Scalable Synthesis of Estrone Amino Acid Ester Intermediates for Enhanced Antitumor Drug Development
The pharmaceutical industry continuously seeks novel strategies to enhance the therapeutic index of potent antitumor agents, particularly those suffering from poor pharmacokinetic profiles. Patent CN102127137A introduces a groundbreaking methodology for synthesizing intermediate compounds essential for producing estrone amino acid esters, specifically designed to overcome the limitations of 2-Methoxyestradiol (2-ME). While 2-ME exhibits broad-spectrum antitumor activity by inhibiting angiogenesis and disrupting microtubule function, its clinical utility has been severely hampered by low water solubility and rapid metabolic clearance. This patent addresses these critical deficiencies by proposing a structural modification strategy focused on the D-ring of the steroid nucleus, specifically at the 17-hydroxyl position. By conjugating various N-benzyloxycarbonyl (Cbz) protected amino acids or small peptide residues to the steroid core, the resulting intermediates serve as precursors to new drug candidates that maintain potent cytotoxicity while offering significantly improved solubility characteristics. This technological advancement represents a pivotal shift in steroid-based drug design, providing a robust chemical platform for developing next-generation oncology therapeutics with enhanced bioavailability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to modifying steroid hormones often involve harsh reaction conditions that can compromise the integrity of the sensitive steroid skeleton, particularly the aromatic A-ring which is crucial for receptor binding affinity. Conventional esterification methods frequently require strong acids or high temperatures that lead to unwanted side reactions, such as dehydration or rearrangement of the steroid backbone, resulting in complex impurity profiles that are difficult to purify. Furthermore, direct coupling of unprotected amino acids to sterically hindered secondary alcohols at the 17-position of the estrane framework typically suffers from low conversion rates and poor regioselectivity. The inherent hydrophobicity of the steroid nucleus also complicates reaction homogeneity, often necessitating the use of exotic solvents or prolonged reaction times that are economically unviable for large-scale manufacturing. These inefficiencies not only drive up production costs but also pose significant challenges in meeting the stringent purity requirements mandated by regulatory bodies for pharmaceutical intermediates, thereby delaying the translation of promising laboratory discoveries into viable clinical candidates.
The Novel Approach
The methodology outlined in patent CN102127137A circumvents these historical challenges through a meticulously optimized two-step synthetic route that prioritizes mild conditions and high selectivity. The process begins with the efficient protection of diverse amino acids or small peptides using benzyl chloroformate in an aqueous alkaline medium, ensuring the formation of stable N-Cbz derivatives without racemization. Subsequently, these activated building blocks are coupled to the steroid nucleus using a DCC/DMAP system in organic solvents like dichloromethane or chloroform at ambient temperatures ranging from 10°C to 30°C. This gentle approach preserves the stereochemical integrity of both the amino acid side chains and the chiral centers of the steroid core. 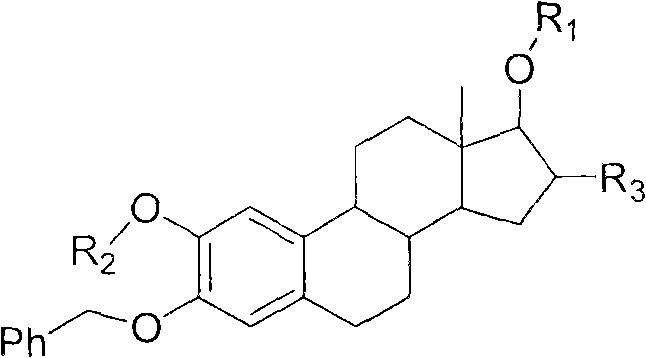 The versatility of this method is demonstrated by its compatibility with a wide array of amino acids, including beta-alanine, phenylalanine, leucine, and lysine, allowing for the fine-tuning of physicochemical properties. By achieving yields as high as 92% in specific embodiments, this novel approach establishes a scalable and reliable pathway for generating high-purity intermediates essential for the commercial production of advanced antitumor agents.
The versatility of this method is demonstrated by its compatibility with a wide array of amino acids, including beta-alanine, phenylalanine, leucine, and lysine, allowing for the fine-tuning of physicochemical properties. By achieving yields as high as 92% in specific embodiments, this novel approach establishes a scalable and reliable pathway for generating high-purity intermediates essential for the commercial production of advanced antitumor agents.
