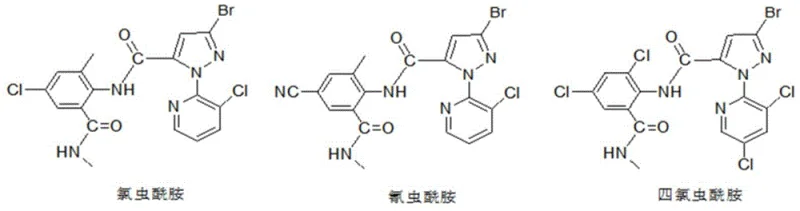
Advanced Continuous Synthesis of Bromopyrazole Intermediates for Diamide Insecticides
Advanced Continuous Synthesis of Bromopyrazole Intermediates for Diamide Insecticides
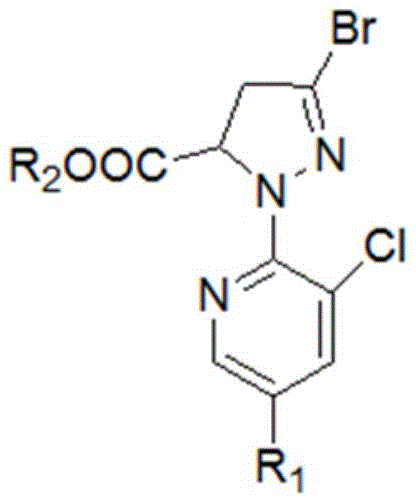
The agrochemical industry is constantly seeking more efficient pathways to produce key intermediates for next-generation insecticides, particularly the diamide class which includes market leaders like Chlorantraniliprole. Patent CN111072630A, published in April 2020, introduces a groundbreaking preparation method for 3-bromo-1-(3-chloro-5-R1-2-pyridyl)-4,5-dihydro-1H-pyrazole-5-carboxylic acid esters. This specific intermediate serves as the critical backbone for synthesizing high-efficiency broad-spectrum pesticides. The disclosed technology represents a significant departure from traditional batch processes by implementing a continuous reaction strategy that telescopes the cyclization and bromination steps. By fundamentally altering the reaction medium from acidic to alkaline during the initial cyclization, the inventors have successfully mitigated the formation of hydrolysis by-products that have long plagued this synthesis. This technical breakthrough not only enhances the chemical purity of the final intermediate but also streamlines the operational workflow, offering a compelling value proposition for manufacturers aiming to optimize their supply chains for diamide insecticides.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
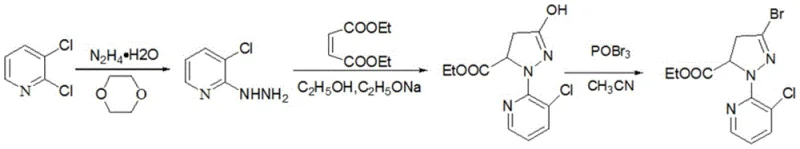
Historically, the synthesis of this vital bromopyrazole intermediate has been fraught with inefficiencies that drive up costs and complicate waste management. Prior art methods, such as those disclosed in WO2004087689 and CN102311424, typically rely on a cyclization step catalyzed by sodium alkoxide followed by a mandatory neutralization with acetic acid and water to precipitate the product. This acidic quenching step is chemically detrimental; it creates an environment where the sensitive 3-hydroxy-pyrazole intermediate is prone to hydrolysis, leading to the formation of unwanted by-products and a consequent reduction in overall yield. Furthermore, the subsequent bromination step in these legacy processes often requires the addition of sodium bicarbonate for neutralization, generating substantial volumes of salt-containing wastewater. These multi-step isolation and purification requirements not only increase the consumption of auxiliary chemicals like acetic acid and bicarbonate but also extend the production cycle time, creating bottlenecks for large-scale manufacturing operations.
