Advanced One-Step Stereoselective Synthesis of Trifluoromethyl Pentenone Derivatives for Pharmaceutical Applications
Advanced One-Step Stereoselective Synthesis of Trifluoromethyl Pentenone Derivatives for Pharmaceutical Applications
The landscape of organic synthesis is constantly evolving to meet the rigorous demands of modern drug discovery, particularly regarding the incorporation of fluorine motifs which enhance metabolic stability and bioavailability. A significant breakthrough in this domain is documented in Chinese Patent CN110698313B, which discloses a highly efficient, one-step preparation method for (Z)-4-trifluoromethyl-5-sulfanyl-4-pentenone derivatives. This technology represents a paradigm shift from traditional multi-step sequences, utilizing a visible-light-driven photoredox catalytic system to achieve remote C-H bond activation and trifluoromethylation simultaneously. For R&D directors and process chemists, this patent offers a compelling solution to the longstanding challenge of selectively functionalizing internal alkynes without triggering unwanted cyclization pathways. The ability to construct these complex fluorinated scaffolds under mild conditions opens new avenues for generating diverse libraries of bioactive molecules, positioning this methodology as a critical asset for any organization seeking a reliable pharmaceutical intermediate supplier capable of delivering high-value building blocks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the selective oxidative synthesis of hydrocarbons into oxygen-containing organic products has been plagued by significant thermodynamic and kinetic hurdles. Traditional approaches often necessitate the use of high temperatures and pressures or stoichiometric amounts of aggressive oxidants to overcome the high activation energy of C-H bonds in saturated hydrocarbons. These harsh conditions frequently lead to poor selectivity, where target products such as ketones or aldehydes are thermodynamically unstable and prone to further deep oxidation, resulting in carboxylic acids or complete mineralization. Furthermore, many conventional radical addition strategies involving alkynes suffer from a lack of control, often undergoing atom migration and subsequent cyclization to form cyclic compounds rather than the desired linear olefin addition products. This tendency towards cyclization severely limits the utility of such reactions for synthesizing open-chain fluorinated pentenones, forcing chemists to rely on longer, less efficient synthetic routes that increase waste and cost.
The Novel Approach
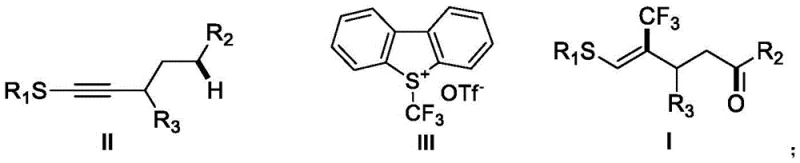
In stark contrast to these legacy methods, the technology described in CN110698313B introduces a sophisticated radical addition-hydrogen migration-oxidation tandem reaction that effectively bypasses cyclization. By employing S-(trifluoromethyl)dibenzothiophenium trifluoromethanesulfonate as a trifluoromethylating reagent and tris(2,2'-bipyridine)ruthenium(II) dichloride as a photocatalyst, the system achieves precise control over the reaction trajectory. The use of dimethyl sulfoxide (DMSO) serves a dual purpose as both the reaction medium and the terminal oxidant, facilitating the conversion of the intermediate radical species into the final ketone functionality under ambient blue LED irradiation. This approach not only realizes the trans-hydrogen trifluoromethylation of non-terminal alkynes but also ensures the exclusive formation of the (Z)-alkene geometry with a remote functional group. The operational simplicity of mixing reagents at room temperature eliminates the need for specialized high-pressure equipment, marking a substantial improvement in process safety and scalability.
