Scalable Synthesis of 3,6-Dichlorosalicylic Acid Intermediates for Commercial Herbicide Production
Introduction to Advanced 3,6-Dichlorosalicylic Acid Manufacturing
The global demand for high-efficiency herbicides continues to drive innovation in agrochemical intermediate synthesis, particularly for established compounds like Dicamba. Patent CN106659162B introduces a transformative methodology for producing 3,6-dichlorosalicylic acid compounds, which serve as critical precursors in the manufacture of this vital herbicide. Unlike legacy processes that rely on hazardous phenol derivatives, this disclosed technology leverages a robust salicylic acid-based pathway that fundamentally alters the economic and safety profile of production. By utilizing widely available salicylic acid as the foundational building block, the method circumvents the supply chain bottlenecks associated with specialized chlorinated phenols. This strategic shift not only enhances the reliability of raw material sourcing but also aligns with modern green chemistry principles by reducing the overall environmental footprint of the synthesis. For R&D directors and procurement strategists, understanding the nuances of this patent is essential for evaluating long-term supply security and cost optimization strategies in the competitive agrochemical market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Dicamba has heavily depended on routes originating from 2,5-dichlorophenol, a pathway fraught with significant operational and safety challenges. As illustrated in prior art schemes, these conventional methods often necessitate harsh reaction conditions, including high-pressure carboxylation steps that require specialized reactor infrastructure and rigorous safety protocols. The reliance on 2,5-dichlorophenol introduces volatility in the supply chain, as this intermediate is subject to market fluctuations and regulatory scrutiny regarding its own synthesis. Furthermore, traditional routes frequently suffer from poor regioselectivity during halogenation, leading to complex mixtures of isomers that are difficult and costly to separate. The generation of substantial hazardous waste streams, particularly from stoichiometric metal reagents and acidic byproducts, imposes a heavy burden on waste treatment facilities and increases the total cost of ownership. These inherent inefficiencies create a fragile manufacturing ecosystem where minor disruptions in precursor availability can halt entire production lines, posing a significant risk to supply chain continuity for downstream herbicide formulators.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a sequential halogenation strategy starting from salicylic acid, offering a streamlined and economically superior alternative. This method employs a controlled bromination followed by precise chlorination steps in an acidic medium, typically utilizing concentrated sulfuric acid or oleum to drive the electrophilic substitution reactions with high fidelity. The process allows for the formation of 5-bromo-3,6-dichlorosalicylic acid intermediates with exceptional regiocontrol, minimizing the formation of unwanted isomers and simplifying downstream purification. By avoiding high-pressure carboxylation, the process can be conducted in standard glass-lined or Hastelloy reactors, drastically reducing capital expenditure requirements for new manufacturing plants. The ability to telescope multiple reaction steps without isolating unstable intermediates further enhances throughput and reduces solvent consumption. This innovative pathway represents a paradigm shift in agrochemical intermediate manufacturing, providing a scalable solution that addresses both the economic and safety deficiencies of legacy technologies.
Mechanistic Insights into Electrophilic Halogenation and Catalytic Debromination
The core chemical innovation lies in the precise control of electrophilic aromatic substitution within a highly acidic reaction medium. The initial bromination of salicylic acid is directed to the 5-position due to the activating effects of the hydroxyl and carboxyl groups, a transformation that is carefully managed to prevent poly-bromination. Subsequent chlorination steps utilize active chlorine sources, such as chlorine gas or trichloroisocyanuric acid, in the presence of catalytic iodine or iodine monochloride. The use of oleum (fuming sulfuric acid) is critical here, as it maintains an anhydrous environment that prevents hydrolysis of the acid chloride species and ensures the solubility of the organic intermediates. The iodine catalyst functions by generating more reactive electrophilic chlorine species, allowing the reaction to proceed at moderate temperatures while maintaining high conversion rates. This mechanistic precision is vital for R&D teams aiming to replicate the process, as deviations in acid concentration or catalyst loading can lead to sulfonation byproducts that compromise yield. The final stage involves a selective catalytic debromination, typically using palladium on carbon under hydrogen atmosphere, which cleanly removes the temporary bromine directing group without affecting the essential chlorine atoms.