Mechanistic Insights into DCC-Mediated Steroid Esterification
The core chemical transformation in this synthesis relies on the activation of the carboxylic acid group of the N-Cbz protected amino acid by 1,3-dicyclohexylcarbodiimide (DCC) to form a highly reactive O-acylisourea intermediate. This activated species is susceptible to nucleophilic attack by the 17-beta-hydroxyl group of the steroid nucleus, a reaction that is significantly accelerated by the nucleophilic catalyst 4-dimethylaminopyridine (DMAP). DMAP functions by forming an even more reactive acylpyridinium ion, which effectively overcomes the steric hindrance presented by the bulky steroid framework at the 17-position. The reaction mechanism proceeds through a tetrahedral intermediate before collapsing to release the desired ester product and dicyclohexylurea (DCU) as a byproduct. The careful control of stoichiometry, specifically using a slight excess of the amino acid derivative (molar ratio 1-1.2:1) and a supplemental addition of DCC, ensures complete consumption of the valuable steroid starting material. 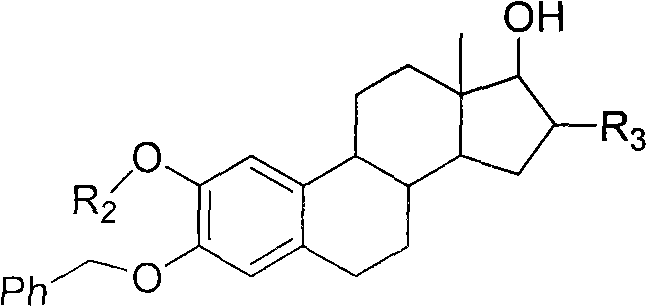 This mechanistic precision is critical for minimizing the formation of unreacted starting materials, which can be notoriously difficult to separate from the product due to similar polarity. The use of mild temperatures prevents the potential elimination reactions that could lead to the formation of unwanted alkene impurities, thereby securing a clean reaction profile suitable for downstream processing.
This mechanistic precision is critical for minimizing the formation of unreacted starting materials, which can be notoriously difficult to separate from the product due to similar polarity. The use of mild temperatures prevents the potential elimination reactions that could lead to the formation of unwanted alkene impurities, thereby securing a clean reaction profile suitable for downstream processing.
Impurity control is rigorously managed through real-time monitoring techniques integrated directly into the workflow. During the initial protection step, the reaction progress is tracked using ninhydrin staining, where the disappearance of the purple color indicates the complete consumption of free primary amines, ensuring that no unprotected amino acid carries over to the coupling stage. In the subsequent esterification phase, thin-layer chromatography (TLC) is employed to verify the complete conversion of the steroid alcohol. The workup procedure is equally sophisticated, involving sequential washes with distilled water and saturated brine to remove water-soluble urea byproducts and residual catalysts. Final purification via column chromatography using ethyl acetate and petroleum ether mixtures, or recrystallization from methanol or ethanol, guarantees that the final intermediate meets the high purity standards required for pharmaceutical applications. This comprehensive approach to quality control ensures that the impurity profile remains well within acceptable limits, facilitating smoother regulatory approval processes for the final drug substance.
How to Synthesize Estrone Amino Acid Ester Intermediates Efficiently
The synthesis of these high-value intermediates requires strict adherence to the optimized parameters defined in the patent to ensure reproducibility and safety. The process is divided into two distinct operational phases: the preparation of the N-Cbz protected amino acid building block and the subsequent coupling with the steroid nucleus. Operators must maintain precise temperature control, particularly during the exothermic addition of benzyl chloroformate, to prevent thermal runaway and ensure selective mono-protection. The detailed standard operating procedures below outline the specific molar ratios, solvent volumes, and reaction times necessary to achieve the reported high yields and purity levels. Following these guidelines allows manufacturing teams to replicate the success of the patent examples, such as the 92% yield achieved in the synthesis of the beta-alanine derivative, while maintaining a safe and controlled production environment.
- Protect amino acids or small peptides using benzyl chloroformate in alkaline aqueous solution at -5°C to 15°C to form N-Cbz protected intermediates.
- Dissolve the protected amino acid and steroid nucleus in organic solvent (e.g., dichloromethane) and add DCC and DMAP catalyst.
