Scalable Green Synthesis of Heterocyclic Aryl Ketones for Pharmaceutical Intermediates Manufacturing
The pharmaceutical and agrochemical industries are constantly seeking sustainable pathways to access high-value carbonyl scaffolds, which serve as critical building blocks for bioactive molecules. Patent CN109134173B, published in March 2021, introduces a transformative approach to synthesizing heterocyclic aryl ketone compounds through a direct, metal-free aerobic oxidation strategy. This technology represents a significant departure from classical methodologies that rely heavily on toxic stoichiometric oxidants and expensive transition metal catalysts. By leveraging molecular oxygen as the terminal oxidant in a polar solvent system, specifically dimethyl sulfoxide (DMSO), this invention achieves the selective functionalization of benzylic Csp3-H bonds. For R&D directors and process chemists, this offers a compelling alternative that simplifies purification workflows and minimizes environmental footprint. The ability to convert simple benzyl heterocycles directly into polysubstituted ketones in a single step underlines the potential for streamlining complex synthetic routes used in the production of active pharmaceutical ingredients (APIs) and advanced intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the preparation of heterocyclic ketone compounds, particularly those containing nitrogen heterocycles, has been fraught with significant operational and environmental challenges. Conventional synthetic routes typically depend on the use of equivalent amounts of toxic and harmful oxidants, such as chromium(VI) reagents, manganese dioxide, or organic peroxides, which generate substantial quantities of hazardous waste streams. Furthermore, many established protocols necessitate the presence of transition metal catalysts to activate the C-H bonds, introducing the risk of heavy metal residues in the final product. For a reliable heterocyclic aryl ketone supplier, removing these trace metals to meet stringent pharmaceutical purity specifications often requires additional, costly purification steps like scavenging or recrystallization. The accumulation of byproducts from stoichiometric oxidants not only complicates downstream processing but also poses severe disposal issues, driving up the overall cost of goods and creating supply chain bottlenecks related to waste management compliance.
The Novel Approach
In stark contrast, the novel approach disclosed in the patent utilizes a green, atom-economical strategy that eliminates the need for exogenous metal catalysts and hazardous oxidants. The core innovation lies in the use of molecular oxygen, an abundant and inexpensive reagent, to drive the oxidative transformation directly. As illustrated in the general reaction scheme below, benzyl heterocyclic compounds are heated in a polar solvent under an oxygen atmosphere to yield the desired ketone products with high efficiency. 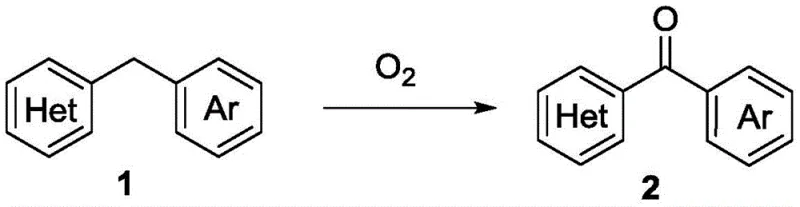 This metal-free protocol not only reduces the raw material costs associated with catalyst procurement but also drastically simplifies the workup procedure, as there are no metal salts to filter or extract. The reaction conditions are relatively mild, typically ranging from 100°C to 160°C, which enhances the safety profile of the process by avoiding the thermal hazards associated with concentrated peroxide solutions. This shift towards aerobic oxidation represents a paradigm shift in cost reduction in pharmaceutical intermediate manufacturing, aligning perfectly with modern green chemistry principles.
This metal-free protocol not only reduces the raw material costs associated with catalyst procurement but also drastically simplifies the workup procedure, as there are no metal salts to filter or extract. The reaction conditions are relatively mild, typically ranging from 100°C to 160°C, which enhances the safety profile of the process by avoiding the thermal hazards associated with concentrated peroxide solutions. This shift towards aerobic oxidation represents a paradigm shift in cost reduction in pharmaceutical intermediate manufacturing, aligning perfectly with modern green chemistry principles.
Mechanistic Insights into DMSO-Promoted Aerobic Oxidation
The mechanistic pathway proposed for this transformation involves a radical-mediated process facilitated by the unique properties of the DMSO solvent. Under thermal conditions, the benzylic methylene group of the heterocyclic substrate undergoes homolytic cleavage or hydrogen atom transfer to generate a reactive methylene radical species. This radical intermediate subsequently captures molecular oxygen to form a superoxide radical adduct. The subsequent steps involve proton abstraction and the elimination of a water molecule to furnish the final carbonyl functionality. This mechanism highlights the dual role of DMSO, acting not merely as a solvent but as a promoter that stabilizes radical intermediates and facilitates the activation of the C-H bond. Understanding this mechanism is crucial for process optimization, as it explains the high selectivity observed for benzylic positions over other potentially oxidizable sites on the heterocyclic ring. The absence of metal centers means that the reaction trajectory is governed purely by electronic and steric factors inherent to the substrate and the solvent system, offering a predictable and robust reaction profile for scale-up.
