Advanced Visible-Light Organocatalysts for Efficient Asymmetric Hydroxylation in Pharmaceutical Manufacturing
The landscape of asymmetric synthesis is undergoing a transformative shift driven by the urgent need for sustainable and energy-efficient manufacturing processes. Patent CN107899611B introduces a groundbreaking class of organic catalysts that seamlessly integrate asymmetric organocatalysis with visible-light photocatalysis. This innovation addresses critical bottlenecks in the production of high-value chiral intermediates, specifically alpha-hydroxy-beta-dicarbonyl compounds, which are pivotal scaffolds in the synthesis of bioactive molecules and agrochemicals. By chemically bonding a chiral organic catalyst, such as a cinchona alkaloid derivative, with a visible-light photosensitizer like tetraphenylporphyrin, the inventors have created a robust bifunctional system. This hybrid architecture enables the activation of C-H bonds and the formation of asymmetric C-O bonds using molecular oxygen as the terminal oxidant under mild visible light irradiation, eliminating the reliance on hazardous UV sources and stoichiometric toxic oxidants.
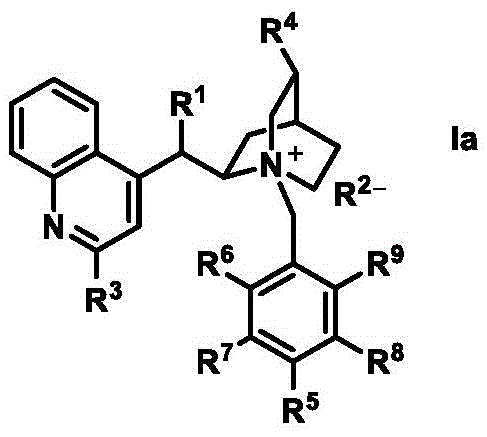
Historically, the asymmetric oxidation of carbonyl compounds has relied heavily on organometallic complexes or discrete dual-catalyst systems that present significant operational challenges. Conventional methods often necessitate the use of ultraviolet light, which poses safety risks and limits penetration depth in large-scale reactors, alongside expensive and potentially explosive oxidants such as organic peroxides or azaoxiranes. Furthermore, traditional organometallic approaches frequently involve complex ligand synthesis and the inevitable contamination of products with trace heavy metals, necessitating costly purification steps to meet stringent pharmaceutical standards. In contrast, the novel approach detailed in this patent leverages a single-molecule bifunctional catalyst that operates under ambient conditions. This strategy not only simplifies the reaction setup by removing the need for external photosensitizers but also utilizes atmospheric oxygen, a vastly superior oxidant in terms of atom economy and environmental safety. The covalent linkage ensures that the chiral environment and the photo-excited state are spatially proximate, enhancing the efficiency of energy and electron transfer processes essential for high enantioselectivity.
Mechanistic Insights into Visible-Light Driven Asymmetric Hydroxylation
The efficacy of this catalytic system stems from its sophisticated dual-activation mechanism, which mimics the precision of enzymatic catalysis. Upon irradiation with visible light (wavelengths ranging from 390 to 780 nm), the porphyrin moiety within the catalyst absorbs photon energy to reach an excited state. This excited species then interacts with ground-state triplet oxygen (3O2) to generate reactive oxygen species, likely singlet oxygen (1O2) or superoxide radicals, which serve as the active oxidizing agents. Simultaneously, the cinchona alkaloid portion of the catalyst engages the beta-dicarbonyl substrate through hydrogen bonding or ion-pairing interactions, organizing the substrate within a rigid chiral pocket. This precise orientation dictates the facial selectivity of the oxygen attack on the enol or enolate intermediate, thereby controlling the stereochemical outcome of the alpha-hydroxylation. The synergy between the photo-redox cycle and the chiral induction cycle allows for the transformation of prochiral substrates into optically active products with high fidelity, as evidenced by the successful synthesis of various alpha-chiral hydroxy-beta-dicarbonyl compounds.
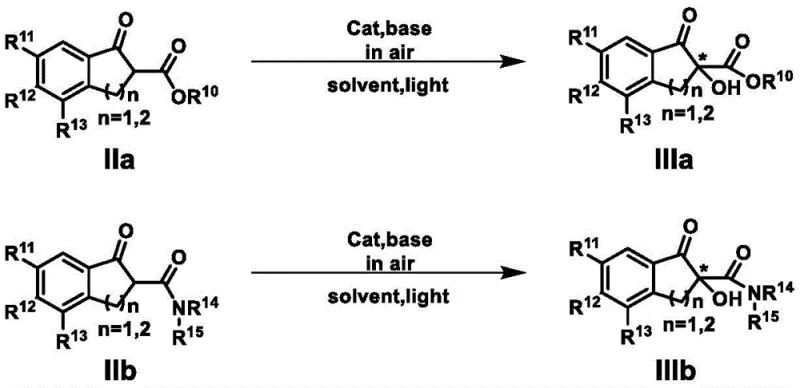
Impurity control is inherently managed by the mildness of the reaction conditions and the specificity of the catalyst. Unlike harsh oxidative conditions that can lead to over-oxidation or degradation of sensitive functional groups, this visible-light protocol operates at temperatures between -20°C and 50°C. The use of molecular oxygen minimizes the formation of side products associated with peroxide decomposition. Furthermore, the catalyst design facilitates easy separation from the reaction mixture; since the catalyst is a large, distinct organic molecule, it can often be removed via standard extraction or chromatography techniques without leaving metal residues. The patent data highlights that the catalyst remains stable throughout the process and can be recovered and reused, which further reduces the risk of catalyst-derived impurities accumulating in the final product stream, ensuring a cleaner impurity profile suitable for downstream pharmaceutical applications.
