Advanced Synthesis of Oxapium Iodide Intermediates for Scalable Pharmaceutical Manufacturing
Advanced Synthesis of Oxapium Iodide Intermediates for Scalable Pharmaceutical Manufacturing
The pharmaceutical industry constantly seeks robust synthetic routes that balance high purity with economic viability, particularly for established therapeutic agents like oxapium iodide, a potent spasmolytic agent marketed as ESPERAN®. Patent CN101121708A introduces a transformative methodology for the preparation of oxapium iodide (Formula Ia) and its analogues, addressing critical bottlenecks in traditional manufacturing. This innovation pivots away from scarce, difficult-to-source precursors, opting instead for a streamlined pathway originating from cyclohexyl phenyl ketone. By re-engineering the synthetic sequence to favor accessible starting materials and simplified purification protocols, this technology offers a compelling value proposition for reliable pharmaceutical intermediate suppliers aiming to optimize their production portfolios. The strategic shift not only enhances the feasibility of large-scale operations but also aligns with modern green chemistry principles by reducing solvent consumption associated with complex chromatographic separations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of oxapium iodide and its structural analogues has been hindered by reliance on specialized starting materials that are neither commercially abundant nor economically efficient to produce. Prior art, such as Japanese patents JP:56-25175 and JP:56-30971, typically utilizes 2-cyclohexyl-2-phenyl-4-tolysulfonyloxy-1,3-dioxolane as a key precursor. The procurement of this tosylate intermediate presents significant logistical challenges, as it is rarely available on the open market, forcing manufacturers to engage in multi-step pre-syntheses that inflate lead times. Furthermore, the purification of intermediates in these legacy processes heavily depends on column chromatography, a technique that is notoriously difficult to scale beyond laboratory settings due to high solvent usage, low throughput, and operational complexity. These factors collectively create a fragile supply chain vulnerable to disruptions and cost volatility, making the conventional route unsuitable for the rigorous demands of industrial API manufacturing.
The Novel Approach
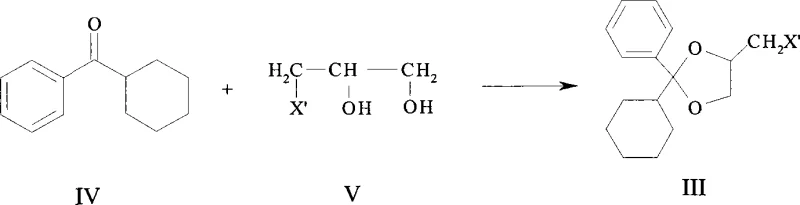
In stark contrast, the methodology disclosed in CN101121708A leverages a direct condensation strategy that bypasses the need for tosylate activation entirely. The novel route initiates with the reaction of cyclohexyl phenyl ketone and 3-chloro-1,2-propanediol under acid catalysis to directly generate the chloromethyl-dioxolane intermediate (Compound III). As illustrated in the reaction scheme above, this approach utilizes commodity chemicals that are inexpensive and readily available globally, effectively decoupling production from niche supply chains. The subsequent transformation involves a nucleophilic substitution with piperidine followed by quaternization, where the final product can be purified through simple solvent crystallization rather than chromatography. This fundamental redesign of the synthetic logic results in a drastic simplification of the operational workflow, enabling cost reduction in pharmaceutical intermediate manufacturing while simultaneously improving overall process safety and environmental compliance through reduced solvent waste.
Mechanistic Insights into Acid-Catalyzed Ketalization and Quaternization
The core of this synthetic innovation lies in the efficient formation of the 1,3-dioxolane ring system via acid-catalyzed ketalization. In the initial step, the carbonyl group of cyclohexyl phenyl ketone undergoes nucleophilic attack by the hydroxyl groups of 3-chloro-1,2-propanediol. The presence of a strong acid catalyst, such as p-toluenesulfonic acid or sulfuric acid, facilitates the protonation of the carbonyl oxygen, increasing its electrophilicity and driving the equilibrium towards the cyclic acetal product. Crucially, the reaction conditions are optimized to operate under reflux in solvents like toluene, often employing a water separator to continuously remove the water by-product, thereby pushing the reaction to completion according to Le Chatelier's principle. This mechanistic precision ensures high conversion rates, with experimental data indicating yields reaching up to 70% for the chloromethyl intermediate, establishing a robust foundation for downstream processing without the accumulation of difficult-to-remove impurities.
