Scalable Nickel-Catalyzed Synthesis of Fluorinated Isoquinoline-1,3-Diones for Commercial Production
Introduction to Advanced Fluorinated Heterocycle Synthesis
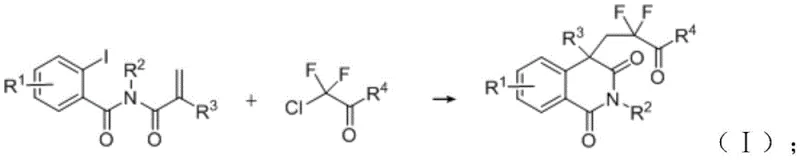
The strategic incorporation of fluorine atoms into heterocyclic scaffolds represents a cornerstone of modern medicinal chemistry, significantly enhancing the metabolic stability and lipophilicity of drug candidates. Patent CN114832862A introduces a groundbreaking catalytic composition specifically designed for the efficient preparation of isoquinoline-1,3-dione compounds, a privileged structure found in numerous bioactive natural products and pharmaceutical agents. This technology addresses the critical industry need for robust, scalable methods to access these valuable fluorinated motifs without relying on prohibitively expensive noble metals or harsh reaction conditions. By leveraging a specialized nickel-based catalytic system, this invention enables the direct introduction of difluoroalkyl groups into the isoquinoline core through a streamlined cross-coupling and cyclization sequence. For R&D directors and procurement specialists alike, this development signals a shift towards more economically viable and operationally simple manufacturing protocols for high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of fluorinated isoquinoline-1,3-diones has been plagued by significant technical and economic hurdles that hinder large-scale adoption. Prior art, such as the visible-light photocatalytic methods reported by Wang's team in 2017, often requires specialized irradiation equipment and suffers from limited penetration depth, making kilogram-scale production challenging and energy-intensive. Furthermore, earlier palladium-catalyzed difluoroalkylation strategies, while effective on small scales, impose a heavy financial burden due to the high cost of palladium precursors and the absolute necessity for rigorous downstream purification to remove toxic metal residues to ppm levels. These conventional routes frequently operate under strict anhydrous conditions or require elevated temperatures that can degrade sensitive functional groups, leading to narrow substrate scopes and inconsistent batch-to-batch reproducibility. Consequently, the reliance on these legacy technologies results in inflated production costs and extended lead times, creating bottlenecks for supply chain managers tasked with securing reliable sources of complex heterocyclic building blocks.
The Novel Approach
In stark contrast, the methodology disclosed in CN114832862A utilizes a cost-effective nickel catalytic system that operates under remarkably mild conditions, typically around 60°C in polar aprotic solvents like DMSO. This novel approach employs a synergistic combination of a nickel bromide catalyst, dual ligands (specifically L3 and DPPE), and a magnesium chloride cocatalyst to drive the reductive coupling of o-iodoacryloylbenzamides with alpha-chloro-difluoro esters. The use of manganese powder as a stoichiometric reductant further simplifies the reaction setup, eliminating the need for external electrical currents or high-energy photon sources. This catalytic composition demonstrates exceptional tolerance for various substituents on the aromatic ring and the amide nitrogen, allowing for the rapid generation of diverse chemical libraries essential for drug discovery campaigns. By replacing precious metals with abundant nickel and utilizing standard heating mantles instead of photoreactors, this technology drastically lowers the barrier to entry for manufacturing these high-purity intermediates.