The Novel Approach
In stark contrast to the fragmented and wasteful conventional protocols, the method described in CN111072630A employs a streamlined, continuous logic that preserves the integrity of the intermediate throughout the synthesis. The core innovation lies in the direct formation of the 3-hydroxy-1-(3-chloro-5-R1-2-pyridyl)-4,5-dihydro-1H-pyrazole-5-carboxylic acid ester sodium salt in an alkaline solution, completely bypassing the need for acidic neutralization. Instead of isolating the free acid or hydroxy form, the process retains the species as a stable sodium salt, which is then directly subjected to bromination using phosphorus oxybromide (POBr3). This approach effectively prevents the hydrolysis side reactions associated with acidic conditions, thereby securing higher reaction yields. Moreover, the ability to recover sodium bromide solids by simple hot filtration after the bromination step eliminates the need for bicarbonate neutralization and organic solvent extraction, drastically simplifying the downstream processing and reducing the environmental footprint of the manufacturing facility.
Mechanistic Insights into Alkaline Cyclization and Direct Bromination
The mechanistic elegance of this new process begins with the nucleophilic attack of the hydrazine group from 3-chloro-5-R1-2-hydrazinopyridine onto the electron-deficient double bond of the maleic acid diester. In the presence of a strong base like sodium ethoxide or sodium methoxide, the reaction proceeds rapidly to form a cyclic pyrazoline ring. Crucially, the alkaline environment ensures that the resulting 3-hydroxy group remains deprotonated as a sodium salt, stabilizing the molecule against the retro-aldol type decomposition or hydrolysis that occurs in acidic media. This stabilization is the key to the improved yield profile. The subsequent step involves the activation of this hydroxy-sodium salt by phosphorus oxybromide. The POBr3 acts as a potent brominating agent, replacing the hydroxyl functionality with a bromine atom via an SN2-type mechanism or through the formation of a reactive phosphate ester intermediate that undergoes nucleophilic substitution by bromide ions. This transformation occurs smoothly in acetonitrile, a solvent that facilitates the solubility of the polar intermediates while allowing for the easy precipitation of the inorganic sodium bromide by-product.
From an impurity control perspective, this mechanism offers distinct advantages over acid-catalyzed routes. In conventional methods, the presence of water during the acid neutralization phase promotes the hydrolysis of the ester moiety or the opening of the pyrazole ring, leading to complex impurity profiles that are difficult to purge. By maintaining anhydrous or low-water conditions during the critical cyclization-to-bromination transition, the new method minimizes these degradation pathways. The direct conversion of the sodium salt means that the reaction mixture remains homogeneous and chemically consistent until the final product precipitation. This consistency allows for tighter control over reaction parameters such as temperature and stoichiometry, ensuring that the final bromopyrazole intermediate meets stringent purity specifications required for downstream coupling reactions in the synthesis of active pharmaceutical ingredients or agrochemical active ingredients. The recovery of sodium bromide also serves as a built-in purification step, removing bromide ions that could otherwise catalyze further unwanted side reactions.
How to Synthesize 3-Bromo-1-(3-chloro-2-pyridyl)-4,5-dihydro-1H-pyrazole-5-carboxylic Ester Efficiently
The synthesis protocol outlined in the patent provides a robust framework for producing this high-value intermediate with minimal operational friction. The process begins by preparing an alkaline alcohol solution, typically using sodium ethoxide in ethanol, which serves as both the catalyst and the reaction medium. 3-Chloro-2-hydrazinopyridine is introduced to this solution, followed by the controlled addition of diethyl maleate. The reaction is heated to reflux to ensure complete cyclization, after which the solvent is distilled off to isolate the crude sodium salt. This salt is then redissolved in acetonitrile and treated with phosphorus oxybromide at elevated temperatures. The beauty of this method lies in its simplicity; it removes the need for pH adjustments, extractions, and multiple drying steps that characterize older technologies. For detailed operational parameters and specific stoichiometric ratios, please refer to the standardized guide below.
- React maleic acid diester with 3-chloro-5-R1-2-hydrazinopyridine in an alkaline alcohol solution to generate the sodium salt of the hydroxy-pyrazole intermediate.
