Advanced Synthetic Route for High-Purity Benzoxazolinone Herbicide Intermediates
The global agrochemical industry is currently witnessing a paradigm shift towards ultra-efficient, low-toxicity herbicides that minimize environmental impact while maximizing crop yield protection. A pivotal development in this sector is detailed in patent CN101270117B, which discloses a novel class of benzoxazolinone compounds exhibiting potent protoporphyrinogen oxidase (PPO) inhibitory activity. Unlike traditional organophosphorus pesticides that pose significant ecological risks, these new heterocyclic structures operate by competitively binding to the active site of the protox enzyme, disrupting chlorophyll synthesis and leading to rapid weed desiccation. For R&D directors and procurement specialists seeking next-generation active ingredients, this technology represents a critical opportunity to diversify product portfolios with high-efficacy solutions. The patent outlines a robust, multi-step synthetic methodology that transforms simple substituted acetophenones into complex, biologically active pyrazolyl-benzoxazolinones, addressing the urgent market demand for sustainable weed control agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of PPO inhibitor herbicides has been plagued by complex reaction pathways that often require hazardous reagents and generate substantial toxic waste. Conventional routes frequently rely on harsh nitration conditions that lack regioselectivity, leading to difficult-to-separate isomeric byproducts that compromise the purity of the final active ingredient. Furthermore, many legacy processes utilize expensive noble metal catalysts for reduction steps, which not only inflate the raw material costs but also introduce stringent requirements for heavy metal removal to meet regulatory standards for agricultural residues. The structural rigidity of older generations of herbicides often limits their efficacy against evolving resistant weed species, necessitating higher application dosages that contradict modern sustainability goals. These operational inefficiencies create bottlenecks in the supply chain, resulting in longer lead times and volatile pricing for key agrochemical intermediates.
The Novel Approach
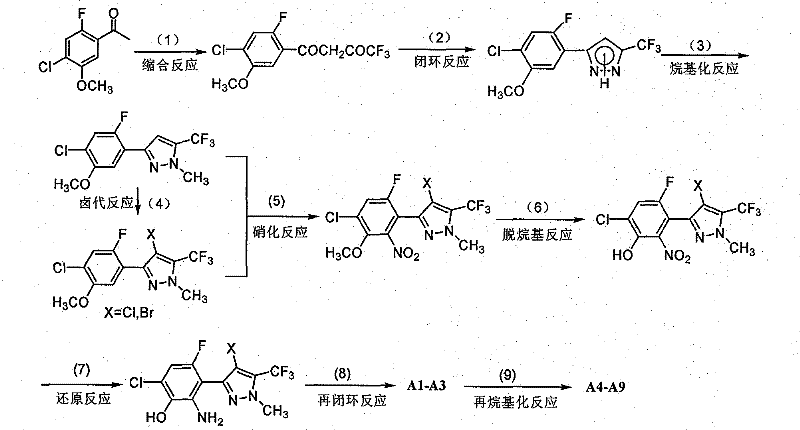
The methodology presented in the patent data introduces a streamlined, modular synthesis strategy that overcomes these historical hurdles through precise control of reaction conditions and substituent positioning. By initiating the sequence with a Claisen-type condensation of 4-chloro-2-fluoro-5-methoxyacetophenone, the process establishes the carbon framework with high atom economy before constructing the nitrogen-containing heterocycles. This approach allows for the systematic introduction of fluorine and chlorine atoms at specific positions on the phenyl ring, which are critical for enhancing metabolic stability and binding affinity to the target enzyme. The use of triphosgene for the final ring-closing step offers a safer and more controllable alternative to phosgene gas, facilitating easier handling in large-scale reactors. Consequently, this novel route delivers a series of compounds, such as A8 and A9, which demonstrate exceptional weed control rates of 50% to 100% across various species at relatively low dosages, validating the superior biological performance of this chemical architecture.
Mechanistic Insights into Pyrazole-Benzoxazolinone Construction
The core of this synthetic innovation lies in the sequential construction of the pyrazole ring followed by the fusion of the benzoxazolinone moiety, a process that requires meticulous management of electronic effects and steric hindrance. The initial cyclization involves the reaction of a trifluoro-beta-diketone intermediate with hydrazine hydrate, where the nucleophilic attack of the hydrazine nitrogen on the carbonyl carbon drives the formation of the five-membered pyrazole ring. Subsequent alkylation with dimethyl sulfate or methyl iodide selectively protects the nitrogen atom, preventing unwanted side reactions during the aggressive nitration phase that follows. The nitration step is particularly crucial, as it introduces the nitro group ortho to the methoxy functionality, setting the stage for the subsequent transformation into the amino precursor required for ring closure. This sequence ensures that the electron-withdrawing trifluoromethyl group on the pyrazole ring stabilizes the intermediate species, allowing the reaction to proceed with high yields even under vigorous acidic conditions.
