Scalable Industrial Synthesis of 6-Bromonaphthalene-1,2-Diamine for HCV Inhibitor Production
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
The synthesis of complex naphthalene derivatives, particularly 1,2-diamines substituted with halogens, has historically presented significant challenges in process chemistry and industrial scalability. Traditional routes often suffer from poor regioselectivity during electrophilic aromatic substitution, leading to difficult-to-separate isomeric mixtures that drastically reduce overall yield and increase purification costs. Furthermore, many existing laboratory-scale methods rely on hazardous reagents or extreme conditions that are impractical for multi-kilogram production, creating a supply chain bottleneck for downstream pharmaceutical applications such as Hepatitis C virus (HCV) inhibitors. The lack of a robust, documented industrial pathway for key intermediates like 6-bromonaphthalene-1,2-diamine has forced manufacturers to rely on inefficient custom synthesis or low-yield exploratory routes, resulting in high material costs and inconsistent quality profiles that fail to meet stringent GMP standards required for API manufacturing.
The Novel Approach
The methodology disclosed in patent CN112679365A introduces a streamlined, four-step synthetic strategy that effectively overcomes these historical barriers through careful optimization of reaction conditions and reagent selection. This novel approach utilizes a protection-group strategy to control regioselectivity, ensuring that the nitro group is installed precisely at the C1 position relative to the amine, while the bromine atom remains intact at the C6 position throughout the sequence. By employing commercially available starting materials like 6-bromo-2-naphthylamine and utilizing standard industrial solvents such as dichloromethane, acetic acid, and toluene, the process eliminates the need for exotic catalysts or specialized high-pressure equipment. The integration of a simple iron powder reduction and a solvent-swap crystallization technique demonstrates a clear focus on operational simplicity and cost-efficiency, transforming a previously elusive intermediate into a viable commodity for large-scale pharmaceutical production.
Mechanistic Insights into Regioselective Nitration and Iron Reduction
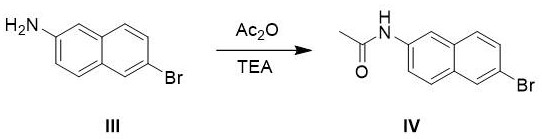
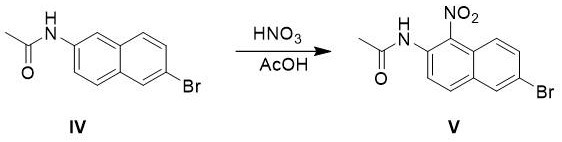
The core chemical innovation of this process lies in the strategic manipulation of electronic effects on the naphthalene ring system to achieve high purity. In the initial acetylation step, the conversion of the free amine to an acetamide (Compound IV) is not merely a protective measure but a critical electronic tuning step. The acetamide group is less activating than the free amine, which prevents over-nitration and oxidative degradation of the ring system when exposed to fuming nitric acid in the subsequent step. This moderation allows the nitration to proceed under mild thermal conditions (35-45°C), favoring the kinetically controlled formation of the 1-nitro isomer (Compound V) while minimizing the formation of dinitro byproducts or oxidation of the sensitive naphthalene core. The use of acetic anhydride alongside nitric acid likely generates the acetyl nitrate species in situ, a potent nitrating agent that operates efficiently in the acetic acid solvent matrix.
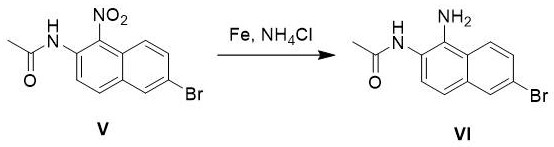
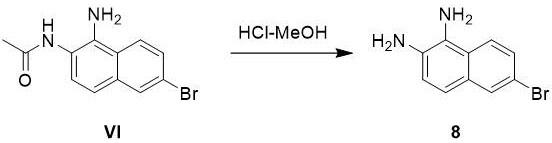
Following nitration, the reduction of the nitro group to the amine is achieved using iron powder in the presence of ammonium chloride, a classic Bechamp-type reduction adapted for modern process efficiency. This heterogeneous reaction proceeds via a single-electron transfer mechanism where the iron surface facilitates the stepwise reduction of the nitro group through nitroso and hydroxylamine intermediates to the final amine (Compound VI). The inclusion of ammonium chloride serves to maintain a slightly acidic environment that prevents the precipitation of insoluble iron oxides on the metal surface, thereby maintaining reaction kinetics and ensuring complete conversion without the need for expensive noble metal catalysts like palladium on carbon. The final hydrolysis step utilizes concentrated hydrochloric acid in methanol to cleave the acetamide protecting group, regenerating the free amine. The subsequent solvent exchange to toluene exploits the differential solubility of the diamine salt versus neutral organic impurities, driving the crystallization of the final product with exceptional purity (>99%) suitable for direct use in coupling reactions for HCV inhibitor synthesis.
How to Synthesize 6-Bromonaphthalene-1,2-Diamine Efficiently
The synthesis protocol outlined in the patent provides a clear, reproducible pathway for manufacturing this critical intermediate, emphasizing temperature control and phase separation techniques to maximize yield. The process begins with the acylation of the starting amine in dichloromethane, followed by a rigorous aqueous workup to remove excess acid and base before proceeding to the nitration stage. Critical attention is paid to the exothermic nature of the nitration, requiring slow addition of the nitrating mixture to maintain the temperature between 35°C and 45°C, which is essential for safety and selectivity. The reduction step utilizes tetrahydrofuran as a solvent to ensure good suspension of the iron powder, and the final isolation involves a clever azeotropic distillation to switch from methanol to toluene, inducing precipitation of the product without the need for energy-intensive chromatography. For detailed operational parameters, stoichiometry, and safety precautions, please refer to the standardized synthesis guide below.
