Advanced Photocatalytic Strategy for Efficient Alkaloid Intermediate Production
Introduction to Next-Generation Alkaloid Synthesis
The pharmaceutical industry continuously seeks robust and scalable methodologies for constructing complex heterocyclic scaffolds, particularly those found in bioactive natural products. Patent CN108707101B introduces a groundbreaking photocatalytic approach for the synthesis of alkaloids, specifically targeting the privileged 2-indolinone skeleton. This structural motif is ubiquitous in potent therapeutic agents such as Ziprasidone, Horsfiline, and Rhyncophylline, which exhibit significant kinase inhibitory and anti-tumor activities. The disclosed technology leverages visible-light photoredox catalysis to achieve direct C-H functionalization and cyclization, bypassing the need for harsh thermal conditions or stoichiometric metal oxidants. By utilizing readily available N-aryl acrylamides as starting materials, this method provides a streamlined pathway to 3-formyl-2-indolinones, which serve as versatile precursors for both indole fused-ring and spirocyclic alkaloids. This innovation represents a paradigm shift towards greener, more sustainable manufacturing processes for high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
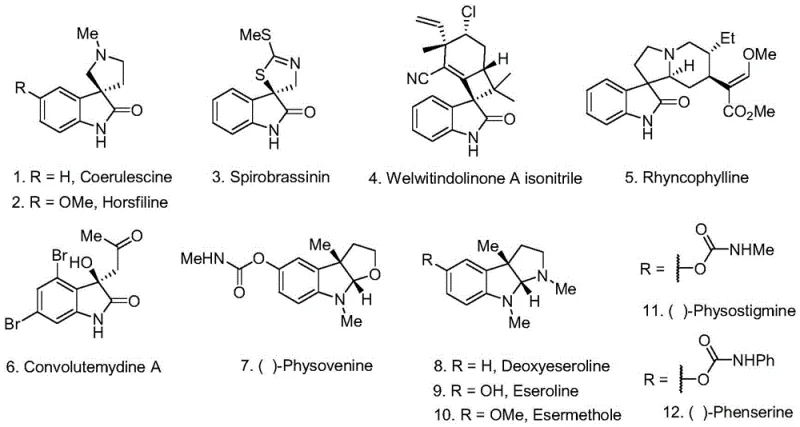
Traditionally, the construction of 3-functionalized 2-indolinones, especially those bearing quaternary carbon centers at the 3-position, has relied heavily on transition metal-catalyzed processes such as Heck reactions or intramolecular lactamizations. These conventional routes often suffer from significant drawbacks that hinder large-scale production. Firstly, they typically require expensive noble metal catalysts like palladium or rhodium, which not only increase raw material costs but also introduce stringent regulatory requirements for residual metal removal in final drug substances. Secondly, these reactions frequently necessitate high temperatures and strong bases, leading to poor functional group tolerance and potential decomposition of sensitive substrates. Furthermore, multi-step sequences are often required to install the necessary functionality at the 3-position, resulting in lower overall yields and increased waste generation. The reliance on stoichiometric oxidants in some older methods further exacerbates environmental concerns and complicates downstream processing, making these routes less attractive for modern cost-sensitive manufacturing environments.
The Novel Approach
In stark contrast, the novel photocatalytic strategy described in the patent offers a direct and efficient alternative that addresses these historical bottlenecks. By employing an organic photocatalyst such as 4CzIPN under visible light irradiation, the reaction proceeds smoothly at room temperature, eliminating the need for external heating and reducing energy consumption. The method utilizes tert-butyl hydroperoxide (TBHP) as a mild oxidant and 1,3-dioxolane as a formyl equivalent, enabling a tandem radical addition-cyclization sequence that directly installs the acetal functionality. This one-pot transformation significantly shortens the synthetic route, improving atom economy and operational simplicity. The subsequent hydrolysis step cleanly reveals the aldehyde group, providing access to 3-formyl-2-indolinones with high purity. This metal-free approach not only lowers the cost of goods but also simplifies the purification workflow, as there is no need for specialized scavengers to remove heavy metal residues, thereby accelerating the timeline from laboratory synthesis to commercial production.