Following the nitration, the demethylation and reduction steps serve as the bridge to the final bicyclic system, utilizing Lewis acids like aluminum trichloride and reducing agents such as iron powder to reveal the reactive amine and phenol functionalities. The final cyclization employs triphosgene as a carbonyl source, reacting with the ortho-amino phenol intermediate to close the oxazolone ring efficiently. This mechanism avoids the formation of urea byproducts often seen with other carbonylating agents, thereby simplifying the downstream purification process. The ability to fine-tune the R-group at the 3-position of the benzoxazolinone ring through a final alkylation step provides immense flexibility, enabling the production of a diverse library of analogs tailored for specific agronomic needs. This mechanistic precision is what distinguishes the process as a viable candidate for commercial scale-up of complex agrochemical intermediates.
How to Synthesize Benzoxazolinone Herbicide Efficiently
The synthesis of these high-value herbicidal compounds is achieved through a carefully orchestrated nine-step sequence that balances reaction efficiency with operational safety. The process begins with the condensation of substituted acetophenone derivatives, followed by cyclization to form the pyrazole core, which serves as the structural anchor for the molecule. Detailed procedural parameters, including specific molar ratios, solvent choices like toluene or DMF, and temperature controls ranging from ice-bath cooling to reflux conditions, are critical for maximizing yield at each stage. For a comprehensive understanding of the exact operational protocols required to replicate this high-purity synthesis in a GMP-compliant environment, please refer to the standardized guide below.
- Perform condensation of substituted acetophenone with ethyl trifluoroacetate followed by cyclization with hydrazine hydrate to form the pyrazole core.
- Execute alkylation and subsequent halogenation using N-halosuccinimide to introduce necessary substituents on the pyrazole ring.
- Complete the synthesis via nitration, demethylation, nitro reduction, and final ring closure with triphosgene to yield the benzoxazolinone structure.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this synthetic route offers compelling advantages that directly address the pain points of cost volatility and supply discontinuity often faced by agrochemical manufacturers. The reliance on commodity chemicals such as substituted acetophenones and common solvents like ethanol and ethyl acetate ensures that the raw material supply chain is robust and less susceptible to geopolitical disruptions. By eliminating the need for exotic catalysts and utilizing standard unit operations like filtration and recrystallization, the process significantly lowers the barrier to entry for contract manufacturing organizations. This accessibility translates into a more competitive pricing structure for the final active ingredient, allowing formulators to offer cost-effective solutions to farmers without compromising on performance. Furthermore, the modular nature of the synthesis allows for flexible production scheduling, where intermediates can be stockpiled and converted to final products based on seasonal demand fluctuations.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of iron powder for reduction drastically reduces the cost of goods sold while simplifying waste treatment protocols. The high yields reported in the patent examples, often exceeding 80% for key steps, minimize raw material wastage and maximize throughput per batch. Additionally, the avoidance of cryogenic conditions or high-pressure hydrogenation equipment reduces capital expenditure requirements for reactor infrastructure. These factors collectively contribute to a leaner manufacturing model that enhances profit margins for producers of herbicide intermediates.
- Enhanced Supply Chain Reliability: The utilization of stable, shelf-stable reagents like triphosgene instead of gaseous phosgene mitigates safety risks and logistical challenges associated with hazardous material transport. The synthesis tolerates a variety of solvents, providing procurement teams with the flexibility to source materials locally based on availability and price fluctuations. This adaptability ensures consistent production output even when specific supply lines are constrained, thereby securing the continuity of supply for downstream formulation partners. The robustness of the chemical transformations also reduces the likelihood of batch failures, further stabilizing the delivery schedule.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, with reaction conditions that can be easily translated from laboratory glassware to industrial-scale stainless steel reactors without significant re-optimization. The waste streams generated are primarily organic solvents and inorganic salts, which can be managed through established recovery and treatment systems, aligning with increasingly stringent environmental regulations. The high selectivity of the reactions minimizes the generation of toxic byproducts, reducing the burden on effluent treatment plants. This environmental compatibility is a crucial asset for manufacturers aiming to maintain their social license to operate in a regulated industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these benzoxazolinone derivatives. The answers are derived directly from the experimental data and structural analysis provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers. Understanding these nuances is essential for evaluating the feasibility of integrating this technology into existing production lines or product development pipelines.
Q: What is the primary advantage of this benzoxazolinone synthesis route?
A: The route utilizes readily available substituted acetophenone starting materials and avoids expensive transition metal catalysts, significantly simplifying purification and reducing production costs.
Q: How does this compound compare to existing PPO inhibitors?
A: The novel structure features a specific substitution pattern (2-fluoro, 4-chloro on the phenyl ring) that demonstrates superior herbicidal activity against resistant weeds like green bristlegrass and abutilon.
Q: Is the process scalable for industrial production?
A: Yes, the reaction conditions utilize standard solvents like toluene and ethanol, and reagents like iron powder and triphosgene, making it highly suitable for commercial scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzoxazolinone Herbicide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this patented synthetic route in reshaping the landscape of modern weed control. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from pilot scale to full manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of agrochemical intermediate delivered meets the highest international standards. We are committed to supporting your R&D efforts by providing custom synthesis services that optimize this chemistry for your specific formulation requirements.
We invite you to engage with our technical procurement team to discuss how we can tailor this synthesis to your volume needs and cost targets. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of adopting this route for your supply chain. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your next generation of herbicide products reaches the market with speed and reliability.
