Advanced Fluoroalkyl-Substituted Phosphine Ligands for High-Performance Transition Metal Catalysis
The pharmaceutical and fine chemical industries are constantly seeking next-generation catalytic solutions to overcome the limitations of traditional synthetic routes. Patent CN114621286A introduces a groundbreaking class of fluoroalkyl-substituted trivalent organic phosphine compounds that address critical gaps in transition metal catalysis. Unlike conventional electron-rich phosphine ligands, these novel compounds incorporate trifluoromethyl and polyfluoroalkyl groups directly onto the phosphorus atom. This structural modification fundamentally shifts the electronic landscape of the catalyst, creating electron-deficient centers that are uniquely suited to accelerate specific elementary steps in catalytic cycles. For R&D directors and process chemists, this represents a significant opportunity to optimize reaction kinetics and access chemical space that was previously difficult to navigate with standard triaryl or trialkyl phosphines.
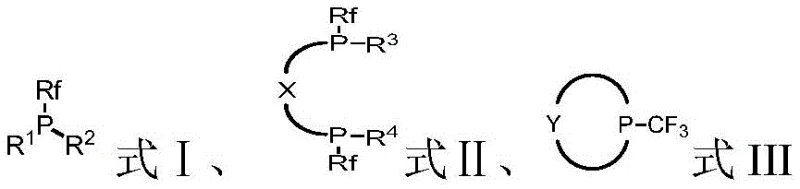
The development of these ligands stems from the recognition that while electron-rich ligands facilitate oxidative addition, electron-deficient ligands are crucial for promoting reductive elimination. By systematically introducing fluorine atoms, which possess the highest electronegativity of all elements, the patent describes a method to finely tune both the steric hindrance and electronic properties of the ligand. This dual tuning capability allows for the creation of highly active catalysts that do not require the delicate balance often needed in traditional ligand design. The versatility of the structure, allowing for various aryl, alkyl, and heterocyclic substituents alongside the fluoroalkyl group, ensures broad applicability across different synthetic challenges in the production of complex pharmaceutical intermediates and agrochemical active ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of specialized phosphine ligands, particularly those containing fluorinated groups, has been fraught with significant technical and economic hurdles. Traditional pathways typically rely on the reaction of phosphine chlorides with organolithium or Grignard reagents. These processes demand stringent cryogenic conditions, often requiring temperatures as low as -78°C to control reactivity and prevent decomposition. Such extreme conditions necessitate specialized equipment, high energy consumption for cooling, and rigorous safety protocols to handle pyrophoric reagents. Furthermore, the introduction of trifluoromethyl groups via these conventional nucleophilic substitutions is often inefficient, suffering from low atom economy and limited substrate scope. The multi-step nature of generating the necessary fluorinated nucleophiles adds complexity, increases waste generation, and prolongs lead times, making these methods less attractive for large-scale commercial manufacturing where cost and throughput are paramount concerns.
The Novel Approach
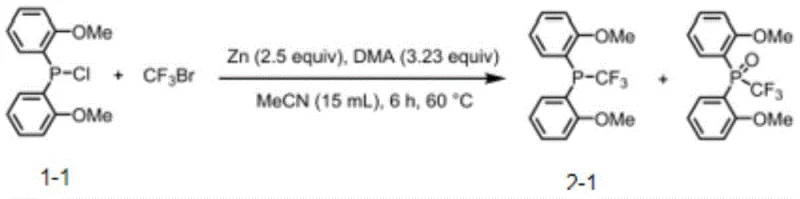
The methodology disclosed in the patent offers a transformative alternative by utilizing a direct, one-step reaction between trivalent phosphine halides and halogenated fluoroalkanes, specifically bromotrifluoromethane (CF3Br), in the presence of metallic zinc. This zinc-mediated trifluoromethylation proceeds under remarkably mild conditions, typically at temperatures ranging from 25°C to 60°C, completely eliminating the need for cryogenic cooling. The use of zinc powder as a reducing agent is not only cost-effective but also safer and easier to handle on an industrial scale compared to organometallic reagents. The addition of DMA (dimethylacetamide) as an additive further enhances the reaction yield and efficiency. This streamlined approach simplifies the workflow, reduces the number of unit operations, and significantly lowers the barrier to entry for producing high-performance fluorinated ligands. The robustness of this method is evidenced by its compatibility with a wide range of functional groups, allowing for the direct synthesis of complex ligand architectures without extensive protecting group strategies.