- Acetylate 6-bromo-2-naphthylamine using acetic anhydride and triethylamine with DMAP catalyst in dichloromethane to form the protected intermediate.
- Perform regioselective nitration using fuming nitric acid and acetic anhydride in acetic acid at 35-45°C, followed by ethanol/water recrystallization.
- Reduce the nitro group using iron powder and ammonium chloride in tetrahydrofuran at 55-65°C, followed by filtration and saline washing.
- Hydrolyze the acetyl group using concentrated hydrochloric acid in methanol, then swap solvent to toluene for crystallization and filtration.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this patented methodology offers substantial strategic benefits by de-risking the sourcing of key HCV inhibitor intermediates. The reliance on bulk commodity chemicals such as acetic anhydride, iron powder, and common organic solvents significantly insulates the manufacturing process from the volatility associated with specialized reagent markets. This shift towards ubiquitous raw materials ensures a stable supply chain that is less susceptible to geopolitical disruptions or supplier monopolies, thereby enhancing the continuity of supply for downstream API production. Furthermore, the elimination of complex purification steps like column chromatography in favor of crystallization and filtration drastically reduces processing time and solvent consumption, leading to a leaner manufacturing footprint that aligns with modern green chemistry initiatives and cost-reduction targets.
- Cost Reduction in Manufacturing: The replacement of catalytic hydrogenation or hydride reductions with iron powder represents a direct and significant decrease in raw material costs, as iron is orders of magnitude cheaper than noble metals or complex reducing agents. Additionally, the high purity achieved through simple recrystallization eliminates the need for expensive preparative HPLC or multiple distillation steps, reducing both utility costs and waste disposal fees associated with solvent-heavy purification protocols. The overall atom economy and yield profile of the four-step sequence contribute to a lower cost of goods sold (COGS), allowing for more competitive pricing in the global pharmaceutical intermediate market.
- Enhanced Supply Chain Reliability: By utilizing a linear synthesis route with robust, high-yielding steps, the process minimizes the risk of batch failures that can disrupt just-in-time delivery schedules. The use of standard reactor configurations (stainless steel for acetylation/nitration, glass-lined for acid steps) means that the process can be easily transferred between different contract manufacturing organizations (CMOs) without requiring specialized hardware investments. This flexibility empowers procurement teams to diversify their supplier base, negotiating better terms and ensuring that production timelines for critical antiviral medications are met consistently without unexpected delays caused by technical bottlenecks.
- Scalability and Environmental Compliance: The process is explicitly designed for industrial scale-up, with demonstrated feasibility from kilogram to multi-hundred-kilogram batches, ensuring that supply can ramp up rapidly to meet market demand surges. The waste streams generated, primarily consisting of iron sludge and aqueous acidic washes, are well-characterized and can be treated using standard effluent treatment plant protocols, simplifying regulatory compliance and environmental permitting. The avoidance of chlorinated solvents in the final crystallization step (using toluene instead) further reduces the environmental impact and simplifies solvent recovery operations, aligning with increasingly strict global environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 6-bromonaphthalene-1,2-diamine, derived directly from the specific innovations detailed in patent CN112679365A. These insights are intended to clarify the process advantages for technical evaluators and supply chain planners who are assessing the viability of this intermediate for their own manufacturing pipelines. Understanding the nuances of the reaction conditions and workup procedures is essential for accurately forecasting production capabilities and quality assurance metrics.
Q: Why is the acetylation step critical before nitration in this synthesis?
A: The acetylation of the amine group serves two crucial purposes: it protects the amine from oxidation by nitric acid and acts as a moderate activating group that directs the incoming nitro group to the desired ortho-position (position 1) on the naphthalene ring, ensuring high regioselectivity and preventing the formation of unwanted isomers.
Q: What are the advantages of using iron powder for reduction in this process?
A: Using iron powder with ammonium chloride offers a significant cost advantage over catalytic hydrogenation or other metal reductions. It utilizes inexpensive, commercially available reagents, avoids the need for high-pressure equipment, and simplifies the workup procedure through simple filtration, making it highly suitable for large-scale industrial production.
Q: How does the final crystallization step ensure high purity of the diamine?
A: The process employs a solvent swap technique where methanol is removed via azeotropy with toluene. Since the final product has low solubility in toluene compared to polar impurities and salts, cooling the toluene mixture induces selective crystallization of the target 6-bromonaphthalene-1,2-diamine, achieving purity levels exceeding 99%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6-Bromonaphthalene-1,2-Diamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development and commercialization of life-saving antiviral therapies. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of 6-bromonaphthalene-1,2-diamine meets the exacting standards required for GMP API synthesis, providing our partners with the confidence needed to accelerate their drug development programs.
We invite you to engage with our technical procurement team to discuss how our optimized manufacturing processes can support your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our efficient synthesis route can lower your overall material costs. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to quality, reliability, and technical excellence in the supply of complex pharmaceutical intermediates.