- Distill to recover the solvent and obtain the crude sodium salt without acid neutralization.
- React the sodium salt directly with phosphorus oxybromide (POBr3) in acetonitrile to effect bromination and isolate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route translates into tangible operational efficiencies and risk mitigation. The most immediate impact is seen in the reduction of raw material costs. By eliminating the requirement for acetic acid and sodium bicarbonate, two commodities that add to the bill of materials without contributing to the final product mass, manufacturers can achieve significant cost savings per kilogram of output. Furthermore, the simplification of the workflow reduces the demand for labor and equipment utilization time, as the number of unit operations is effectively halved. The ability to recover sodium bromide also opens up potential revenue streams or waste disposal cost offsets, adding another layer of economic benefit to the process. These factors combined make the sourcing of intermediates produced via this method highly attractive for companies looking to optimize their cost of goods sold (COGS).
- Cost Reduction in Manufacturing: The elimination of acid neutralization and bicarbonate quenching steps removes the need for purchasing and handling these auxiliary chemicals, directly lowering the variable cost of production. Additionally, the higher yield achieved by preventing hydrolysis means that less starting material is wasted, improving the overall material efficiency of the plant. The reduction in solvent usage for extractions further contributes to lower operating expenses, making the process economically superior to legacy methods.
- Enhanced Supply Chain Reliability: Simplified processes are inherently more reliable because they have fewer points of failure. By removing complex workup procedures like pH adjustment and liquid-liquid extraction, the risk of batch failure due to operational error is minimized. This reliability ensures a more consistent supply of the intermediate, reducing the likelihood of stockouts that could disrupt the production of the final insecticide. The robustness of the sodium salt intermediate also allows for more flexible scheduling and potentially longer hold times if necessary, providing greater agility to the supply chain.
- Scalability and Environmental Compliance: The reduction in wastewater generation is a critical advantage for scaling up production. Traditional methods generate large volumes of salt-laden water that require expensive treatment before discharge. This new method drastically cuts down on effluent volume, easing the burden on wastewater treatment facilities and ensuring compliance with increasingly strict environmental regulations. The ability to filter off solid sodium bromide simplifies waste handling, turning a liquid waste problem into a manageable solid waste stream, which facilitates easier permitting and expansion of manufacturing capacity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis technology. These answers are derived directly from the experimental data and comparative analysis presented in patent CN111072630A, providing a clear understanding of why this method is becoming the preferred choice for modern agrochemical manufacturing. Understanding these nuances is essential for technical teams evaluating process transfers or capacity expansions.
Q: What is the primary advantage of the sodium salt method over conventional acid neutralization?
A: The primary advantage is the elimination of hydrolysis by-products. Conventional methods require adding acetic acid and water to neutralize the cyclization mixture, which often causes the sensitive intermediate to hydrolyze, lowering yield. The sodium salt method bypasses this acidic environment entirely.
Q: How does this process impact wastewater generation?
A: This process significantly reduces wastewater generation by omitting the neutralization steps that typically produce large volumes of salt-containing water. Additionally, sodium bromide by-products can be recovered by filtration rather than washed away.
Q: Can this method be scaled for commercial production of Chlorantraniliprole intermediates?
A: Yes, the process is designed for continuous operation and simplifies unit operations by removing extraction and neutralization steps, making it highly suitable for commercial scale-up in agrochemical manufacturing facilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Bromo-1-(3-chloro-2-pyridyl)-4,5-dihydro-1H-pyrazole-5-carboxylic Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize that the efficiency of your final product depends heavily on the quality and consistency of your intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the innovative continuous synthesis methods described in recent patents can be seamlessly translated into industrial reality. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of bromopyrazole intermediate meets the exacting standards required for the synthesis of high-performance diamide insecticides. We are committed to delivering not just a chemical, but a reliable supply solution that supports your long-term strategic goals.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic advantages of switching to this greener, more efficient process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your volume needs, ensuring that your supply chain remains competitive and resilient in the evolving global market.