Mechanistic Insights into Photoredox-Catalyzed Remote C-H Activation
The core innovation of this methodology lies in its unique mechanistic pathway, which diverges from standard radical alkyne functionalization. Upon irradiation with blue light, the ruthenium photocatalyst enters an excited state, initiating the single-electron transfer processes required to generate the trifluoromethyl radical from the dibenzothiophenium salt. This electrophilic radical adds regioselectively to the alkyne thioether substrate, forming a vinyl radical intermediate. Crucially, instead of undergoing the typical 5-exo-trig cyclization observed in many radical cascades, this intermediate undergoes a 1,5-hydrogen atom transfer (HAT) from a remote sp3 C-H bond. This step generates a carbon-centered radical at the delta-position relative to the sulfur atom, setting the stage for the subsequent oxidation event. The DMSO solvent then acts as the hydrogen acceptor and oxidant, converting this alkyl radical into the corresponding carbonyl group while regenerating the catalytic cycle. This intricate dance of radical intermediates ensures that the reaction terminates at the ketone stage, preserving the olefinic double bond and the stereochemical integrity of the molecule.
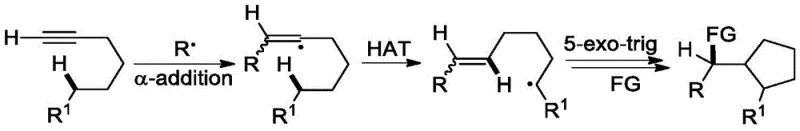
From an impurity control perspective, this mechanism offers distinct advantages by minimizing the formation of cyclic byproducts that are notoriously difficult to separate from linear analogs. The high regioselectivity of the initial radical addition, driven by the electronic properties of the thioether group, ensures that the trifluoromethyl group is installed at the correct position adjacent to the sulfur moiety. Furthermore, the mild oxidative conditions provided by DMSO prevent the over-oxidation of the sulfide group to sulfoxides or sulfones, which are common side reactions in stronger oxidative environments. The result is a clean reaction profile where the primary impurities are typically unreacted starting materials or minor regioisomers, both of which can be easily removed via standard silica gel chromatography. This high level of chemical fidelity is essential for pharmaceutical applications where strict limits on genotoxic impurities and structural analogs must be maintained throughout the supply chain.
How to Synthesize (Z)-4-trifluoromethyl-5-sulfanyl-4-pentenone Efficiently
The practical implementation of this synthesis is remarkably straightforward, designed to be accessible even in laboratories without specialized high-pressure infrastructure. The general protocol involves combining the alkyne thioether substrate with the trifluoromethylating reagent, the ruthenium catalyst, and sodium bicarbonate as a base in anhydrous DMSO. The reaction mixture is stirred under a nitrogen atmosphere at room temperature while being irradiated with a standard 20W blue LED lamp for approximately 8 hours. Following the completion of the reaction, indicated by TLC or LC-MS analysis, the workup procedure is simple and robust. The reaction is quenched with water, and the product is extracted into ethyl acetate. After washing the organic phase with brine and drying over anhydrous sodium sulfate, the crude material is purified by flash column chromatography to yield the pure (Z)-4-trifluoromethyl-5-sulfanyl-4-pentenone derivative. Detailed standardized synthesis steps are provided in the guide below.
- Mix alkyne thioether, S-(trifluoromethyl)dibenzothiophenium triflate, Ru(bpy)3Cl2 catalyst, and NaHCO3 in DMSO under nitrogen.
- Irradiate the reaction mixture with 20W blue LEDs at room temperature (25°C) for 8 hours to initiate radical addition and oxidation.
- Quench with water, extract with ethyl acetate, wash, dry over sodium sulfate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this photoredox methodology translates into tangible strategic benefits regarding cost structure and supply reliability. The elimination of harsh reaction conditions significantly reduces the energy consumption associated with heating and cooling cycles, leading to a lower overall carbon footprint and reduced utility costs per kilogram of product. Moreover, the use of DMSO as a dual-purpose solvent and oxidant simplifies the bill of materials by removing the need for separate, potentially hazardous oxidizing agents like peroxides or chromates. This consolidation of reagents not only lowers raw material costs but also streamlines inventory management and reduces the regulatory burden associated with storing and transporting dangerous goods. The mild nature of the reaction also extends the lifespan of reactor vessels and piping by minimizing corrosion, thereby reducing capital expenditure on maintenance and equipment replacement over the long term.
