Advanced Metal-Free Acylation Technology for Scalable Pharmaceutical Intermediate Production
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic methodologies that balance efficiency with safety and regulatory compliance. Patent CN114380718B introduces a transformative approach to the acylation of aniline compounds, addressing critical limitations associated with traditional protecting group strategies. This innovation utilizes diethyl azodicarboxylate (DEAD) as a novel acyl source under mild alkaline promotion, effectively bypassing the need for highly toxic reagents like ethyl chloroformate or harsh high-pressure carbon monoxide systems. By operating at moderate temperatures between 50-90°C and achieving completion within 30-60 minutes, this technology offers a streamlined pathway for producing high-purity carbamate intermediates essential for drug synthesis. For R&D directors and process chemists, this represents a significant leap forward in designing safer, more sustainable routes for complex molecule assembly, particularly for amino-cyanobenzene derivatives widely used in agrochemical and pharmaceutical applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the protection of aniline amino groups has relied heavily on reagents that pose significant operational and environmental hazards. The industry standard often involves ethyl chloroformate, a compound notorious for its high toxicity, flammability, and severe irritation to the respiratory tract and conjunctiva, necessitating expensive containment infrastructure and rigorous safety protocols. Alternatively, carbonylation strategies utilizing carbon monoxide or carbon dioxide-alcohol systems frequently demand extreme reaction conditions, including temperatures up to 800°C and pressures reaching 5 MPa, alongside the use of precious transition metal catalysts such as palladium, rhodium, or copper. These conventional methods not only inflate capital expenditure due to the need for specialized high-pressure reactors but also introduce complex purification challenges related to removing trace heavy metals, which is a critical bottleneck for regulatory approval in pharmaceutical manufacturing.
The Novel Approach
In stark contrast, the methodology disclosed in CN114380718B leverages commercially available azo compounds, specifically DEAD, to achieve rapid acylation under remarkably mild conditions. This metal-free protocol eliminates the requirement for high-pressure equipment and toxic gaseous reagents, instead utilizing simple inorganic bases like sodium carbonate or potassium phosphate in common polar aprotic solvents. The reaction proceeds efficiently at temperatures ranging from 50-90°C, drastically reducing energy consumption and thermal stress on sensitive substrates. Furthermore, the process demonstrates exceptional functional group tolerance, successfully accommodating electron-withdrawing substituents such as cyano, fluoro, chloro, and methoxy groups without compromising yield or selectivity. This shift towards milder, metal-free chemistry aligns perfectly with modern green chemistry principles, offering a reliable pharmaceutical intermediate supplier a distinct competitive advantage in terms of process safety and scalability.

Mechanistic Insights into DEAD-Mediated Base-Promoted Acylation
The mechanistic pathway of this transformation is elegantly simple yet highly effective, relying on the unique reactivity of the azo functionality under basic conditions. The reaction initiates when oxygen anions from the base, such as carbonate, nucleophilically attack the nitrogen atom of the diethyl azodicarboxylate, generating a reactive zwitterionic intermediate. This species subsequently abstracts a proton from the aniline substrate, facilitating the formation of a nitrogen anion on the aniline ring while simultaneously activating the ester carbonyl of the azo-reagent. The resulting nitrogen anion then performs a nucleophilic addition onto the activated ester carbonyl, forming a tetrahedral intermediate that collapses via electron transfer to release the target ethyl carbamate product. This mechanism avoids the formation of stable metal-ligand complexes, thereby preventing catalyst deactivation and ensuring consistent reaction kinetics across diverse substrate classes.
From an impurity control perspective, this mechanism offers substantial benefits by minimizing side reactions commonly associated with transition metal catalysis, such as homocoupling or over-reduction. The absence of metal centers eliminates the risk of metal-induced decomposition of sensitive functional groups like nitriles or halides, which are prevalent in many bioactive scaffolds. Additionally, the byproduct profile is significantly cleaner, primarily consisting of hydrazine derivatives that are easily removed during the aqueous workup and silica gel chromatography steps described in the patent examples. This high level of chemoselectivity ensures that the final product maintains a superior purity profile, reducing the burden on downstream purification units and enhancing the overall mass balance of the manufacturing process.
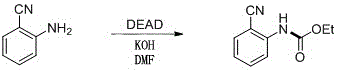
How to Synthesize Ethyl Carbamates Efficiently
The practical implementation of this acylation strategy is straightforward and amenable to standard laboratory and pilot plant equipment. The general procedure involves charging a reaction vessel with the aniline substrate, a slight excess of DEAD, and a stoichiometric amount of base, followed by the addition of a solvent such as DMSO or DMF. The mixture is then heated to the specified temperature range, typically around 70°C, and monitored via TLC until conversion is complete, usually within one hour. Upon completion, the reaction is quenched with saturated ammonium chloride, and the product is isolated through liquid-liquid extraction with ethyl acetate, followed by drying and solvent removal. For detailed operational parameters, specific stoichiometric ratios, and purification gradients tailored to different substrates, please refer to the standardized synthesis guide below.