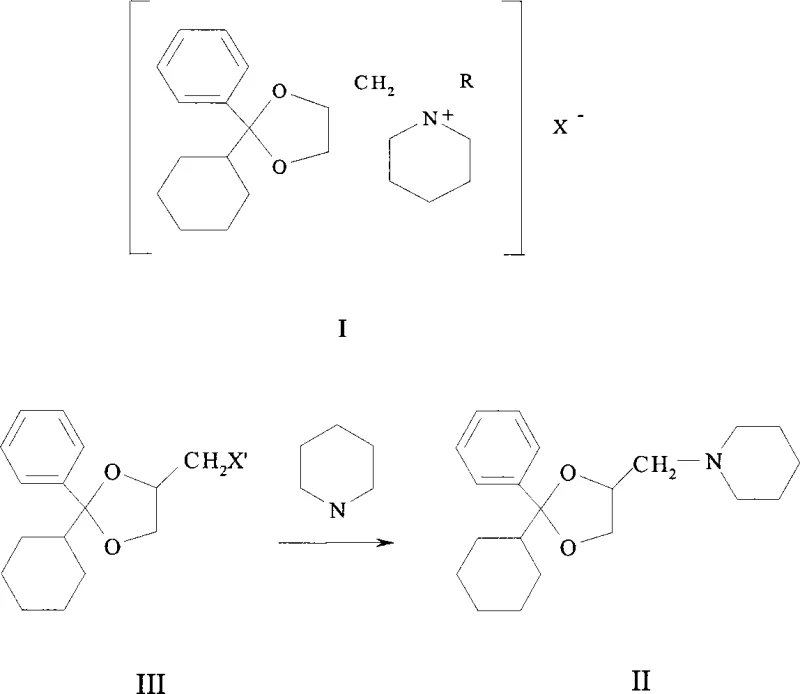
Following the formation of the dioxolane scaffold, the process proceeds through a classic nucleophilic substitution mechanism where the chloride leaving group is displaced by piperidine. As shown in the reaction diagram, the secondary amine attacks the chloromethyl carbon, displacing the chloride ion to form the tertiary amine intermediate (Compound II). This step is typically conducted in polar aprotic or protic solvents like acetonitrile or ethanol under reflux conditions to overcome the activation energy barrier. The final stage involves the quaternization of the piperidine nitrogen using an alkyl halide, specifically methyl iodide in the preferred embodiment. This exothermic reaction proceeds smoothly at ambient temperatures, converting the neutral amine into the pharmacologically active quaternary ammonium salt. The entire sequence is designed to minimize side reactions, ensuring that the impurity profile remains manageable and amenable to purification via recrystallization, which is critical for meeting the stringent quality standards required for high-purity pharmaceutical intermediates.
How to Synthesize Oxapium Iodide Efficiently
The practical implementation of this synthesis requires careful control of reaction parameters to maximize yield and purity while maintaining operational simplicity. The process is divided into three distinct stages: the initial ketalization to form the chloromethyl intermediate, the amination step to introduce the piperidine moiety, and the final quaternization to generate the iodide salt. Each stage has been optimized in the patent examples to demonstrate reproducibility across different solvent systems, including toluene, benzene, acetonitrile, and ethanol. Operators should note that while the reaction temperatures can vary from 0°C to 150°C depending on the specific step, the preferred embodiments utilize reflux conditions for the condensation and substitution steps to ensure kinetic efficiency. For detailed standard operating procedures and specific stoichiometric ratios derived from the patent examples, please refer to the structured guide below.
- Condense cyclohexyl phenyl ketone with 3-chloro-1,2-propanediol under acid catalysis (e.g., tosic acid) in toluene to form the chloromethyl-dioxolane intermediate.
- React the chloromethyl intermediate with piperidine in a solvent like acetonitrile under reflux to achieve nucleophilic substitution, forming the piperidinylmethyl derivative.