Furthermore, the impurity profile generated by this metal-free oxidation is significantly cleaner compared to traditional methods. Without transition metals, there is no risk of metal-catalyzed side reactions such as over-oxidation to carboxylic acids or unwanted coupling reactions that often plague metal-mediated processes. The primary byproduct is water, which is easily removed during the aqueous workup or drying stages. Experimental data indicates that the reaction tolerates a wide variety of functional groups, including electron-withdrawing nitro and chloro substituents, as well as electron-donating methoxy groups, without compromising yield. For instance, the oxidation of 4-benzylpyridine proceeds smoothly to give the corresponding ketone in 87% yield, demonstrating the efficacy of the system even with basic nitrogen heterocycles. 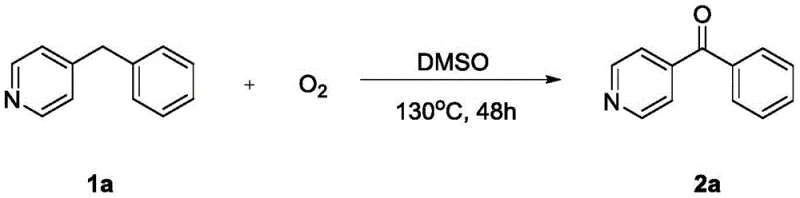 This tolerance is vital for the synthesis of complex drug candidates where orthogonal protecting group strategies might otherwise be required. Additionally, control experiments shown in the patent reveal that substrates lacking the activating heterocyclic ring or possessing aliphatic chains do not react under these conditions, underscoring the chemoselectivity of the method.
This tolerance is vital for the synthesis of complex drug candidates where orthogonal protecting group strategies might otherwise be required. Additionally, control experiments shown in the patent reveal that substrates lacking the activating heterocyclic ring or possessing aliphatic chains do not react under these conditions, underscoring the chemoselectivity of the method. 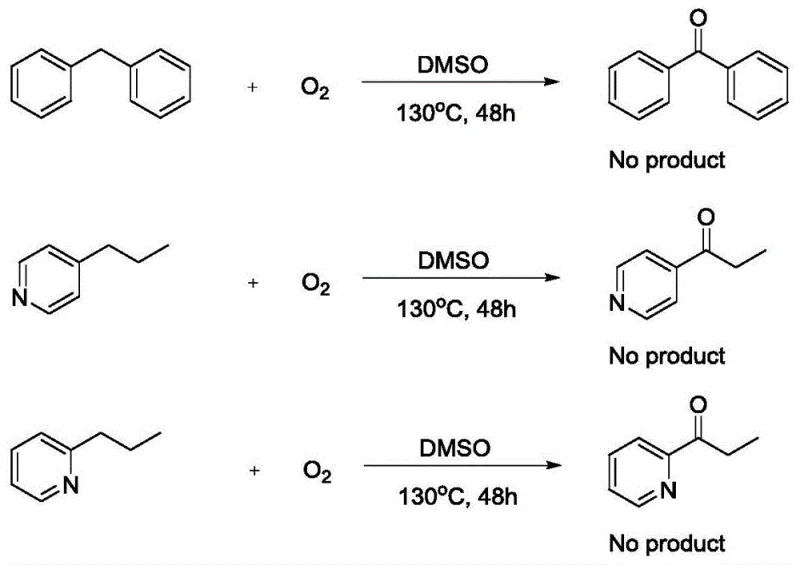 This selectivity ensures that sensitive functionalities elsewhere in a molecule remain intact, preserving the structural integrity of advanced intermediates.
This selectivity ensures that sensitive functionalities elsewhere in a molecule remain intact, preserving the structural integrity of advanced intermediates.
How to Synthesize Heterocyclic Aryl Ketones Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to reaction parameters to maximize yield and safety. The general procedure involves charging a reaction vessel with the benzyl heterocyclic substrate and dimethyl sulfoxide, followed by establishing an oxygen atmosphere. The mixture is then heated to the optimized temperature, typically between 130°C and 140°C, and stirred for a prolonged period, often ranging from 48 to 60 hours, to ensure complete conversion. Following the reaction, the mixture is cooled, quenched with water, and extracted with organic solvents like ethyl acetate. The detailed standardized synthesis steps for specific substrates are outlined in the guide below.