- Cost Reduction in Manufacturing: The process achieves cost optimization primarily through the simplification of the synthetic route. By accomplishing the trifluoromethylation and oxidation in a single pot, the need for intermediate isolation and purification steps is completely eradicated. This telescoping of operations drastically reduces solvent usage, labor hours, and processing time. Additionally, the high atom economy of the radical addition means that a larger proportion of the starting mass ends up in the final product, minimizing waste disposal fees. The avoidance of expensive transition metals beyond the catalytic amount of ruthenium further contributes to a leaner cost structure, making the final API intermediate more price-competitive in the global market.
- Enhanced Supply Chain Reliability: The robustness of this reaction conditions enhances supply chain resilience by reducing the risk of batch failures due to thermal runaways or sensitivity to moisture and oxygen. Since the reaction proceeds at room temperature, it is less susceptible to fluctuations in utility supply, such as steam pressure variations, which can plague high-temperature processes. The wide substrate scope demonstrated in the patent, accommodating various aryl and alkyl groups, allows for flexible sourcing of starting materials. If a specific alkyne thioether becomes unavailable, alternative substrates can often be substituted without re-optimizing the entire process, ensuring continuous production flow and mitigating the risk of stockouts for downstream customers.
- Scalability and Environmental Compliance: Scaling photochemical reactions has historically been challenging, but the use of simple LED arrays makes this process highly amenable to scale-up in flow reactors or large batch vessels with adequate light penetration. The environmental profile is superior to traditional methods, as the primary byproduct is the reduced form of the dibenzothiophenium salt, which is easier to handle than heavy metal waste. The absence of volatile organic solvents other than DMSO and ethyl acetate simplifies solvent recovery and recycling operations. This alignment with green chemistry principles facilitates easier regulatory approval and supports corporate sustainability goals, making the manufacturing process more attractive to environmentally conscious partners and investors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this trifluoromethylation technology. These answers are derived directly from the experimental data and claims within the patent documentation to ensure accuracy and relevance for technical decision-makers. Understanding these nuances is critical for evaluating the feasibility of integrating this route into existing manufacturing pipelines.
Q: What is the primary advantage of this photoredox method over traditional oxidation?
A: Unlike traditional methods requiring harsh temperatures or strong oxidants that lead to over-oxidation, this method uses mild blue light irradiation and DMSO as a dual solvent/oxidant, ensuring high selectivity for the ketone product without degrading the sensitive olefin structure.
Q: Does this reaction require expensive transition metal removal steps?
A: The process utilizes a ruthenium catalyst at low loading (0.01-0.05 equivalents). While standard purification like column chromatography is used, the mild conditions minimize metal complexation issues often seen in high-temperature processes, simplifying downstream processing compared to heavy metal-catalyzed alternatives.
Q: What is the stereoselectivity of the resulting alkene?
A: The reaction specifically yields the (Z)-isomer of the 4-trifluoromethyl-5-sulfanyl-4-pentenone derivative with high regio- and stereoselectivity, avoiding the formation of cyclic byproducts common in radical alkyne additions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (Z)-4-trifluoromethyl-5-sulfanyl-4-pentenone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic route described in CN110698313B for advancing fluorinated drug candidates. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to industrial manufacturing is seamless. Our facilities are equipped with state-of-the-art photochemical reactors and stringent purity specifications are maintained through our rigorous QC labs, guaranteeing that every batch of (Z)-4-trifluoromethyl-5-sulfanyl-4-pentenone meets the highest international standards. We understand that consistency is key in pharmaceutical supply, and our dedicated process development team is ready to optimize this specific route to maximize yield and minimize impurities for your specific application.
We invite you to leverage our technical expertise to accelerate your project timelines. By contacting our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage potential partners to reach out for specific COA data and route feasibility assessments to verify how this innovative chemistry can enhance your supply chain efficiency. Let us collaborate to bring your next-generation fluorinated therapeutics to market faster and more economically.
