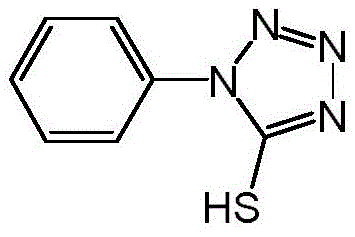
Scalable Production of 1-Phenyl-5-Mercapto Tetrazole for Statin Intermediates
The pharmaceutical industry's relentless pursuit of efficient lipid-lowering therapies has placed significant demand on the supply chain for key statin intermediates, specifically 1-phenyl-5-mercapto tetrazole (CAS 86-93-1). As detailed in the recent patent CN113372287A, a breakthrough synthetic methodology has emerged that addresses long-standing safety and efficiency bottlenecks in producing this critical heterocyclic building block. This innovative approach utilizes 1-phenyl-5-hydroxytetrazole as a robust starting material, transforming it through a strategic two-step sequence that eliminates the need for hazardous reagents like phenyl thioisocyanate. By leveraging a sulfonate activation strategy followed by a mild thionation reaction, this process delivers the target molecule with exceptional purity and yield metrics that exceed 90%. For R&D teams and procurement specialists alike, this represents a paradigm shift towards greener, safer, and more economically viable manufacturing protocols for high-purity statin intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 1-phenyl-5-mercapto tetrazole has been plagued by severe safety hazards and operational complexities that drive up costs and risk profiles. One prevalent legacy method involves the reaction of carbon disulfide with aniline, a pathway notorious for generating toxic sulfide smoke upon decomposition, posing acute health risks to plant personnel and requiring expensive scrubbing systems. Another common route relies on phenyl thioisocyanate reacting with sodium azide; however, phenyl thioisocyanate is not only highly toxic and combustible but also possesses strong irritant properties that complicate handling and storage logistics. These conventional pathways often suffer from multi-step procedures with lower overall atom economy, leading to increased waste generation and higher disposal costs. Furthermore, the reliance on such dangerous raw materials creates significant supply chain vulnerabilities, as regulatory scrutiny on hazardous chemical transport continues to tighten globally, potentially causing delays and price volatility for buyers seeking a reliable pharmaceutical intermediates supplier.
The Novel Approach
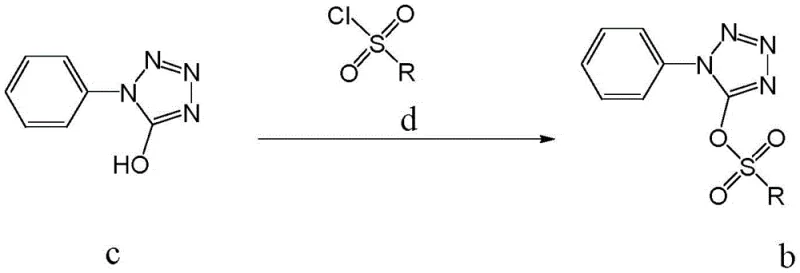
In stark contrast to these hazardous legacy routes, the novel methodology disclosed in the patent introduces a remarkably safe and streamlined synthetic architecture centered on the activation of 1-phenyl-5-hydroxytetrazole. This approach bypasses the need for toxic isocyanates entirely by first converting the hydroxyl group into a superior leaving group via sulfonation. As illustrated in the reaction scheme below, the starting material (Compound c) reacts with various sulfonyl chlorides (Compound d) in the presence of an acid-binding agent to form a stable sulfonate intermediate (Compound b). This activation step is crucial as it renders the tetrazole ring susceptible to nucleophilic attack by sulfur sources in the subsequent step without requiring harsh conditions or transition metal catalysts. The simplicity of this design allows for operation at moderate temperatures between 20°C and 50°C, drastically reducing energy consumption compared to high-temperature pyrolysis methods. This strategic pivot not only enhances operator safety but also simplifies the purification workflow, making it an ideal candidate for cost reduction in API manufacturing environments where margin compression is a constant challenge.