- Mix the benzyl heterocyclic substrate with dimethyl sulfoxide (DMSO) solvent in a reaction vessel under an oxygen atmosphere.
- Heat the reaction mixture to a temperature between 100°C and 160°C and stir continuously for 24 to 60 hours to promote C-H bond oxidation.
- Upon completion, cool the mixture, quench with water, extract with ethyl acetate, dry over sodium sulfate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this aerobic oxidation technology offers tangible benefits that extend beyond mere chemical elegance. The elimination of transition metal catalysts removes a major cost driver associated with precious metal procurement and the subsequent analytical testing required to certify low residual metal levels. This simplification of the supply chain reduces the dependency on volatile metal markets and mitigates the risk of supply disruptions caused by geopolitical factors affecting mining outputs. Moreover, the use of molecular oxygen as the oxidant replaces expensive and hazardous stoichiometric reagents, leading to substantial cost savings in raw material expenditure. The simplified workup procedure, which avoids complex filtration or chelation steps, translates directly into reduced processing time and lower labor costs per kilogram of product. These factors collectively contribute to a more resilient and cost-effective manufacturing process for high-purity heterocyclic aryl ketones.
- Cost Reduction in Manufacturing: The removal of transition metal catalysts and toxic stoichiometric oxidants fundamentally alters the cost structure of the synthesis. By avoiding expensive palladium, copper, or iron catalysts, the direct material costs are significantly lowered. Furthermore, the absence of heavy metals eliminates the need for specialized scavenger resins or extensive purification protocols designed to meet strict regulatory limits for metal residues in APIs. This streamlining of the downstream processing phase reduces solvent consumption and waste disposal fees, resulting in a leaner and more economical production cycle that enhances overall profit margins for large-scale manufacturing operations.
- Enhanced Supply Chain Reliability: The reagents required for this transformation, namely benzyl heterocycles, DMSO, and oxygen, are commodity chemicals with robust and stable global supply chains. Unlike specialized ligands or air-sensitive catalysts that may have long lead times or single-source suppliers, these inputs are readily available from multiple vendors. This abundance ensures continuous production capability and reduces the risk of batch delays due to material shortages. The stability of the reaction conditions also allows for flexible scheduling and inventory management, enabling manufacturers to respond quickly to fluctuating market demands for key pharmaceutical intermediates without being constrained by fragile supply networks.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process is inherently safer and more scalable than traditional oxidation methods. The use of oxygen gas, while requiring appropriate safety controls, avoids the thermal runaway risks associated with handling large quantities of organic peroxides. The generation of water as the primary byproduct aligns with green chemistry metrics, minimizing the volume of hazardous waste that requires treatment. This environmental compatibility facilitates easier permitting for new production lines and supports corporate sustainability goals. The straightforward nature of the reaction allows for seamless scale-up from gram-scale laboratory experiments to multi-ton commercial production, ensuring consistent quality and yield across different batch sizes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this aerobic oxidation technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the scope and limitations of the method. Understanding these details is essential for evaluating the feasibility of integrating this route into existing manufacturing portfolios.
Q: Does this synthesis method require transition metal catalysts?
A: No, the method described in patent CN109134173B operates without transition metal catalysts, utilizing DMSO and molecular oxygen to drive the oxidation, which significantly reduces heavy metal contamination risks.
Q: What is the primary oxidant used in this green synthesis route?
A: Molecular oxygen (O2) serves as the terminal oxidant, making the process environmentally friendly and cost-effective compared to traditional stoichiometric oxidants like peroxides or chromates.
Q: What types of heterocyclic substrates are compatible with this oxidation protocol?
A: The protocol demonstrates broad substrate scope, successfully oxidizing benzyl pyridines, benzothiazoles, benzimidazoles, and xanthene derivatives to their corresponding ketones with high selectivity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Heterocyclic Aryl Ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of adopting green and efficient synthetic methodologies like the one described in CN109134173B. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are successfully translated into robust industrial processes. We are committed to delivering high-purity heterocyclic aryl ketones that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to handle metal-free oxidations and manage oxygen-mediated reactions safely positions us as a preferred partner for companies seeking to optimize their supply chains for next-generation pharmaceutical intermediates.
We invite you to collaborate with us to evaluate the potential of this technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this aerobic oxidation route. Please contact us to request specific COA data for relevant ketone intermediates and to discuss route feasibility assessments tailored to your target molecules. By partnering with us, you gain access to a reliable supply of complex fine chemicals produced with a focus on sustainability, quality, and cost-efficiency.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