How to Synthesize Bifunctional Catalyst Ia-1 Efficiently
The preparation of these advanced catalysts is designed to be scalable and straightforward, utilizing standard organic synthesis techniques that are easily adaptable to industrial settings. The synthesis typically involves a nucleophilic substitution reaction where a halogenated photosensitizer precursor reacts with a hydroxyl-functionalized chiral amine under basic conditions. For instance, the preparation of catalyst Ia-1 involves reacting a cinchonidine derivative with a bromomethyl-substituted tetraphenylporphyrin in a solvent like dichloromethane. The reaction proceeds under nitrogen protection to prevent unwanted side reactions, with the addition of an aqueous base to facilitate the coupling. This modular approach allows for the variation of both the chiral backbone and the photosensitizer unit, enabling fine-tuning of the catalyst's properties for specific substrate classes.
- Prepare the reaction mixture by dissolving the asymmetric organic catalyst (e.g., Cinchonidine derivative Cn-1) and the visible light photosensitizer (e.g., TPP-1) in an appropriate solvent such as dichloromethane.
- Maintain an inert atmosphere using nitrogen protection and add an aqueous alkaline solution, such as 50% KOH, to initiate the coupling reaction at room temperature.
- Stir the mixture for 2 to 24 hours, then quench with water, extract the organic phase, dry over anhydrous sodium sulfate, and purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this technology offers substantial strategic benefits beyond mere technical performance. The shift towards visible-light photocatalysis represents a move away from resource-intensive processes that rely on precious metals and hazardous reagents. By eliminating the need for expensive transition metal catalysts and toxic oxidants, manufacturers can achieve significant cost reductions in raw material procurement and waste disposal. The simplified workup procedures, which avoid complex metal scavenging steps, translate directly into reduced processing time and lower operational expenditures. Additionally, the use of air as an oxidant removes the logistical burden and safety costs associated with storing and handling unstable peroxide reagents, thereby enhancing overall plant safety and regulatory compliance.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the elimination of costly noble metal catalysts and the replacement of expensive stoichiometric oxidants with free molecular oxygen. This fundamental change in reagent strategy drastically lowers the variable cost per kilogram of the produced intermediate. Furthermore, the ability to recycle the catalyst multiple times without significant loss of activity means that the effective catalyst loading can be kept extremely low over the lifetime of the production campaign. The mild reaction conditions also reduce energy consumption related to heating or cooling, contributing to a leaner and more cost-effective manufacturing footprint that aligns with modern green chemistry principles.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of readily available starting materials such as cinchona alkaloids and porphyrins, which are commercially accessible and do not suffer from the geopolitical supply constraints often associated with rare earth metals. The robustness of the catalyst, which maintains performance over multiple cycles as demonstrated in the patent examples, ensures consistent production throughput and reduces the frequency of catalyst replenishment orders. This stability minimizes the risk of production stoppages due to catalyst degradation or supply shortages, providing a more predictable and reliable source of high-purity pharmaceutical intermediates for downstream formulation.
- Scalability and Environmental Compliance: Scaling photochemical reactions has historically been challenging due to light penetration issues, but the use of visible light and the specific reactor configurations implied by this technology mitigate these concerns. The process generates minimal hazardous waste, primarily consisting of benign organic solvents that can be recovered and reused, significantly lowering the environmental burden. This aligns perfectly with increasingly strict global environmental regulations regarding waste discharge and carbon footprint. The ability to run reactions at ambient pressure and near-ambient temperatures simplifies the engineering requirements for scale-up, allowing for safer and faster transition from laboratory benchtop to commercial-scale production facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this visible-light catalytic technology. These insights are derived directly from the experimental data and embodiments provided in the patent documentation, offering clarity on performance metrics and operational parameters. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this methodology into existing production lines.
Q: What are the primary advantages of this bifunctional catalyst over traditional metal-based systems?
A: Unlike traditional organometallic systems that often require toxic heavy metals and harsh oxidants like peroxides, this bifunctional organocatalyst utilizes molecular oxygen from air as a green oxidant under mild visible light irradiation, significantly reducing environmental impact and purification costs.
Q: Can the catalyst be recycled for multiple reaction cycles?
A: Yes, the patent data demonstrates that the catalyst exhibits excellent stability and can be recovered from the organic layer after extraction and reused for multiple cycles while maintaining high catalytic activity and enantioselectivity.
Q: What types of substrates are compatible with this visible-light hydroxylation method?
A: The system shows broad substrate applicability, effectively catalyzing the asymmetric alpha-hydroxylation of various beta-dicarbonyl compounds, including indanone and tetralone derivatives with different electronic substituents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bifunctional Organocatalyst Supplier
As the demand for sustainable and efficient synthetic routes grows, NINGBO INNO PHARMCHEM stands ready to support your development goals with our expertise in advanced catalytic technologies. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale discovery to industrial manufacturing is seamless and robust. Our commitment to quality is reflected in our stringent purity specifications and rigorous QC labs, which guarantee that every batch of catalyst or intermediate meets the highest international standards required by the pharmaceutical industry.
We invite you to engage with our technical procurement team to discuss how this visible-light organocatalyst technology can optimize your specific synthesis challenges. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the potential economic benefits tailored to your production volume. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive innovation and efficiency in your supply chain.
