Advanced Synthesis of Trans-3-Thiophenyl-Gamma-Lactone for Commercial Flavor Applications
Advanced Synthesis of Trans-3-Thiophenyl-Gamma-Lactone for Commercial Flavor Applications
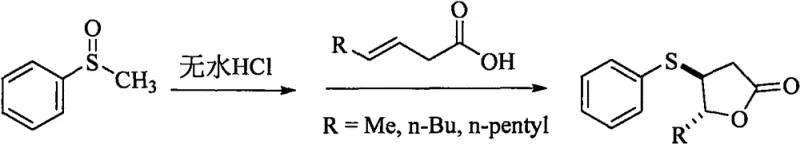
The global demand for high-purity sulfur-containing organic compounds continues to surge, driven by their critical roles in the flavor, fragrance, and pharmaceutical sectors. Patent CN113754616A introduces a groundbreaking preparation method for trans-3-thiophenyl-gamma-lactone, a compound renowned for its potent meat-like flavor profile. This technology addresses long-standing challenges in organic synthesis by offering a route that combines exceptional stereoselectivity with operational simplicity. For R&D directors and procurement specialists, understanding the nuances of this patent is essential for securing a reliable supply chain of high-value flavor intermediates. The method leverages the electrophilic properties of sulfur in a novel way, utilizing methyl phenyl sulfoxide and anhydrous hydrogen chloride to achieve yields ranging from 80% to 91%. This represents a significant leap forward compared to traditional methodologies that often struggle with isomeric purity and reagent stability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of thiophenyl groups into organic frameworks has relied heavily on reagents such as phenylsulfinylchloride, diphenyldisulfide, or direct thiophenolation using thiophenol. These conventional pathways are fraught with significant technical and commercial drawbacks that hinder efficient manufacturing. A primary concern for process chemists is the inherent instability of many sulfur-based electrophiles, which complicates storage, handling, and precise dosing during large-scale reactions. Furthermore, traditional methods involving the 1,4-conjugate addition of thiophenol to alpha,beta-unsaturated ketones frequently result in poor stereoselectivity. This lack of control leads to the formation of complex mixtures containing both cis and trans stereoisomers, necessitating costly and time-consuming purification steps to isolate the desired bioactive or flavor-active trans-isomer. The presence of unwanted isomers not only reduces the overall yield but can also negatively impact the sensory profile of the final flavor application, making these older routes commercially unviable for high-specification markets.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN113754616A utilizes a highly efficient strategy centered on the reaction between methyl phenyl sulfoxide and trans-3-olefinic acids under anhydrous acidic conditions. This approach fundamentally shifts the paradigm by using stable, readily available starting materials that eliminate the safety hazards associated with volatile thiols or unstable sulfinyl chlorides. The core innovation lies in the generation of anhydrous hydrogen chloride in situ, which activates the sulfoxide for a highly selective electrophilic addition to the olefinic double bond. This specific activation pathway ensures that the cyclization proceeds with rigorous stereocontrol, exclusively favoring the formation of the trans-3-thiophenyl-gamma-lactone structure. By bypassing the formation of cis-isomers entirely, this novel approach streamlines the downstream processing workflow, significantly reducing solvent consumption and waste generation associated with chromatographic separation of isomers.
Mechanistic Insights into Anhydrous HCl-Mediated Thiophenylation
To fully appreciate the technical superiority of this synthesis, one must delve into the mechanistic details of the electrophilic addition and subsequent lactonization. The reaction initiates with the interaction between methyl phenyl sulfoxide and anhydrous hydrogen chloride in an acetonitrile solvent system. The anhydrous nature of the HCl is critical, as water would compete for the electrophilic species and hydrolyze sensitive intermediates, leading to byproduct formation. Upon heating to 100°C, the activated sulfur species undergoes a concerted addition across the double bond of the trans-3-olefinic acid. This step is governed by the electronic properties of the sulfoxide oxygen and the protonated intermediate, which directs the attack to occur in a manner that preserves the trans-geometry of the starting alkene. The resulting intermediate then undergoes an intramolecular nucleophilic attack by the carboxylic acid group onto the sulfonium center, displacing the methyl group and closing the gamma-lactone ring. This cascade of events is highly exothermic and self-propagating once initiated, ensuring complete conversion of the starting materials within a 10-hour reflux period.
Impurity control in this system is inherently robust due to the specificity of the anhydrous conditions and the stoichiometry of the reagents. The use of a slight excess of methyl phenyl sulfoxide relative to the olefinic acid ensures that the limiting reagent is fully consumed, minimizing the presence of unreacted starting material in the crude mixture. Furthermore, the absence of strong oxidizing agents or radical initiators prevents the formation of sulfones or disulfides, which are common contaminants in other sulfur chemistry protocols. The reaction mixture, upon completion, contains primarily the target lactone and benign byproducts like methanol or dimethyl sulfide derivatives, which are easily removed during the aqueous workup and rotary evaporation steps. This clean reaction profile allows for high-purity isolation via standard silica gel column chromatography using petroleum ether and ethyl acetate gradients, yielding a product that meets stringent quality specifications for flavor and fragrance applications without the need for recrystallization or distillation.