Mechanistic Insights into Zinc-Mediated Trifluoromethylation
The core innovation lies in the ability of the zinc/DMA system to activate the carbon-bromine bond of CF3Br, generating a reactive trifluoromethyl species that can effectively substitute the halogen on the phosphorus atom. Mechanistically, the zinc metal serves as a single-electron transfer agent, facilitating the formation of radical or organozinc intermediates that react with the phosphine chloride precursor. The presence of DMA likely stabilizes these intermediates or activates the zinc surface, ensuring a smooth conversion even at moderate temperatures. This mechanism bypasses the high energy barriers associated with direct nucleophilic attack by unstable trifluoromethyl anions. From a catalytic perspective, the resulting phosphine ligands exert a profound influence on the transition metal center, typically palladium or nickel. The strong electron-withdrawing nature of the CF3 group decreases the electron density on the phosphorus, which in turn renders the metal center more electrophilic. This electronic modulation is critical for accelerating the reductive elimination step, where the final product is released from the metal catalyst, often the bottleneck in cross-coupling reactions.
Furthermore, the steric environment around the phosphorus atom can be precisely engineered by selecting appropriate R groups (such as bulky aryl or cycloalkyl substituents) in conjunction with the fluoroalkyl moiety. This steric bulk prevents the formation of inactive catalyst aggregates and promotes the formation of low-coordinate, highly active metal species. The synergy between the electronic deficiency induced by the fluorine atoms and the steric protection provided by the substituents results in ligands that exhibit superior stability and turnover frequencies. This level of control allows chemists to tailor the catalyst specifically for challenging substrates, such as sterically hindered aryl chlorides or electron-deficient heterocycles, which are common motifs in modern drug discovery. The ability to fine-tune these parameters without compromising the simplicity of the ligand synthesis is a key advantage for developing robust manufacturing processes.
How to Synthesize Fluoroalkyl-Substituted Phosphine Compounds Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production. The process begins with the suspension of zinc powder in a solvent system comprising acetonitrile and potentially toluene or benzene, depending on solubility requirements. The phosphine chloride substrate is added, followed by the controlled introduction of the DMA additive. The reaction vessel is then pressurized or bubbled with CF3Br gas, maintaining a slight excess to drive the reaction to completion. Heating the mixture to 60°C for approximately 6 hours typically yields the crude product, which can be purified using standard silica gel chromatography. The detailed standardized synthesis steps for specific derivatives are provided in the guide below, ensuring reproducibility and consistency for technical teams looking to implement this chemistry.
- Prepare the reaction mixture by combining the trivalent phosphine chloride precursor with zinc powder (2.5 equivalents) and DMA additive (3.23 equivalents) in acetonitrile solvent.
- Introduce bromotrifluoromethane (CF3Br) gas into the reaction system, ensuring a molar ratio of approximately 2.32 equivalents relative to the phosphine chloride substrate.
- Heat the reaction mixture to 60°C and stir for 6 hours to facilitate the substitution reaction, followed by purification via silica gel chromatography to isolate the target fluoroalkyl phosphine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this technology offers tangible benefits beyond mere chemical performance. The shift from cryogenic, multi-step syntheses to a mild, one-pot process translates directly into significant cost reduction in fine chemical intermediates manufacturing. By eliminating the need for specialized low-temperature reactors and the handling of hazardous pyrophoric reagents, capital expenditure and operational safety costs are drastically reduced. The use of commodity chemicals like zinc powder and CF3Br ensures a stable and reliable supply chain, mitigating the risks associated with sourcing specialized organometallic reagents that may have long lead times or volatile pricing. Additionally, the simplified workup and purification procedures reduce solvent consumption and waste generation, aligning with increasingly stringent environmental regulations and sustainability goals.