Mechanistic Insights into Photoredox-Catalyzed Cyclization
The core of this technological advancement lies in the intricate photoredox catalytic cycle that drives the formation of the indolinone core. Upon irradiation with blue LED light, the organic photocatalyst (e.g., 4CzIPN) enters an excited state, facilitating single-electron transfer (SET) processes that generate reactive radical species from the peroxide oxidant. These radicals abstract hydrogen atoms from the 1,3-dioxolane solvent, creating alpha-oxy radicals that add selectively to the electron-deficient double bond of the N-aryl acrylamide substrate. This radical addition triggers a cascade of intramolecular cyclization events, effectively closing the five-membered lactam ring while simultaneously establishing the quaternary carbon center at the 3-position. The versatility of this mechanism allows for the accommodation of various electronic and steric environments on the aromatic ring, ensuring broad substrate scope. The intermediate 3-acetal-2-indolinone is then subjected to acidic hydrolysis, which cleaves the acetal protecting group to yield the desired 3-formyl product. This mechanistic pathway avoids the high-energy barriers associated with thermal cyclization, allowing for the preservation of sensitive functional groups that would otherwise degrade under traditional conditions.
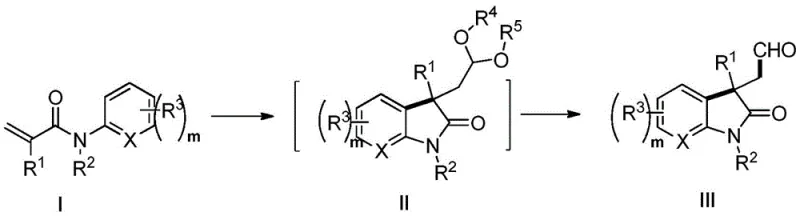
Following the formation of the 3-formyl-2-indolinone intermediate, the synthetic utility is extended through reductive transformations to access complex alkaloid architectures. The aldehyde moiety serves as a pivotal handle for further derivatization, enabling the construction of indole fused-ring systems or spirocyclic frameworks depending on the reduction conditions employed. For instance, treatment with lithium aluminum hydride (LiAlH4) can reduce both the lactam carbonyl and the aldehyde, followed by spontaneous cyclization to form tetrahydrofuroindole structures. Alternatively, reductive amination with amines in the presence of reducing agents like sodium cyanoborohydride allows for the introduction of nitrogen-containing side chains, leading to pyrroloindoline alkaloids. This modularity is crucial for medicinal chemistry campaigns where rapid exploration of structure-activity relationships (SAR) is required. The ability to diverge from a common intermediate to multiple distinct scaffolds enhances the value proposition of this synthetic route, making it a powerful tool for the discovery and development of new therapeutic candidates targeting various biological pathways.
How to Synthesize 3-Formyl-2-indolinone Efficiently
The practical implementation of this photocatalytic protocol is designed for ease of operation and scalability, making it highly suitable for process chemistry teams aiming to optimize production workflows. The reaction setup involves standard photoreactors equipped with blue LED arrays, which are increasingly common in modern synthesis laboratories. The use of commercially available reagents such as TBHP and 1,3-dioxolane ensures that supply chain logistics remain straightforward and cost-effective. Detailed standardized synthetic steps for the preparation of key intermediates and final alkaloids are provided in the guide below, outlining specific molar ratios, solvent choices, and workup procedures that have been validated to deliver consistent high yields. This level of procedural clarity minimizes the risk of batch-to-batch variability and facilitates technology transfer from R&D to pilot plant operations.
- React N-aryl acrylamide with a formyl equivalent (like 1,3-dioxolane) and oxidant (TBHP) under blue LED light with 4CzIPN catalyst.
- Hydrolyze the resulting 3-acetal-2-indolinone intermediate using acidic aqueous solution to reveal the aldehyde group.
- Reduce the 3-formyl-2-indolinone using LiAlH4 or reductive amination conditions to form the final indole fused or spiro alkaloids.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this photocatalytic methodology offers substantial strategic benefits that align with the goals of cost reduction and operational resilience. The elimination of precious metal catalysts removes a significant variable cost component and mitigates the supply risk associated with fluctuating prices of metals like palladium and iridium. Furthermore, the mild reaction conditions reduce the demand for specialized high-pressure or high-temperature equipment, allowing for production in standard glass-lined reactors or even continuous flow setups, which enhances manufacturing flexibility. The simplified purification process, devoid of heavy metal scavenging steps, shortens the overall production cycle time and reduces the consumption of auxiliary materials such as silica gel and filtration aids. These factors collectively contribute to a more lean and agile manufacturing process, capable of responding quickly to market demands while maintaining strict quality standards required for pharmaceutical ingredients.
