Advanced One-Step Synthesis of Brominated 1,4-Naphthoquinone for Commercial Scale-Up
Introduction to High-Efficiency Brominated Naphthoquinone Synthesis
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for more efficient, cost-effective, and environmentally sustainable synthetic routes. A significant breakthrough in this domain is documented in Chinese Patent CN108069845B, which details a novel one-step preparation method for brominated 1,4-naphthoquinones. This technology addresses the longstanding challenges associated with synthesizing these critical scaffolds, which are ubiquitous in natural products and bioactive molecules. Traditionally, accessing these structures required laborious multi-step sequences involving the separate construction of the quinone core followed by halogenation. The disclosed innovation fundamentally shifts this paradigm by enabling a direct, telescoped transformation from a readily available dinitro-naphthalene precursor. For R&D directors and process chemists, this represents a substantial opportunity to streamline development timelines, while supply chain leaders will recognize the inherent value in a process that reduces unit operations and potential failure points. As a reliable pharmaceutical intermediate supplier, understanding and leveraging such patented methodologies is crucial for maintaining a competitive edge in the global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of brominated naphthoquinones was fraught with inefficiencies and selectivity issues. Literature surveys reveal a fragmented landscape of methods, each with distinct drawbacks that hinder commercial viability. For instance, early approaches involved the oxidation of dinaphthols using superoxides or the oxygen oxidation of tetralones under basic conditions, which often suffered from harsh reaction conditions and moderate yields. More critically, the direct bromination of existing naphthoquinone skeletons frequently led to poor regioselectivity. As illustrated in the prior art, attempting to brominate polyhydroxy naphthoquinones or simple naphthols often resulted in over-bromination, yielding unwanted dibromo or tetrabromo byproducts rather than the desired mono- or di-brominated targets. Specifically, direct bromination of 2,6-dihydroxynaphthalene derivatives typically fails to stop at the desired stage, producing complex mixtures that are difficult to purify. Furthermore, some legacy methods rely on expensive or hazardous reagents, such as specific iodobenzene dipoles or specialized oxidants, which escalate the cost of goods sold (COGS) and complicate waste management protocols. These conventional pathways, characterized by their multi-step nature and lack of precision, create significant bottlenecks in the manufacturing of high-purity OLED materials or API intermediates.
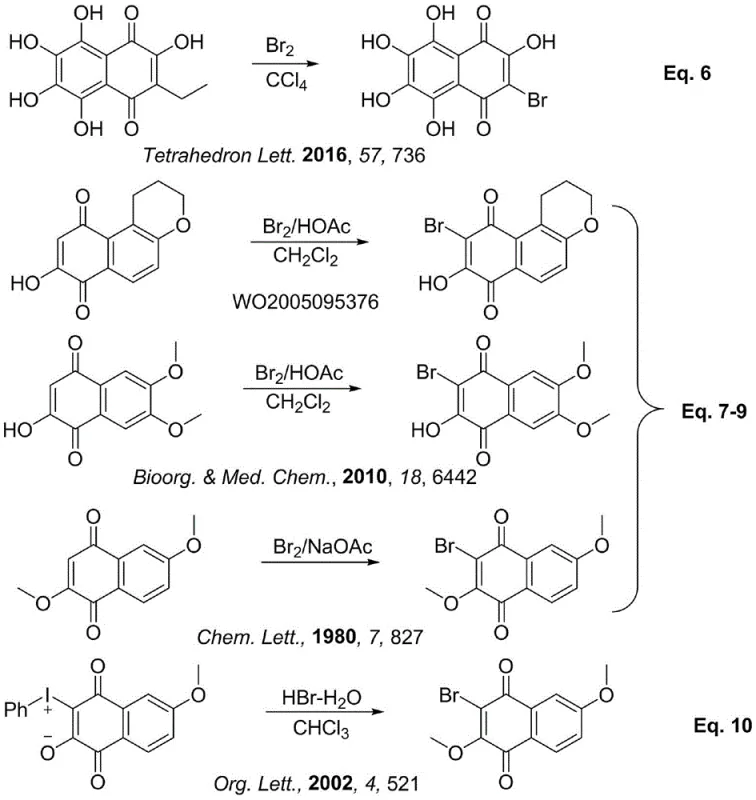
The Novel Approach
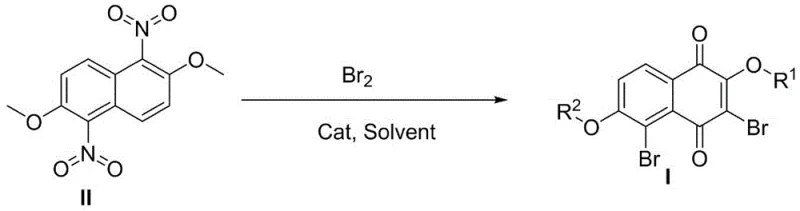
In stark contrast to the fragmented prior art, the method disclosed in CN108069845B offers a unified, high-efficiency solution. The core of this innovation lies in the utilization of 1,5-dinitro-2,6-dimethoxynaphthalene as a strategic precursor. This specific substitution pattern activates the naphthalene ring in a unique way, allowing for simultaneous bromination and oxidative demethylation/oxidation in a single reactor vessel. Instead of isolating a naphthoquinone intermediate and then subjecting it to a separate halogenation step, this process achieves both transformations concurrently. The reaction proceeds smoothly in the presence of a catalyst, such as anhydrous ferric chloride, and a solvent like glacial acetic acid. This one-pot strategy not only drastically simplifies the operational workflow but also enhances the overall yield by minimizing material loss associated with intermediate isolation and purification. The ability to directly access 3,5-dibromo-2-hydroxy-6-methoxy-1,4-naphthoquinone or its fully methoxylated analogues from a common dinitro starting material provides a versatile platform for derivative synthesis. This approach effectively bypasses the regioselectivity pitfalls of direct naphthol bromination, ensuring that the bromine atoms are installed precisely at the 3 and 5 positions, which is critical for downstream coupling reactions in drug discovery.