- Perform quaternization by reacting the piperidine derivative with methyl iodide in methanol or ethanol at room temperature to yield the final oxapium iodide salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible strategic benefits that extend beyond mere technical feasibility. The most significant advantage is the radical simplification of the purification process; by eliminating the need for column chromatography, manufacturers can drastically reduce the volume of organic solvents consumed per kilogram of product. This reduction directly translates to lower waste disposal costs and a smaller environmental footprint, aligning with increasingly strict global regulations on industrial emissions. Furthermore, the reliance on commodity starting materials like cyclohexyl phenyl ketone mitigates the risk of supply disruptions often associated with specialized fine chemicals. This stability allows for more accurate forecasting and inventory management, ensuring continuous production schedules even in volatile market conditions. The ability to purify the final product through simple crystallization also implies a faster turnaround time between batches, enhancing the overall agility of the manufacturing facility.
- Cost Reduction in Manufacturing: The economic impact of this new method is profound, primarily driven by the substitution of expensive, hard-to-source tosylate precursors with cheap, bulk-available ketones and diols. Additionally, the removal of chromatographic purification steps eliminates the capital expenditure and operational costs associated with silica gel columns and the vast quantities of elution solvents they require. This streamlining of the process flow reduces labor hours and energy consumption, resulting in substantial cost savings that can be passed down the supply chain or reinvested into R&D. The higher overall yield reported in the patent examples further amplifies these savings by maximizing the output from every unit of raw material input, thereby improving the gross margin profile of the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: From a logistics perspective, sourcing cyclohexyl phenyl ketone and 3-chloro-1,2-propanediol is significantly less risky than procuring custom-synthesized tosylate intermediates. These base chemicals are produced by multiple vendors globally, creating a competitive marketplace that prevents price gouging and ensures consistent availability. This diversification of the supply base protects the manufacturer from single-source failures and geopolitical disruptions. Moreover, the simplified process chemistry reduces the complexity of the manufacturing schedule, allowing for more flexible production planning. The robustness of the reaction conditions means that minor variations in raw material quality can be tolerated without compromising the final product specification, adding another layer of resilience to the supply chain.
- Scalability and Environmental Compliance: The transition from laboratory-scale chromatography to industrial-scale crystallization is a critical enabler for commercial viability. Crystallization is a unit operation that scales linearly and predictably, whereas chromatography often faces diminishing returns and engineering challenges at the ton-scale. This scalability ensures that the process can meet the demands of commercial scale-up of complex antispasmodic intermediates without requiring prohibitive capital investment in new equipment. Environmentally, the reduction in solvent usage and waste generation supports corporate sustainability goals and facilitates compliance with environmental protection laws. The use of common solvents like ethanol and acetonitrile, which have well-established recovery and recycling protocols, further enhances the green credentials of the manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in CN101121708A, offering clarity on how this method outperforms legacy technologies. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer and for commercial teams assessing the long-term value of adopting this new supply channel. The answers reflect a commitment to transparency and technical accuracy, ensuring that all stakeholders have a clear understanding of the process capabilities and limitations.
Q: What is the primary advantage of the new synthesis method for oxapium iodide described in patent CN101121708A?
A: The primary advantage is the elimination of column chromatography purification. The new method uses readily available cyclohexyl phenyl ketone and allows for simple solvent crystallization, significantly reducing production costs and complexity compared to prior art using tosylate intermediates.
Q: What are the typical reaction conditions for the initial ketalization step?
A: The initial condensation between cyclohexyl phenyl ketone and 3-chloro-1,2-propanediol is typically conducted under reflux conditions (approx. 110°C in toluene) for about 12 hours using an acid catalyst such as p-toluenesulfonic acid or sulfuric acid, often with a water separator to drive the equilibrium.
Q: How does this process improve supply chain reliability for antispasmodic drug manufacturers?
A: By utilizing commodity chemicals like cyclohexyl phenyl ketone and 3-chloro-1,2-propanediol instead of specialized tosylate precursors, the process ensures a stable supply of raw materials. Furthermore, the simplified purification via crystallization rather than chromatography enhances batch consistency and throughput.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Oxapium Iodide Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced pharmaceutical intermediates requires more than just a patent; it demands deep process engineering expertise and a commitment to quality excellence. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this novel synthesis are fully realized in a GMP-compliant environment. We understand that reducing lead time for high-purity pharmaceutical intermediates is critical for our partners' drug development timelines. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of oxapium iodide or its analogues meets the highest international standards for safety and efficacy.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this cost-effective and scalable synthetic route. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Together, we can optimize your supply chain, reduce manufacturing costs, and accelerate the delivery of essential antispasmodic therapies to patients worldwide, solidifying your position as a leader in the generic and specialty pharmaceutical markets.