Mechanistic Insights into Nickel-Catalyzed Reductive Cyclization
The efficacy of this transformation relies on a sophisticated catalytic cycle where the nickel center undergoes precise oxidation state changes to facilitate bond formation. Initially, the active Ni(0) species, generated in situ from the Ni(II) precursor and manganese reductant, undergoes oxidative addition into the carbon-iodine bond of the o-iodoacryloylbenzamide substrate. This key step forms an aryl-nickel(II) intermediate which is stabilized by the bidentate phosphine and nitrogen-containing ligands, preventing premature decomposition or aggregation of the metal center. Subsequently, the difluoroalkyl radical, generated via single-electron transfer or halogen atom abstraction from the alpha-chloro-difluoro ester, adds to the activated alkene moiety or interacts directly with the nickel center to form a new carbon-carbon bond. The resulting organonickel species then undergoes migratory insertion and subsequent reductive elimination to close the isoquinoline ring, regenerating the active catalyst for the next turnover. This mechanistic pathway is highly selective, effectively suppressing competing homocoupling side reactions that often plague radical-based cross-coupling processes.
Impurity control in this system is inherently managed by the specific choice of the MgCl2 cocatalyst and the dual-ligand environment, which modulates the electronic properties of the nickel center to favor the desired cyclization pathway. The presence of magnesium ions likely assists in the activation of the carbon-chlorine bond in the difluoro reagent or stabilizes the transition state during the ring-closing step, thereby minimizing the formation of open-chain byproducts or dehalogenated starting materials. Furthermore, the mild reaction temperature of 60°C prevents thermal degradation of the sensitive difluoro ester functionality, ensuring that the final product retains its structural integrity and fluorine content. For quality assurance teams, this means a cleaner crude reaction profile that simplifies downstream purification, requiring less aggressive chromatography conditions and reducing solvent consumption. The robustness of this mechanism across different substrates, including those with electron-withdrawing or electron-donating groups, underscores its reliability for producing consistent, high-quality batches of isoquinoline-1,3-dione derivatives.
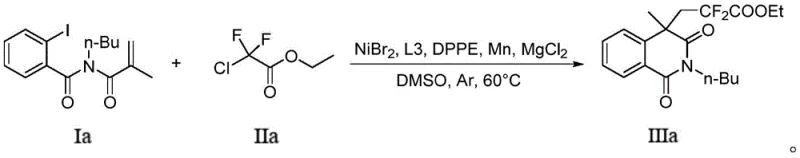
How to Synthesize Ethyl 4,4-difluoro-2-(n-butyl)-1,3-dioxo-1,2,3,4-tetrahydroisoquinoline-4-acetate Efficiently
To demonstrate the practical application of this patented technology, we focus on the synthesis of a representative ethyl ester derivative, which serves as a versatile intermediate for further functionalization. The procedure outlined in the patent exemplifies the operational simplicity of the method, requiring only standard Schlenk techniques and common laboratory reagents to achieve respectable yields. The reaction leverages the specific molar ratios defined in the invention, ensuring optimal catalyst turnover and substrate conversion while minimizing waste. Detailed below is the strategic overview of the synthesis, highlighting the critical parameters that must be controlled to replicate the success reported in the intellectual property documentation.
- Charge a Schlenk bottle with NiBr2 catalyst, L3 and DPPE ligands, Mn reducing agent, and MgCl2 cocatalyst under inert atmosphere.
- Add o-iodoacryloylbenzamide substrate and difluoro compound in DMSO solvent, then heat to 60°C for 24 hours.
- Quench with saturated ammonium chloride, extract with ethyl acetate, and purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this nickel-catalyzed protocol offers transformative benefits for organizations seeking to optimize their manufacturing spend and secure resilient supply chains for critical intermediates. The substitution of palladium with nickel represents a direct and substantial reduction in raw material costs, as nickel salts are orders of magnitude cheaper and more abundant than their noble metal counterparts. This cost efficiency is compounded by the elimination of expensive photocatalytic equipment and the associated energy costs, allowing production facilities to utilize existing standard reactor infrastructure without capital expenditure upgrades. Moreover, the use of commercially available ligands and simple inorganic additives like magnesium chloride ensures that the supply chain for reagents is robust and unlikely to face geopolitical or logistical disruptions. For procurement managers, this translates to a more predictable cost structure and reduced vulnerability to price volatility in the precious metals market.
