Advanced Resolution of Axially Chiral Oxindole Styrenes for Commercial Catalysis
The landscape of asymmetric synthesis is constantly evolving, driven by the pharmaceutical industry's insatiable demand for high-purity chiral building blocks. Patent CN111848322B introduces a groundbreaking methodology for the resolution of axially chiral oxindole-substituted styrene compounds, addressing a critical gap in current synthetic capabilities. Traditionally, accessing these complex stereoisomers has been fraught with challenges, often requiring harsh conditions or expensive transition metal catalysts that complicate downstream processing. This patent discloses a novel organocatalytic approach that leverages chiral phosphoric acids to achieve kinetic resolution with exceptional efficiency. By utilizing a mild reaction environment and readily available starting materials, this technology offers a robust pathway for generating structurally diverse axially chiral scaffolds. For R&D directors and process chemists, this represents a significant opportunity to streamline the production of high-value intermediates used in next-generation therapeutic agents and advanced catalytic systems.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of axially chiral styrene skeletons has relied heavily on nucleophilic addition reactions of alkynes or palladium-catalyzed coupling of aryl halides with hydrazones. These conventional pathways often suffer from significant drawbacks that impede their utility in commercial manufacturing. For instance, transition metal-catalyzed processes frequently necessitate rigorous exclusion of air and moisture, increasing operational complexity and cost. Furthermore, the removal of trace heavy metal residues from the final product is a stringent regulatory requirement for pharmaceutical intermediates, adding costly purification steps such as scavenging or recrystallization. Additionally, many existing methods exhibit limited substrate scope, failing to accommodate diverse functional groups without compromising yield or stereoselectivity. The reliance on stoichiometric chiral auxiliaries in some older protocols further exacerbates atom economy issues, generating substantial waste and reducing overall process efficiency.
The Novel Approach
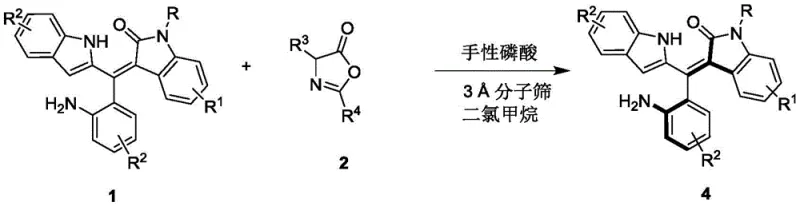
In stark contrast, the methodology outlined in CN111848322B employs a kinetic resolution strategy mediated by chiral phosphoric acid organocatalysts. This approach operates under remarkably mild conditions, typically between 0°C and 10°C in dichloromethane, which significantly reduces energy consumption and thermal stress on sensitive functional groups. The use of 3Å molecular sieves as an additive ensures the reaction environment remains anhydrous, protecting the catalyst's integrity and enhancing reproducibility. This organocatalytic system eliminates the need for toxic transition metals, thereby simplifying the purification workflow and ensuring a cleaner impurity profile. The reaction demonstrates broad substrate tolerance, accommodating various substituents on the indole and phenyl rings while maintaining high enantiomeric excess. This versatility makes it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates, offering a sustainable and economically viable alternative to traditional metal-catalyzed routes.
Mechanistic Insights into Chiral Phosphoric Acid Catalyzed Resolution
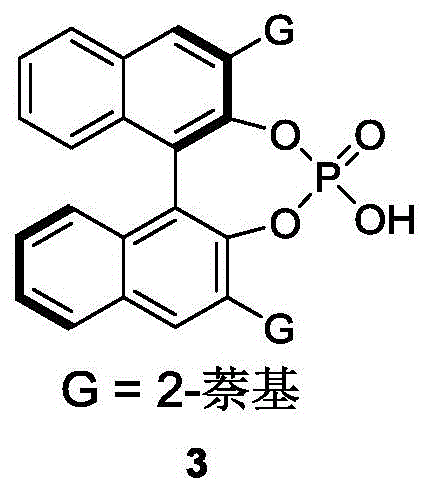
The core of this technological advancement lies in the precise stereochemical control exerted by the chiral phosphoric acid catalyst. These catalysts, often derived from BINOL scaffolds substituted with bulky groups like 2-naphthyl, function through a dual hydrogen-bonding activation mechanism. The acidic proton of the phosphate group activates the electrophilic species, while the phosphoryl oxygen interacts with the nucleophile or substrate, organizing the transition state within a well-defined chiral pocket. This rigid spatial arrangement forces the reaction to proceed through a specific trajectory, effectively discriminating between the enantiomers of the racemic starting material. The steric bulk of the catalyst's 3,3'-substituents plays a pivotal role in this differentiation, blocking one face of the substrate and directing the attack to the other. Such mechanistic precision allows for the isolation of the desired axially chiral product with optical purity reaching up to 96% ee, as demonstrated in the patent examples.
From an impurity control perspective, the mild nature of this organocatalytic cycle is paramount. High-temperature processes often promote racemization or decomposition of axially chiral compounds due to the relatively low rotational barrier of the biaryl bond. By maintaining the reaction temperature at 0°C, the kinetic stability of the product is preserved, preventing erosion of optical purity over time. Furthermore, the absence of reactive metal species minimizes the formation of metal-complexed byproducts that are notoriously difficult to separate. The use of molecular sieves not only sequesters water produced during the reaction but also prevents hydrolysis of sensitive intermediates. This combination of low thermal energy and chemical inertness ensures a clean reaction profile, facilitating easier downstream processing and yielding a high-purity final product suitable for sensitive biological applications.