- Cost Reduction in Manufacturing: The transition to a metal-free catalytic system fundamentally alters the cost structure of producing these complex intermediates. By replacing expensive transition metal complexes with organic photocatalysts that can potentially be recovered or used in low loadings, the direct material costs are significantly lowered. Additionally, the ambient temperature operation drastically cuts energy expenditures related to heating and cooling, which is a major overhead in large-scale chemical synthesis. The reduction in waste generation, particularly heavy metal contaminated waste, also lowers disposal costs and environmental compliance fees, contributing to a more sustainable and economically viable production model that improves the overall margin profile for the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: Reliance on scarce or geographically concentrated resources poses a risk to supply continuity, but this method utilizes widely available organic reagents and common solvents. The robustness of the reaction against moisture and oxygen variations, combined with the use of stable photocatalysts, ensures consistent output even with minor fluctuations in raw material quality. This reliability is critical for maintaining uninterrupted supply to downstream drug manufacturers. Moreover, the potential for using polymer-supported photocatalysts, as hinted in some embodiments of the patent, opens the door to heterogeneous catalysis which further simplifies catalyst recovery and reuse, stabilizing the supply chain against catalyst shortages and price volatility.
- Scalability and Environmental Compliance: Scaling photochemical reactions has historically been challenging due to light penetration issues, but advancements in flow chemistry and high-surface-area reactors have made this highly feasible. The modular nature of LED lighting allows for linear scale-up without the exponential safety risks associated with thermal runaways in exothermic reactions. From an environmental standpoint, the process adheres to green chemistry principles by minimizing hazardous reagents and waste. The absence of toxic heavy metals simplifies the environmental impact assessment and regulatory filing process, accelerating time-to-market for new drugs. This alignment with global sustainability goals enhances the corporate reputation of manufacturers adopting this technology and ensures long-term compliance with increasingly stringent environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocatalytic synthesis platform. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on the feasibility and advantages of the method for industrial application. Understanding these details helps stakeholders make informed decisions about integrating this technology into their existing manufacturing portfolios.
Q: What is the primary advantage of this photocatalytic method over traditional metal catalysis?
A: The primary advantage is the elimination of expensive and toxic transition metals like palladium or ruthenium. This method uses an organic photocatalyst (4CzIPN) under visible light at room temperature, significantly simplifying purification and reducing heavy metal contamination risks in the final API.
Q: What are the typical reaction conditions for the cyclization step?
A: The reaction proceeds under mild conditions, typically at room temperature (15-35°C) using a 5W blue LED light source. It requires an inert atmosphere (nitrogen or argon) and common solvents like 1,3-dioxolane, making it safer and more energy-efficient than high-temperature thermal processes.
Q: Can this method be applied to diverse substrate scopes?
A: Yes, the patent demonstrates excellent functional group tolerance. Various substituents on the aromatic ring, including electron-donating groups like methoxy and electron-withdrawing groups like esters or cyano groups, are well-tolerated, allowing for the synthesis of a wide library of alkaloid analogs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Formyl-2-indolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this photocatalytic technology in advancing the synthesis of complex alkaloid intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions seamlessly from benchtop discovery to full-scale manufacturing. Our state-of-the-art facilities are equipped with advanced photoreactors and rigorous QC labs capable of meeting stringent purity specifications required for global pharmaceutical markets. We are committed to delivering high-quality intermediates that adhere to the highest standards of safety and efficacy, leveraging our technical expertise to optimize yields and minimize impurities throughout the production process.
We invite you to collaborate with us to explore the full capabilities of this innovative synthetic route for your specific drug development needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this metal-free approach can enhance your bottom line. Please contact us to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary molecules. Let us be your trusted partner in navigating the complexities of modern chemical synthesis and securing a reliable supply chain for your critical pharmaceutical ingredients.