Mechanistic Insights into Sulfonate Activation and Thionation
The core chemical innovation of this process lies in the precise manipulation of the tetrazole ring's electronic properties through sulfonate ester formation. In the first stage, the hydroxyl group at the 5-position of the tetrazole ring acts as a nucleophile, attacking the sulfur atom of the sulfonyl chloride reagent. This reaction, facilitated by an organic base such as triethylamine or pyridine, effectively converts the poor leaving hydroxyl group into a sulfonate ester, which is an excellent leaving group due to the resonance stabilization of the resulting sulfonate anion. This activation is fundamental because the direct displacement of a hydroxyl group on an aromatic-like heterocycle is energetically unfavorable; the sulfonate intermediate lowers the activation energy barrier significantly. The choice of R group on the sulfonyl chloride (whether phenyl, benzyl, or alkyl) offers tunability, allowing chemists to optimize the steric and electronic environment for the subsequent substitution, ensuring that side reactions such as hydrolysis are minimized during the workup phase.
Following activation, the second critical mechanistic event is the nucleophilic thionation, where the sulfonate group is displaced by a sulfur nucleophile. As shown in the transformation below, reagents such as thiourea or sodium hydrosulfide serve as the sulfur source, attacking the C5 position of the tetrazole ring. The reaction proceeds via an SNAr-like or addition-elimination mechanism where the electron-withdrawing nature of the adjacent nitrogen atoms in the tetrazole ring facilitates the displacement. The use of a mixed solvent system comprising ethanol and water is particularly ingenious; it ensures adequate solubility for both the organic intermediate and the inorganic or polar thionating agents while maintaining a homogeneous reaction medium. The subsequent addition of sodium hydroxide serves to hydrolyze any intermediate thiouronium salts formed when using thiourea, liberating the free thiol group. This meticulous control over reaction conditions prevents the formation of disulfide impurities, a common issue in thiol chemistry, thereby securing the high-purity profile required for downstream pharmaceutical applications.

How to Synthesize 1-Phenyl-5-Mercapto Tetrazole Efficiently
The practical execution of this synthesis is designed for seamless integration into existing pilot and production facilities, requiring only standard glass-lined or stainless steel reactors equipped with reflux condensers and nitrogen inlet ports. The process begins with the dissolution of 1-phenyl-5-hydroxytetrazole in dichloromethane, followed by the controlled addition of the acid-binding agent and the sulfonyl chloride solution, maintaining the temperature strictly between 20°C and 50°C to prevent exothermic runaway. Once the sulfonate intermediate is formed and isolated via standard extraction and recrystallization techniques, it is subjected to the thionation step in an ethanol-water mixture under a nitrogen blanket to exclude oxygen, which could otherwise oxidize the sensitive thiol product. Detailed standardized operating procedures regarding stoichiometry, specifically the molar ratios of reagents like thiourea (1:1 to 1:3 relative to substrate) and the concentration of NaOH (0.8-4N), are critical for maximizing the reported yields of over 90%. For a complete breakdown of the specific experimental parameters and workup protocols validated in the patent examples, please refer to the structured guide below.
- Activate 1-phenyl-5-hydroxytetrazole by reacting with a sulfonyl chloride (e.g., p-toluenesulfonyl chloride) and an acid-binding agent like triethylamine in dichloromethane at 20-50°C to form the sulfonate intermediate.
- Perform a thionation reaction by refluxing the sulfonate intermediate with a thionating agent such as thiourea or sodium hydrosulfide in an ethanol-water mixture under nitrogen atmosphere.