Impurity control is a paramount concern in the synthesis of pharmaceutical and agrochemical intermediates, and this patent offers sophisticated mechanisms to manage the impurity profile. The selective debromination step is particularly crucial, as it converts the 5-bromo-3,6-dichlorosalicylic acid intermediate into the desired 3,6-dichlorosalicylic acid with high specificity. The patent highlights that impurities such as iodine-containing species, which can poison the debromination catalyst, are effectively managed through specific workup procedures like suspension in organic solvents such as xylene. Furthermore, the direct filtration of products from the oleum medium minimizes exposure to water, preventing the degradation of sensitive intermediates. For quality assurance teams, understanding these purification dynamics is essential for establishing robust specification limits. The ability to produce intermediates with minimal halogenated byproducts ensures that the final herbicide meets stringent regulatory standards for residue levels. This level of chemical control demonstrates a deep understanding of reaction kinetics and thermodynamics, providing a reliable framework for consistent commercial production.
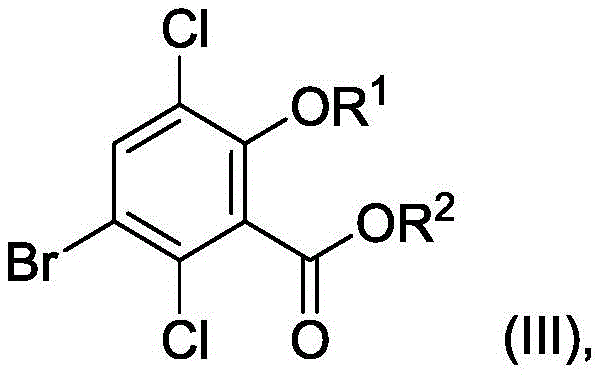
How to Synthesize 3,6-Dichlorosalicylic Acid Efficiently
Implementing this synthesis route requires a disciplined approach to reaction engineering and process safety management. The procedure begins with the dissolution of salicylic acid in concentrated sulfuric acid, followed by the controlled addition of bromine to generate 5-bromosalicylic acid. Once this intermediate is formed, the reaction medium is adjusted, often by adding oleum, to facilitate the subsequent chlorination using chlorine gas or solid chlorinating agents. The reaction temperature must be meticulously maintained, typically ranging from 0°C to 40°C depending on the specific step, to balance reaction rate with selectivity. Following the formation of the dibromodichloro intermediate, the mixture undergoes a catalytic hydrogenolysis step to remove the bromine atom. Detailed operational parameters, including stirring rates, gas flow velocities, and quenching protocols, are critical for maximizing yield and ensuring operator safety. For technical teams looking to adopt this technology, the following guide outlines the standardized synthesis steps derived from the patent examples, serving as a foundational reference for process development and scale-up activities.
- Brominate salicylic acid in concentrated sulfuric acid to form 5-bromosalicylic acid.
- Chlorinate the intermediate using chlorine gas or TCICA in oleum to yield 5-bromo-3,6-dichlorosalicylic acid.
- Perform catalytic hydrogenolysis to selectively remove the bromine atom, yielding the final 3,6-dichlorosalicylic acid product.
Commercial Advantages for Procurement and Supply Chain Teams
The adoption of this salicylic acid-based synthesis route offers profound commercial advantages that extend far beyond simple chemical yield improvements. For procurement managers, the shift to salicylic acid as a starting material represents a significant de-risking of the supply chain, as this commodity chemical is produced globally in massive volumes with stable pricing dynamics. Unlike specialized chlorinated phenols which may have limited suppliers, salicylic acid offers a diversified sourcing landscape that protects against regional supply disruptions. The elimination of high-pressure reaction steps reduces the need for expensive, custom-fabricated reactor vessels, allowing for the utilization of standard chemical processing equipment. This flexibility lowers the barrier to entry for new manufacturing sites and facilitates faster capacity expansion in response to market demand. Additionally, the telescoped nature of the reactions minimizes the number of isolation and drying steps, which directly translates to reduced energy consumption and lower utility costs per kilogram of product. These structural efficiencies create a leaner manufacturing model that is more resilient to external economic shocks and raw material price volatility.