- Cost Reduction in Manufacturing: The economic advantage of this process is primarily driven by the replacement of high-cost palladium catalysts with inexpensive nickel bromide, which significantly lowers the bill of materials for every kilogram of product produced. Additionally, the mild reaction conditions reduce energy consumption compared to high-temperature or high-pressure alternatives, contributing to lower utility costs over the lifecycle of the manufacturing campaign. The simplified workup procedure, which avoids complex metal scavenging steps required for palladium removal, further reduces processing time and the consumption of specialized purification resins. These cumulative savings allow for a more competitive pricing strategy for the final API intermediate, enhancing margin potential for downstream pharmaceutical products.
- Enhanced Supply Chain Reliability: By utilizing reagents that are commodity chemicals rather than specialized proprietary catalysts, manufacturers can source materials from multiple global suppliers, mitigating the risk of single-source dependency. The stability of the catalytic system allows for longer shelf-life of prepared reaction mixtures if necessary, providing flexibility in production scheduling and inventory management. Furthermore, the broad substrate scope means that a single standardized protocol can be adapted for various analogues, reducing the need for extensive process re-validation when switching between different product grades. This flexibility ensures continuous supply even when specific starting materials face temporary shortages, as the process can be quickly adjusted to alternative substrates within the defined scope.
- Scalability and Environmental Compliance: The process is inherently scalable because it avoids the light-penetration limitations of photochemistry, allowing for the use of large-volume stirred tank reactors without loss of efficiency. The use of manganese as a reductant generates benign inorganic byproducts that are easier to treat and dispose of compared to the heavy metal waste streams associated with traditional cross-coupling methods. Operating at 60°C in DMSO also aligns well with green chemistry principles by minimizing volatile organic compound emissions and reducing the thermal load on facility cooling systems. These factors collectively simplify the environmental permitting process and reduce the overall ecological footprint of the manufacturing operation, aligning with increasingly stringent global sustainability mandates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this nickel-catalyzed synthesis platform. These answers are derived directly from the experimental data and technical specifications provided in the patent literature to ensure accuracy and relevance for industry stakeholders. Understanding these details is crucial for process chemists evaluating the feasibility of technology transfer and for business leaders assessing the strategic value of this manufacturing route.
Q: Why is nickel preferred over palladium for this isoquinoline-1,3-dione synthesis?
A: Nickel catalysts are significantly more cost-effective than palladium and exhibit higher oxidative addition activity due to lower electronegativity, allowing for milder reaction conditions and broader substrate tolerance without expensive metal removal steps.
Q: What are the critical reaction conditions for maximizing yield in this coupling?
A: Optimal yields are achieved at 60°C in DMSO solvent using a specific ligand combination of L3 and DPPE, with magnesium chloride as a crucial cocatalyst to enhance nickel activity and manganese powder as the terminal reductant.
Q: Can this process be scaled for industrial production of pharmaceutical intermediates?
A: Yes, the process utilizes readily available reagents, operates at moderate temperatures (60°C), and avoids sensitive photochemical equipment or high-pressure systems, making it highly suitable for commercial scale-up in standard reactor vessels.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isoquinoline-1,3-dione Supplier
At NINGBO INNO PHARMCHEM, we recognize the pivotal role that advanced catalytic technologies play in accelerating drug development timelines and reducing time-to-market for novel therapeutics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory benchtop to industrial reactor is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of isoquinoline-1,3-dione intermediate meets the exacting standards required by global regulatory agencies. Our commitment to quality and consistency makes us a trusted partner for pharmaceutical companies seeking to secure their supply of complex fluorinated heterocycles.
We invite you to engage with our technical procurement team to discuss how this innovative nickel-catalyzed route can be integrated into your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this methodology for your pipeline candidates. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your unique molecular targets, ensuring that your supply chain is optimized for both performance and profitability.