- Complete the synthesis by adding aqueous NaOH, continuing reflux, followed by acidification, extraction with dichloromethane, drying, and distillation under reduced pressure to isolate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers profound strategic advantages that extend far beyond simple unit cost calculations. By eliminating the requirement for phenyl thioisocyanate, a reagent classified as toxic and combustible with stringent storage regulations, companies can significantly reduce their insurance premiums and compliance overheads associated with hazardous material handling. The process operates under mild thermal conditions and utilizes ubiquitous solvents like dichloromethane and ethanol, which are readily available in global chemical markets, thereby insulating the supply chain from the volatility often seen with specialty reagents. Furthermore, the high yield efficiency (>90%) directly translates to reduced raw material consumption per kilogram of finished product, optimizing the cost of goods sold (COGS) without compromising on quality. This efficiency also means less waste generation, simplifying effluent treatment processes and aligning with increasingly rigorous environmental, social, and governance (ESG) mandates that multinational corporations must adhere to.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by its catalyst-free nature and high atom economy. Traditional methods often require expensive transition metal catalysts or complex purification steps to remove heavy metal residues, which adds substantial cost and time to the production cycle. By avoiding these catalysts entirely, the new method removes the need for costly scavenging resins and extensive analytical testing for metal content, streamlining the quality control workflow. Additionally, the high conversion rates minimize the loss of valuable starting materials, ensuring that every dollar spent on raw inputs contributes maximally to the final output. This lean manufacturing approach allows for substantial cost savings in API manufacturing, providing a competitive edge in pricing negotiations with downstream drug developers who are constantly pressured to lower healthcare costs.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of commoditized raw materials that are less prone to geopolitical supply shocks. Unlike specialized isocyanates which may have limited suppliers and long lead times, sulfonyl chlorides and thiourea are produced at massive scales globally, ensuring consistent availability. The robustness of the reaction conditions also means that the process is less sensitive to minor fluctuations in utility supplies or ambient conditions, reducing the risk of batch failures that can disrupt delivery schedules. This reliability is crucial for reducing lead time for high-purity pharmaceutical intermediates, enabling manufacturers to respond more agilely to sudden spikes in demand for statin medications. Consequently, partners can maintain lower safety stock levels while still guaranteeing continuity of supply, freeing up working capital for other strategic investments.
- Scalability and Environmental Compliance: From a scale-up perspective, the simplicity of the two-step sequence facilitates a smooth transition from laboratory grams to commercial tons. The absence of exotic reagents or extreme pressures means that existing infrastructure can often be utilized without major capital expenditure on new reactor types. Environmentally, the process generates fewer hazardous by-products; the primary waste streams consist of benign salts and recoverable solvents, which are easier to treat than the toxic sulfide wastes associated with carbon disulfide routes. This alignment with green chemistry principles not only mitigates regulatory risk but also enhances the brand reputation of the manufacturer as a sustainable partner. The ability to commercial scale-up of complex heterocycles safely and cleanly makes this technology a future-proof asset for long-term production planning.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived directly from the patent's experimental data and background analysis. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their current manufacturing portfolios. The answers provided reflect the specific advantages in safety, yield, and operational simplicity that distinguish this method from prior art, offering clarity on how it resolves historical pain points in tetrazole chemistry.
Q: Why is this new method safer than traditional routes for 1-phenyl-5-mercapto tetrazole?
A: Traditional methods often utilize highly toxic and combustible phenyl thioisocyanate or generate toxic sulfide smoke from carbon disulfide decomposition. This patented route avoids these hazardous reagents entirely by using stable sulfonyl chlorides and common thionating agents like thiourea, significantly improving operational safety.
Q: What yields can be expected from this catalyst-free synthesis process?
A: The patent data indicates that this efficient preparation method achieves high yields exceeding 90% for the target compound. Specifically, examples demonstrate overall yields ranging from 82.6% to 90.2%, which is competitive with or superior to existing industrial methods while eliminating the need for expensive transition metal catalysts.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process is explicitly designed for industrial scalability. It utilizes common solvents like dichloromethane and ethanol, operates at moderate temperatures (20-50°C for activation, reflux for thionation), and involves straightforward workup procedures such as extraction and distillation, making it ideal for reliable pharmaceutical intermediates supplier operations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Phenyl-5-Mercapto Tetrazole Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to safer, more efficient synthetic routes is critical for the long-term sustainability of the pharmaceutical supply chain. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a GMP-compliant manufacturing environment. We are committed to delivering high-purity 1-phenyl-5-mercapto tetrazole that meets stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our dedication to quality assurance means that every batch is thoroughly characterized to ensure it is free from the toxic impurities associated with older synthesis methods, providing peace of mind for your R&D and production teams.
We invite you to engage with our technical procurement team to discuss how this advanced manufacturing technology can be tailored to your specific volume requirements and cost targets. By partnering with us, you gain access to a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this catalyst-free route for your specific application. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and efficiency in your statin intermediate supply chain.