- Cost Reduction in Manufacturing: The process achieves substantial cost savings by eliminating the need for expensive transition metal catalysts in the halogenation steps and replacing them with catalytic amounts of iodine, which can be recovered and recycled. The use of chlorine gas, a low-cost industrial commodity, further drives down reagent expenses compared to complex organic chlorinating agents. By reducing the number of unit operations through telescoping, the process minimizes labor hours and solvent losses, contributing to a lower overall cost of goods sold. These cumulative savings allow for more competitive pricing strategies in the global agrochemical market without compromising margin integrity.
- Enhanced Supply Chain Reliability: Utilizing salicylic acid ensures a robust and continuous supply of starting materials, as it is a staple chemical with well-established logistics networks worldwide. The simplified process flow reduces the dependency on niche intermediates that are prone to allocation during peak seasons. Furthermore, the stability of the intermediates allows for safer storage and transportation, mitigating risks associated with hazardous material handling. This reliability is crucial for maintaining consistent production schedules and meeting delivery commitments to downstream herbicide manufacturers, thereby strengthening long-term customer relationships.
- Scalability and Environmental Compliance: The synthetic route is inherently scalable, having been demonstrated to work effectively from laboratory bench scales to multi-ton commercial batches. The reduction in hazardous waste generation, particularly through the avoidance of heavy metal catalysts and the efficient use of acid media, simplifies wastewater treatment and disposal compliance. This environmental advantage aligns with increasingly stringent global regulations on industrial emissions and effluent quality. Companies adopting this technology can position themselves as sustainability leaders, appealing to eco-conscious partners and regulators while avoiding potential fines and remediation costs associated with older, dirtier technologies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and process descriptions found within the patent documentation, providing clarity on feasibility and performance. Understanding these details is vital for stakeholders evaluating the technology for potential licensing or internal development. The answers reflect the practical realities of scaling this chemistry and highlight the specific benefits that differentiate it from conventional approaches.
Q: What are the advantages of the salicylic acid route over traditional phenol routes for Dicamba production?
A: The salicylic acid route avoids hazardous high-pressure reactions and utilizes readily available starting materials, significantly improving process safety and reducing capital expenditure compared to traditional 2,5-dichlorophenol methods.
Q: How is high purity achieved in the final 3,6-dichlorosalicylic acid product?
A: High purity is ensured through a selective catalytic debromination step using palladium or platinum catalysts, which effectively removes bromine impurities while preserving the critical chlorine substituents required for biological activity.
Q: Can this synthesis method be scaled for industrial manufacturing?
A: Yes, the process is designed for scalability, utilizing telescoped reactions that minimize isolation steps and solvent usage, making it highly suitable for commercial scale-up of complex agrochemical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,6-Dichlorosalicylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of secure and efficient supply chains for high-value agrochemical intermediates like 3,6-dichlorosalicylic acid. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with advanced analytical instrumentation. Our facility is designed to handle complex halogenation chemistries safely and efficiently, leveraging the latest process innovations to maximize yield and minimize environmental impact. By partnering with us, you gain access to a supply chain that is not only robust but also adaptable to your specific formulation needs.
We invite you to engage with our technical procurement team to discuss how our manufacturing capabilities can support your business goals. We offer a Customized Cost-Saving Analysis to demonstrate the economic benefits of switching to our optimized supply model. Please contact us to request specific COA data and route feasibility assessments tailored to your project requirements. Let us be your trusted partner in driving efficiency and growth in the agrochemical sector.