- Cost Reduction in Manufacturing: The elimination of cryogenic conditions (-78°C) and the replacement of expensive organolithium reagents with inexpensive zinc powder fundamentally alter the cost structure of ligand production. This process intensification reduces energy consumption for cooling and heating, while the higher atom economy of the direct trifluoromethylation minimizes raw material waste. Consequently, the overall cost of goods sold (COGS) for these high-value ligands can be substantially lowered, making advanced catalytic solutions accessible for broader commercial applications without compromising quality or purity standards.
- Enhanced Supply Chain Reliability: Relying on stable, shelf-stable reagents like phosphine chlorides and zinc powder enhances supply chain resilience. Unlike moisture-sensitive Grignard reagents that require immediate use or special storage, the inputs for this process are robust and widely available from multiple global suppliers. This diversification of the supply base reduces the risk of production stoppages due to raw material shortages. Furthermore, the milder reaction conditions allow for processing in a wider range of manufacturing facilities, increasing flexibility and reducing dependency on specialized contract manufacturing organizations with niche capabilities.
- Scalability and Environmental Compliance: The straightforward nature of the zinc-mediated reaction facilitates seamless scale-up from gram to ton quantities. The absence of highly exothermic steps typical of organolithium additions improves process safety and control at larger scales. Moreover, the reduced generation of hazardous waste and the use of less toxic solvents contribute to a greener manufacturing footprint. This alignment with green chemistry principles not only simplifies regulatory compliance but also enhances the corporate sustainability profile, which is increasingly important for partnerships with major pharmaceutical and agrochemical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of these fluoroalkyl-substituted phosphine ligands. These insights are derived directly from the patent specifications and experimental data, providing a reliable foundation for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this technology into existing workflows and assessing its potential impact on project timelines and budgets.
Q: What is the primary advantage of fluoroalkyl-substituted phosphine ligands over traditional triarylphosphines?
A: The introduction of strongly electron-withdrawing fluoroalkyl groups, such as trifluoromethyl, significantly alters the electronic properties of the phosphorus center. This electron deficiency promotes the reductive elimination step in transition metal catalytic cycles, which is often the rate-determining step, thereby enhancing overall reaction efficiency and yield in coupling reactions.
Q: How does the zinc-mediated synthesis method improve scalability compared to conventional routes?
A: Conventional methods often rely on cryogenic conditions (-78°C) and pyrophoric reagents like organolithiums or Grignard reagents. The patented zinc-mediated approach operates at mild temperatures (60°C) using stable zinc powder and gaseous CF3Br. This eliminates the need for expensive cryogenic infrastructure and enhances operational safety, making commercial scale-up from kilograms to tons significantly more feasible and cost-effective.
Q: In which types of organic reactions have these ligands demonstrated superior performance?
A: These ligands have shown exceptional catalytic activity in carbon-carbon bond coupling reactions, such as Negishi and Sonogashira couplings, as well as carbon-heteroatom bond formations like C-N coupling. The unique steric and electronic tuning allows for high turnover numbers and excellent yields even with challenging substrates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluoroalkyl Phosphine Ligands Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating advanced academic research into commercial reality. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the innovative synthesis methods described in patent CN114621286A can be effectively deployed at an industrial level. Our state-of-the-art facilities are equipped to handle the specific requirements of fluorine chemistry and phosphine handling, adhering to stringent purity specifications and rigorous QC labs to guarantee the highest quality ligands for your catalytic processes. We understand the critical role these ligands play in your synthesis and are committed to delivering consistent, high-performance materials.
We invite you to collaborate with us to explore how these novel ligands can optimize your specific synthetic routes. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your project needs, demonstrating the economic viability of switching to this superior technology. Please contact our technical procurement team to request specific COA data and route feasibility assessments, and let us help you accelerate your development timeline with reliable, high-quality catalytic solutions.