Mechanistic Insights into FeCl3-Catalyzed Oxidative Bromination
The success of this one-step transformation hinges on the intricate interplay between the electron-withdrawing nitro groups, the Lewis acidic catalyst, and the oxidizing capability of the bromine source. Mechanistically, the presence of the two nitro groups at the 1 and 5 positions of the naphthalene ring significantly deactivates the aromatic system towards electrophilic attack, yet paradoxically, they facilitate the specific oxidative cleavage required to form the quinone moiety. The ferric chloride catalyst serves a dual purpose: it acts as a Lewis acid to activate the bromine molecule for electrophilic substitution and potentially assists in the hydrolysis or oxidation of the methoxy groups to carbonyls under the reflux conditions. The reaction temperature, optimized between 60°C and 120°C, provides the necessary thermal energy to overcome the activation barrier for the oxidative cyclization while maintaining control over the bromination extent. Crucially, the nitro groups act as protecting-directing groups; they prevent the uncontrolled poly-bromination observed in simpler naphthalene systems by modulating the electron density of the ring. This ensures that bromination occurs selectively at the activated positions ortho to the methoxy groups, which are subsequently oxidized. The result is a clean conversion to the 1,4-naphthoquinone core with bromine atoms strategically placed for further functionalization. This mechanistic elegance allows for the production of complex halogenated quinones without the need for protecting group strategies that would otherwise add steps and cost to the synthesis.
From an impurity control perspective, this mechanism offers distinct advantages over traditional routes. In conventional direct bromination, the lack of directing groups often leads to a statistical distribution of brominated species, including tribromo and tetrabromo impurities that are structurally similar and difficult to remove. In the patented process, the electronic bias imposed by the 1,5-dinitro pattern inherently suppresses the formation of these over-brominated side products. The reaction pathway is channeled towards the formation of the 3,5-dibromo species as the thermodynamic and kinetic major product. Furthermore, the use of glacial acetic acid as a solvent aids in solubilizing the polar intermediates while providing a mildly acidic environment that stabilizes the quinone product against degradation. The final workup, involving simple cooling and filtration, leverages the low solubility of the product in cold acetic acid to achieve high purity without the need for extensive chromatography. This robustness in impurity profile is a key selling point for regulatory filings, where controlling genotoxic impurities and closely related substances is paramount for API manufacturers.
How to Synthesize 3,5-Dibromo-2-hydroxy-6-methoxy-1,4-naphthoquinone Efficiently
Implementing this synthesis requires careful attention to the preparation of the starting material and the precise control of reaction parameters. The process begins with the quantitative nitration of 2,6-dimethoxynaphthalene to generate the essential 1,5-dinitro-2,6-dimethoxynaphthalene intermediate. Once this precursor is secured, the one-pot conversion can be executed with high reproducibility. The protocol involves suspending the dinitro compound in glacial acetic acid with a catalytic amount of anhydrous ferric chloride, followed by heating to initiate the reaction. Subsequent addition of liquid bromine drives the halogenation and oxidation sequence to completion. The detailed standardized synthesis steps, including specific molar ratios, temperature profiles, and workup procedures, are outlined below to ensure successful replication in a pilot or production setting.