How to Synthesize Trans-3-Thiophenyl-Gamma-Lactone Efficiently
Implementing this synthesis in a pilot or production plant requires careful attention to the generation and handling of anhydrous hydrogen chloride gas. The process begins with the controlled dripping of concentrated hydrochloric acid into concentrated sulfuric acid to generate dry HCl gas, which is then passed through a drying tower containing allochroic silica gel to ensure absolute moisture removal. This dry gas is absorbed into acetonitrile to create a standardized reagent solution, which is then charged into a reactor containing methyl phenyl sulfoxide. The detailed standardized synthesis steps, including specific molar ratios, temperature ramping rates, and quenching procedures, are outlined in the structured guide below to ensure reproducibility and safety compliance.
- Generate anhydrous hydrogen chloride gas by dripping concentrated HCl into concentrated sulfuric acid and drying through a silica gel tower.
- Absorb the anhydrous HCl into acetonitrile, add methyl phenyl sulfoxide, and heat the mixture to 100°C.
- Add trans-3-olefinic acid to the reaction mixture and reflux for 10 hours to obtain the target lactone with high stereoselectivity.
Commercial Advantages for Procurement and Supply Chain Teams
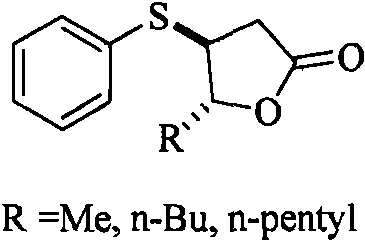
For procurement managers and supply chain heads, the transition to this patented methodology offers substantial strategic benefits that extend beyond mere chemical yield. The reliance on commodity chemicals such as methyl phenyl sulfoxide and various trans-3-olefinic acids (where R can be methyl, n-butyl, or n-pentyl) drastically simplifies the raw material sourcing landscape. Unlike specialized thiophenylating agents that may have limited suppliers and long lead times, the precursors for this reaction are widely available in the global chemical market, ensuring supply continuity even during periods of market volatility. This accessibility translates directly into reduced procurement risk and greater negotiating power when contracting with upstream vendors for bulk quantities of starting materials.
- Cost Reduction in Manufacturing: The economic implications of this process are profound, primarily driven by the elimination of expensive and hazardous reagents. By avoiding the use of unstable phenylsulfinylchloride or malodorous thiophenol, manufacturers save significantly on safety infrastructure, specialized containment equipment, and waste disposal costs associated with sulfur odors. The high stereoselectivity of the reaction means that nearly all input mass is converted into the desired product, effectively maximizing atom economy and reducing the cost per kilogram of the final active ingredient. Additionally, the simplified workup procedure, which avoids complex fractional distillations or multiple recrystallizations, lowers energy consumption and labor hours, contributing to a leaner and more cost-effective production model.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes to a more predictable and reliable manufacturing schedule. Because the reagents are stable and the reaction tolerates standard industrial glass-lined or stainless steel reactors, there is minimal risk of batch failure due to reagent degradation or sensitivity to trace impurities. This reliability allows for tighter production planning and shorter lead times for fulfilling customer orders. The ability to produce various derivatives (valerolactone, octalactone, nonalactone) using the same core platform technology further enhances supply chain flexibility, allowing manufacturers to pivot quickly between different flavor profiles based on market demand without retooling entire production lines.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process aligns well with modern green chemistry principles. The use of acetonitrile as a solvent, which is easily recovered and recycled, minimizes volatile organic compound (VOC) emissions. The absence of heavy metal catalysts or toxic sulfur byproducts simplifies wastewater treatment and reduces the regulatory burden associated with hazardous waste disposal. As global regulations on chemical manufacturing tighten, adopting a cleaner synthesis route like this future-proofs the supply chain against potential compliance issues, ensuring uninterrupted operations and maintaining a positive corporate sustainability profile.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear basis for decision-making regarding technology adoption and product specification.
Q: What are the primary advantages of this synthesis method over conventional thiophenylation?
A: Unlike conventional methods using thiophenol which suffer from poor stereoselectivity and cis/trans mixtures, this patent describes a route yielding exclusively the trans-isomer with 80-91% efficiency.
Q: Is this process suitable for large-scale manufacturing of flavor intermediates?
A: Yes, the process utilizes stable, commercially available raw materials like methyl phenyl sulfoxide and avoids unstable reagents, making it highly scalable for industrial production.
Q: What represents the key impurity control mechanism in this reaction?
A: The use of anhydrous HCl generated in situ ensures a controlled acidic environment that promotes specific cyclization while minimizing side reactions common in wet acidic conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trans-3-Thiophenyl-Gamma-Lactone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity sulfur-containing intermediates in the development of next-generation flavor and fragrance formulations. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of trans-3-thiophenyl-gamma-lactone meets the exacting standards required by top-tier multinational corporations.
We invite you to collaborate with us to leverage this innovative synthesis route for your specific application needs. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this technology can optimize your bill of materials. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and sample quantities for your evaluation.
