded by the PTC system minimizes over-alkylation or side reactions on the aldehyde group. The result is a crude product with purity levels often exceeding 99.0% by HPLC directly after filtration. This high level of intrinsic purity reduces the burden on downstream purification units, such as recrystallization or column chromatography, thereby lowering the overall cost of goods sold (COGS) and minimizing solvent waste generation. For R&D teams, this implies a more robust process window where minor fluctuations in reaction parameters do not compromise the quality of the final API intermediate.
How to Synthesize 3-Ethoxy-4-Methoxybenzaldehyde Efficiently
Implementing this synthesis route requires precise attention to reagent ratios and mixing dynamics to maximize the benefits of the phase transfer system. The process is designed to be operationally simple, avoiding the need for specialized high-pressure reactors or inert atmosphere gloveboxes, which makes it highly accessible for standard multipurpose chemical plants. The following overview outlines the critical operational parameters derived from the patent examples, emphasizing the simplicity of the work-up procedure which is a major driver for cost reduction.
- Dissolve a strong base such as sodium hydroxide or potassium carbonate in water within a reaction vessel to create an alkaline aqueous medium.
- Add isovanillin, a phase transfer catalyst like tetrabutylammonium bromide, and bromoethane to the mixture under stirring conditions.
- Maintain the reaction temperature between 0-60°C for 3-6 hours, then perform suction filtration to isolate the high-purity solid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this aqueous PTC methodology translates into tangible strategic advantages beyond mere chemical elegance. The elimination of hazardous reagents like diethyl sulfate removes a significant bottleneck in raw material sourcing and storage, as these substances often require specialized containment and transport logistics. By switching to bromoethane and common inorganic bases, the supply chain becomes more resilient and less susceptible to regulatory restrictions on toxic chemicals. Moreover, the use of water as a solvent drastically reduces the volume of organic waste requiring incineration or treatment, aligning production with increasingly stringent global environmental regulations and corporate sustainability goals.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the simplification of downstream processing. Traditional methods involving DMF require energy-intensive distillation to recover the solvent and extensive washing to remove trace residues, which inflates utility costs and extends batch cycle times. In this novel aqueous process, the product precipitates or can be isolated via simple filtration, bypassing the need for complex solvent recovery systems. Additionally, the high molar yield exceeding 95% ensures better utilization of raw materials, effectively lowering the cost per kilogram of the intermediate. The avoidance of expensive, toxic alkylating agents further contributes to a leaner cost structure, allowing for competitive pricing in the global API market.
- Enhanced Supply Chain Reliability: Reliability in the supply of critical intermediates like 3-ethoxy-4-methoxybenzaldehyde is paramount for uninterrupted API production. This method utilizes commodity chemicals—sodium hydroxide, bromoethane, and water—which are widely available from multiple global suppliers, reducing the risk of single-source dependency. The robustness of the reaction conditions, which do not require cryogenic cooling or high vacuum, means that production can be maintained even during periods of utility fluctuation. This operational stability ensures consistent delivery schedules, mitigating the risk of stockouts that could halt downstream drug manufacturing lines.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces unforeseen challenges, particularly regarding heat management and waste disposal. This aqueous protocol is inherently scalable because water acts as an excellent heat sink, managing the exotherm of the alkylation reaction safely even in large-scale reactors. The "green" nature of the process, characterized by the absence of chlorinated solvents and toxic sulfates, simplifies the permitting process for new manufacturing sites. Facilities can achieve higher throughput with lower environmental footprints, ensuring long-term compliance with evolving environmental, social, and governance (ESG) standards without the need for costly retrofits to waste treatment infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, aiming to clarify the practical benefits for potential partners and licensees.
Q: Why is the aqueous PTC method superior to traditional DMF-based alkylation?
A: Traditional methods often rely on polar aprotic solvents like DMF, which are difficult to remove and recover, leading to complex post-treatment and higher environmental costs. The patented aqueous method eliminates the need for organic solvents, allowing for simple filtration and significantly reducing three-waste treatment complexity.
Q: Does this process avoid the use of highly toxic sulfating agents?
A: Yes, unlike prior art routes that utilize diethyl sulfate or dimethyl sulfate—substances known for their extreme toxicity and carcinogenic potential—this novel protocol employs bromoethane as the alkylating agent, drastically improving operational safety and environmental compliance.
Q: What purity levels can be achieved with this synthesis route?
A: The method consistently delivers product with HPLC purity exceeding 99.0%, often reaching 99.9% without the need for complex recrystallization or chromatographic purification, making it ideal for direct use in sensitive pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Ethoxy-4-Methoxybenzaldehyde Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to greener, more efficient synthesis routes is critical for the future competitiveness of the pharmaceutical supply chain. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patented aqueous method are fully realized in a GMP-compliant manufacturing environment. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 3-ethoxy-4-methoxybenzaldehyde meets the exacting standards required for psoriasis therapeutic applications. Our commitment to quality assurance ensures that our clients receive intermediates that facilitate smooth downstream processing and final drug approval.
We invite global pharmaceutical partners to collaborate with us to leverage this advanced technology for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that optimize both cost and supply security for your critical API programs.