- Stir at 10-30°C for 10-20 hours, monitor by TLC, then purify via column chromatography or recrystallization to obtain the final intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this synthesis route offers substantial benefits in terms of cost efficiency and supply chain resilience. The reliance on commodity chemicals such as sodium hydroxide, benzyl chloroformate, and common amino acids ensures that raw material sourcing is stable and not subject to the volatility associated with exotic reagents. The elimination of cryogenic conditions, with reactions proceeding effectively between -5°C and 30°C, significantly reduces the energy burden on manufacturing facilities compared to processes requiring deep freezing or high-temperature reflux. This energy efficiency translates directly into lower operational expenditures, making the production of these complex intermediates more economically viable. Furthermore, the simplicity of the workup procedure, which utilizes standard extraction and filtration techniques rather than complex distillation or specialized chromatography, streamlines the manufacturing timeline and reduces the requirement for highly specialized labor.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by utilizing widely available and inexpensive coupling agents like DCC and solvents like dichloromethane, avoiding the need for precious metal catalysts often found in alternative cross-coupling methodologies. The high atom economy of the esterification step minimizes waste generation, reducing the costs associated with waste disposal and environmental compliance. Additionally, the ability to recover and recycle solvents such as ethyl acetate and petroleum ether further enhances the overall economic profile of the synthesis. By maximizing the yield of the valuable steroid starting material through optimized stoichiometry, the cost per kilogram of the final intermediate is drastically lowered, providing a competitive edge in the pricing of the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route ensures consistent supply continuity, as it is less susceptible to variations in raw material quality compared to more sensitive enzymatic or biocatalytic processes. The use of standard chemical equipment and glassware means that production can be easily transferred between different manufacturing sites without the need for specialized hardware investments. The stability of the N-Cbz protecting group allows for the storage of the amino acid intermediates, creating a buffer stock that can mitigate supply disruptions. This flexibility enables procurement managers to plan inventory more effectively, reducing the risk of production stoppages due to raw material shortages and ensuring timely delivery to downstream drug formulation partners.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of toxic heavy metals make this process inherently safer and easier to scale from laboratory benchtop to industrial tonnage production. The generation of dicyclohexylurea as a solid byproduct facilitates its removal via simple filtration, minimizing the liquid waste load and simplifying effluent treatment protocols. The process aligns well with green chemistry principles by reducing solvent usage through efficient concentration steps and avoiding the generation of hazardous gaseous byproducts. This environmental compatibility not only reduces regulatory hurdles but also enhances the corporate sustainability profile of the manufacturing entity, appealing to environmentally conscious stakeholders and investors in the global pharmaceutical market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these estrone amino acid ester intermediates. The answers are derived directly from the experimental data and technical specifications provided in patent CN102127137A, ensuring accuracy and relevance for R&D and procurement professionals. Understanding these details is crucial for evaluating the feasibility of integrating this technology into existing drug development pipelines and for assessing the quality attributes of the supplied materials.
Q: What is the primary advantage of modifying the 17-position of 2-Methoxyestradiol?
A: Modifying the 17-position (D-ring) preserves the antitumor activity of the A and B rings while significantly improving water solubility and bioavailability, addressing the key clinical limitation of native 2-ME.
Q: Which coupling agents are utilized in this synthesis protocol?
A: The process employs 1,3-dicyclohexylcarbodiimide (DCC) as the condensing agent and 4-dimethylaminopyridine (DMAP) as the catalyst to ensure efficient ester bond formation under mild conditions.
Q: How is reaction completion monitored during the amino acid protection step?
A: Reaction progress is strictly monitored using ninhydrin colorimetric testing to ensure the disappearance of free amine groups, supplemented by thin-layer chromatography (TLC) for precise endpoint determination.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Estrone Amino Acid Ester Intermediate Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis, leveraging deep expertise in steroid chemistry to bring complex pathways like the one described in CN102127137A to commercial reality. Our facility is equipped to handle the nuanced requirements of steroid functionalization, ensuring that every batch of estrone amino acid ester intermediate meets stringent purity specifications through our rigorous QC labs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, guaranteeing that your supply needs are met with consistency and reliability. Our commitment to quality ensures that the critical stereochemical integrity of the steroid nucleus is preserved throughout the manufacturing process, delivering intermediates that are ready for immediate downstream processing into final drug substances.
We invite global pharmaceutical partners to collaborate with us to optimize their supply chains for next-generation antitumor agents. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and project timelines. We encourage you to reach out today to obtain specific COA data and route feasibility assessments that demonstrate how our manufacturing capabilities can accelerate your drug development programs. Let us be your trusted partner in transforming innovative patent technologies into life-saving medicines.