- Charge a reaction vessel with the aniline substrate, diethyl azodicarboxylate (DEAD), and a suitable inorganic or organic base such as sodium carbonate or potassium phosphate.
- Add an appropriate polar aprotic solvent like DMSO, DMF, or acetonitrile to achieve a substrate concentration of 0.05-0.2 mol/L and heat the mixture to 50-90°C.
- Maintain reaction for 30-60 minutes, quench with saturated ammonium chloride, extract with ethyl acetate, and purify the crude residue via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this DEAD-mediated acylation technology translates into tangible operational efficiencies and risk mitigation. By transitioning away from hazardous reagents like ethyl chloroformate and high-pressure gas systems, facilities can significantly reduce their insurance premiums and safety compliance costs. The reliance on commercially available, shelf-stable reagents ensures a consistent supply chain, mitigating the risks associated with the logistics of toxic gases or specialized catalysts that may face geopolitical or availability constraints. Furthermore, the simplified workup procedure, which avoids complex metal scavenging resins, streamlines the production cycle, allowing for faster batch turnover and improved asset utilization rates in multipurpose manufacturing plants.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts such as palladium or rhodium directly lowers the raw material cost per kilogram of the intermediate. Additionally, removing the need for high-pressure reactors reduces capital depreciation costs and maintenance expenses associated with specialized equipment. The mild reaction conditions also result in lower energy consumption for heating and cooling cycles, contributing to a reduced overall utility bill. Qualitatively, the simplified purification process reduces the volume of silica gel and solvents required for chromatography, further driving down the cost of goods sold without compromising product quality.
- Enhanced Supply Chain Reliability: Utilizing commodity chemicals like DEAD and sodium carbonate ensures that the supply chain is not vulnerable to the bottlenecks often seen with specialty catalysts or toxic gases. The robustness of the reaction across various substituted anilines means that a single platform technology can be applied to multiple products, simplifying inventory management and raw material sourcing. This flexibility allows manufacturers to respond more agilely to market demands, ensuring continuous production even if specific substrate availability fluctuates. The stability of the reagents also permits longer storage times, reducing waste from expired materials and enhancing overall supply chain resilience.
- Scalability and Environmental Compliance: The metal-free nature of this process inherently simplifies environmental compliance, as there is no need for extensive wastewater treatment to remove heavy metal residues. This aligns with increasingly stringent global environmental regulations, reducing the risk of fines and shutdowns. The mild conditions and lack of high-pressure requirements make the process inherently safer to scale from kilogram to tonne levels, minimizing the engineering controls needed for scale-up. Consequently, this technology supports sustainable manufacturing goals by reducing the generation of hazardous waste and lowering the carbon footprint associated with energy-intensive high-pressure operations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this acylation technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a factual basis for decision-making. Understanding these details is crucial for evaluating the feasibility of integrating this method into existing production lines or new product development pipelines.
Q: What are the primary safety advantages of using DEAD over ethyl chloroformate for aniline acylation?
A: Unlike ethyl chloroformate, which is highly toxic, flammable, and a severe respiratory irritant, the DEAD-mediated process operates under significantly milder conditions without generating hazardous acidic byproducts, thereby enhancing operator safety and reducing containment costs.
Q: Does this metal-free acylation method tolerate sensitive functional groups like nitriles and halogens?
A: Yes, the protocol demonstrates excellent functional group tolerance, successfully acylating substrates containing electron-withdrawing groups such as cyano, fluoro, chloro, and bromo moieties without requiring protective group strategies or harsh transition metal catalysts.
Q: How does the absence of transition metals impact downstream purification and regulatory compliance?
A: Eliminating transition metal catalysts like palladium or copper removes the need for complex and costly metal scavenging steps, simplifying the purification workflow and ensuring the final API intermediate meets stringent residual metal specifications required by global regulatory bodies.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ethyl Carbamate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting innovative synthetic routes that enhance both product quality and process safety. Our team of expert process chemists has extensively evaluated the DEAD-mediated acylation technology described in CN114380718B and confirmed its potential for large-scale application. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab to plant is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of ethyl carbamate intermediate delivered meets the highest international standards for pharmaceutical and agrochemical use.
We invite you to collaborate with us to leverage this advanced technology for your specific project needs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this metal-free process for your specific molecule. We encourage you to contact us today to discuss your requirements,索取 specific COA data for our reference standards, and obtain detailed route feasibility assessments tailored to your production targets. Let us help you optimize your supply chain with safer, more efficient, and cost-effective chemical solutions.