- Quantitatively prepare the key intermediate 1,5-dinitro-2,6-dimethoxynaphthalene from 2,6-dimethoxynaphthalene via nitration.
- Perform a one-pot reaction mixing the dinitro intermediate with anhydrous ferric chloride and glacial acetic acid, followed by the addition of liquid bromine.
- Reflux the mixture at 60°C to 120°C until conversion is complete, then cool to precipitate the product, filter, and wash with glacial acetic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology translates into tangible strategic benefits that extend beyond mere chemical curiosity. The consolidation of multiple synthetic steps into a single pot operation fundamentally alters the cost structure of manufacturing these specialized intermediates. By eliminating the isolation and purification of the intermediate naphthoquinone, the process reduces solvent consumption, labor hours, and equipment occupancy time. This telescoping effect leads to significantly reduced operational expenditures, making the final brominated product more price-competitive in a margin-sensitive market. Moreover, the reliance on commodity chemicals like ferric chloride, glacial acetic acid, and liquid bromine ensures that the raw material supply chain is robust and less susceptible to the volatility associated with exotic reagents or precious metal catalysts. This stability is crucial for long-term supply agreements with multinational pharmaceutical clients who demand continuity of supply.
- Cost Reduction in Manufacturing: The elimination of intermediate isolation steps drastically cuts down on processing time and solvent usage, which are major cost drivers in fine chemical synthesis. By avoiding the need for column chromatography in the standard workup—relying instead on simple filtration and washing—the process minimizes waste generation and disposal costs. The use of inexpensive iron-based catalysts instead of noble metals further lowers the input cost, allowing for substantial cost savings in API manufacturing without compromising on quality. This economic efficiency enables suppliers to offer more competitive pricing models while maintaining healthy margins.
- Enhanced Supply Chain Reliability: The starting material, 2,6-dimethoxynaphthalene, is commercially available and can be sourced from multiple vendors, reducing the risk of single-source dependency. The simplicity of the reaction setup, which does not require specialized high-pressure equipment or cryogenic conditions, means that the process can be easily transferred between different manufacturing sites or scaled up rapidly to meet surges in demand. This flexibility ensures that lead times for high-purity pharmaceutical intermediates can be kept short and predictable, safeguarding the production schedules of downstream drug manufacturers against unexpected disruptions.
- Scalability and Environmental Compliance: The one-pot nature of the reaction inherently reduces the environmental footprint by minimizing the volume of waste solvents and aqueous streams generated per kilogram of product. The straightforward workup procedure, which avoids complex extractions or distillations, simplifies the engineering controls required for scale-up from laboratory grams to multi-tonne commercial batches. This ease of scale-up facilitates the commercial production of complex polymer additives or electronic chemicals that require similar halogenated quinone motifs. Additionally, the high atom economy of incorporating bromine directly into the scaffold reduces the burden on waste treatment facilities, aligning with increasingly stringent global environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity on the feasibility and scope of the technology. Understanding these nuances is essential for technical teams evaluating the integration of this method into their existing manufacturing portfolios.
Q: What is the primary advantage of the method in CN108069845B?
A: The primary advantage is the telescoping of bromination and oxidation into a single one-pot reaction step, which eliminates the need for isolating the naphthoquinone skeleton prior to halogenation, thereby simplifying the workflow and reducing waste.
Q: Which catalyst system is preferred for this transformation?
A: The patent specifies anhydrous ferric chloride (FeCl3) as the preferred catalyst, typically used in amounts ranging from 17 mol% to 50 mol% relative to the substrate, often in conjunction with glacial acetic acid as both solvent and promoter.
Q: How does this method control over-bromination compared to prior art?
A: By utilizing the 1,5-dinitro-2,6-dimethoxynaphthalene precursor, the electronic properties of the ring direct bromination specifically to the 3 and 5 positions, avoiding the formation of tetra-brominated byproducts common in direct bromination of unsubstituted naphthols.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Brominated 1,4-Naphthoquinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced synthetic methodologies like the one described in CN108069845B. Our team of expert process chemists is dedicated to translating such innovative laboratory protocols into robust, GMP-compliant manufacturing processes. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop to plant floor is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of brominated naphthoquinone delivered meets the exacting standards required for pharmaceutical and electronic applications. We understand that consistency and quality are non-negotiable in the fine chemical industry, and our commitment to excellence is reflected in our comprehensive quality assurance systems.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific project needs. Whether you require custom synthesis of novel derivatives or large-scale supply of standard intermediates, our technical procurement team is ready to assist. Please contact us to request a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to reach out for specific COA data and route feasibility assessments to determine how this efficient one-step process can optimize your supply chain and reduce your overall manufacturing costs.