How to Synthesize Axially Chiral Oxindole Styrenes Efficiently
The synthesis protocol described in the patent is designed for operational simplicity and robustness, making it highly attractive for process development teams. The procedure involves mixing the racemic oxindole-substituted styrene precursor with an azlactone derivative in the presence of the chiral catalyst. The reaction is heterogeneous due to the presence of molecular sieves, which requires efficient stirring to ensure mass transfer. Monitoring is typically conducted via thin-layer chromatography (TLC), allowing for real-time assessment of conversion. Once the reaction reaches completion, the workup is straightforward, involving simple filtration to remove the solid sieves followed by solvent evaporation. The crude product is then purified using standard silica gel column chromatography, a technique familiar to any synthetic laboratory. For a detailed breakdown of the specific molar ratios, solvent volumes, and purification parameters, please refer to the standardized guide below.
- Combine Compound 1 and Compound 2 in dichloromethane with 3Å molecular sieves and 10 mol% chiral phosphoric acid catalyst.
- Stir the reaction mixture at 0°C to 10°C and monitor progress via TLC until completion.
- Filter off the molecular sieves, concentrate the filtrate, and purify the residue using silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this organocatalytic resolution method presents compelling economic and logistical benefits. The shift away from precious metal catalysts like palladium or rhodium immediately removes a major cost driver and supply chain bottleneck associated with volatile metal prices. Moreover, the simplified purification process reduces the consumption of specialized scavenging resins and solvents, leading to significant cost savings in raw material expenditure. The mild reaction conditions also translate to lower energy requirements for heating or cooling, contributing to a reduced carbon footprint and lower utility costs. From a supply continuity perspective, the starting materials are commercially accessible or easily synthesized, ensuring a stable feedstock for continuous manufacturing operations. The robustness of the reaction against minor variations in conditions further enhances reliability, minimizing batch failures and ensuring consistent delivery schedules for downstream clients.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the associated heavy metal removal steps drastically lowers the cost of goods sold. Organocatalysts are generally more affordable and easier to handle than their metallic counterparts, reducing both material and safety compliance costs. The high atom economy of the resolution process means less waste generation, which in turn lowers waste disposal fees. Additionally, the ability to run the reaction at near-ambient temperatures reduces the energy load on reactor jackets and chillers. These cumulative efficiencies result in a leaner manufacturing process that maximizes margin potential without compromising on quality standards.
- Enhanced Supply Chain Reliability: Relying on organocatalysis mitigates the risk of supply disruptions often seen with specialty metal catalysts that may have single-source suppliers. The reagents used, such as dichloromethane and molecular sieves, are commodity chemicals with stable global supply chains. The simplicity of the workup procedure reduces the turnaround time between batches, allowing for higher throughput in existing facilities. This agility enables manufacturers to respond more quickly to fluctuating market demands. Furthermore, the high selectivity of the process reduces the need for extensive recycling of unreacted material, streamlining the overall production flow and ensuring a steady output of high-purity intermediates.
- Scalability and Environmental Compliance: The protocol is inherently scalable, as it avoids exothermic hazards associated with some metal-catalyzed couplings. The use of standard solvents and purification techniques facilitates technology transfer from lab to pilot to commercial scale. Environmentally, the process aligns with green chemistry principles by avoiding toxic heavy metals and operating under mild conditions. This reduces the burden on wastewater treatment systems and simplifies regulatory compliance regarding residual metal limits in APIs. The high yield and selectivity also mean less solvent is required per kilogram of product, reducing the overall environmental impact of the manufacturing campaign.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and performance of this resolution technology. These insights are derived directly from the experimental data and specifications provided in the patent documentation. Understanding these details is crucial for evaluating the feasibility of integrating this method into your existing production workflows. The answers highlight the versatility and robustness of the chiral phosphoric acid system.
Q: What enantiomeric excess (ee) can be achieved with this resolution method?
A: The patented method achieves high enantioselectivity, with experimental results demonstrating ee values up to 96% for various substrates.
Q: What type of catalyst is required for this asymmetric resolution?
A: The process utilizes a chiral phosphoric acid catalyst, specifically BINOL-derived derivatives such as compound 3, which effectively controls stereochemistry.
Q: Are the reaction conditions suitable for large-scale manufacturing?
A: Yes, the reaction operates at mild temperatures (0-10°C) in common solvents like dichloromethane, avoiding extreme conditions that hinder scalability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Axial Chiral Oxindole Styrene Supplier
The technical innovations described in CN111848322B underscore the immense potential of axially chiral oxindole styrenes in modern asymmetric synthesis. At NINGBO INNO PHARMCHEM, we possess the expertise to translate such complex laboratory methodologies into reliable commercial realities. As a premier CDMO partner, we have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch meets the exacting standards required by global pharmaceutical regulators. We understand the critical nature of chiral integrity and employ advanced analytical techniques to guarantee optical purity throughout the manufacturing lifecycle.
We invite you to collaborate with us to leverage this cutting-edge resolution technology for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and quality targets. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments. Let us help you secure a competitive advantage through superior supply chain solutions and advanced chemical manufacturing capabilities.
