Revolutionizing Gamma-Aryl Ketone Production with Cost-Efficient Nickel Catalysis for Commercial Scale
The landscape of organic synthesis for complex ketone derivatives is undergoing a significant transformation, driven by the urgent need for more sustainable and cost-effective catalytic systems. Patent CN112110801B introduces a groundbreaking methodology for the synthesis of gamma-aryl substituted ketone compounds, utilizing tertiary cyclobutanol derivatives and halogenated aromatic hydrocarbons as key substrates. This innovation specifically leverages a nickel-catalyzed system to achieve selective remote gamma-position arylation, a transformation that has historically been challenging to execute with high precision. By shifting away from traditional noble metal catalysts, this technology offers a compelling value proposition for the fine chemical and pharmaceutical industries, promising to streamline the production of high-value intermediates while drastically reducing raw material costs associated with precious metal procurement.
Historically, the functionalization of arylalkyl ketones at the alpha and beta positions has been well-established through various mature synthetic routes. However, achieving high selectivity at the remote gamma-position has remained a formidable obstacle for process chemists. Conventional approaches often rely on radical-mediated beta-homolytic cleavage processes, which typically necessitate the use of stoichiometric strong oxidants or complex photocatalytic setups. These traditional methods not only generate substantial chemical waste, complicating downstream purification and environmental compliance, but they also involve cumbersome operational steps that hinder scalability. Furthermore, existing transition metal-catalyzed alternatives have predominantly depended on expensive noble metals such as palladium or rhodium, creating a significant economic bottleneck for large-scale manufacturing operations.
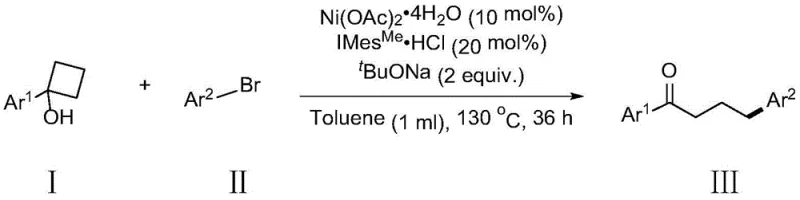
The novel approach detailed in this patent fundamentally disrupts these limitations by employing an inexpensive nickel catalytic system, specifically nickel acetate tetrahydrate, to drive the reaction. This method capitalizes on the inherent ring strain of tertiary cyclobutanol derivatives, facilitating a beta-carbon elimination pathway that selectively installs the aryl group at the gamma-position. Unlike previous literature reports that required costly palladium or rhodium complexes, this nickel-based protocol utilizes earth-abundant metals, thereby aligning with green chemistry principles and significantly lowering the barrier to entry for commercial production. The reaction conditions are robust, operating effectively in common organic solvents like toluene at elevated temperatures, demonstrating a practical feasibility that is highly attractive for industrial adoption.
Mechanistic Insights into Nickel-Catalyzed Beta-Carbon Elimination
The core of this synthetic breakthrough lies in the unique mechanistic pathway involving nickel-catalyzed beta-carbon elimination. The reaction initiates with the oxidative addition of the aryl bromide to the low-valent nickel species generated in situ from the nickel acetate precursor and the N-heterocyclic carbene ligand. This active nickel complex then interacts with the tertiary cyclobutanol substrate, where the relief of ring strain serves as the thermodynamic driving force for the cleavage of the carbon-carbon bond adjacent to the hydroxyl-bearing carbon. This beta-carbon elimination step is critical, as it generates a distal organonickel intermediate that positions the metal center precisely at the gamma-carbon relative to the incipient carbonyl group.
Following the carbon-carbon bond cleavage, the resulting alkoxide-nickel species undergoes reductive elimination to forge the new carbon-carbon bond between the gamma-position of the ketone chain and the aryl group from the bromide. This mechanism ensures high regioselectivity, avoiding the formation of unwanted alpha- or beta-arylated byproducts that often plague less selective methods. The use of a bulky carbene ligand, derived from the specified imidazolium salt precursor, plays a pivotal role in stabilizing the active nickel species and promoting the specific elimination pathway over competing side reactions. This precise control over the catalytic cycle is essential for maintaining high purity profiles in the final product, minimizing the burden on downstream purification processes.
How to Synthesize Gamma-Aryl Substituted Ketones Efficiently
The synthesis protocol outlined in the patent provides a clear and reproducible pathway for generating these valuable ketone derivatives. The process involves combining the substituted cyclobutanol and aryl bromide substrates with the nickel catalyst system in a suitable organic solvent. The reaction is conducted under an inert atmosphere to prevent catalyst deactivation, typically requiring heating to temperatures around 130°C for a duration of approximately 36 hours to ensure complete conversion. Following the reaction period, a straightforward workup procedure involving dilution, adsorption onto silica gel, and column chromatography allows for the isolation of the target gamma-aryl ketone with high purity.
- Prepare the reaction mixture by combining substituted cyclobutanol, aryl bromide, nickel acetate tetrahydrate catalyst, carbene precursor, and sodium tert-butoxide in toluene solvent.
- Heat the reaction mixture to 130°C under nitrogen protection and stir for 36 hours to facilitate the ring-opening and arylation process.
- Cool the reaction, dilute with ethyl acetate, adsorb onto silica gel, and purify via column chromatography to isolate the target gamma-aryl ketone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this nickel-catalyzed technology presents a strategic opportunity to optimize manufacturing costs and enhance supply reliability. The substitution of expensive noble metal catalysts with nickel acetate tetrahydrate represents a direct and significant reduction in raw material expenditure. Since nickel salts are commodity chemicals with stable global supply chains, manufacturers can mitigate the price volatility often associated with precious metals like palladium and rhodium. This stability allows for more accurate long-term budgeting and reduces the risk of production delays caused by catalyst shortages or sudden price spikes in the precious metals market.
- Cost Reduction in Manufacturing: The economic impact of switching to a nickel-based catalyst system is profound, primarily due to the drastic difference in metal pricing. Nickel acetate is orders of magnitude cheaper than palladium or rhodium complexes, which directly lowers the bill of materials for every batch produced. Additionally, the elimination of stoichiometric oxidants required in radical-mediated methods further reduces reagent costs and waste disposal fees. This cumulative effect results in a leaner cost structure, enabling competitive pricing for the final pharmaceutical intermediates while maintaining healthy profit margins for the manufacturer.
- Enhanced Supply Chain Reliability: Relying on earth-abundant metals like nickel significantly de-risks the supply chain compared to dependence on geographically concentrated precious metals. Nickel is widely mined and processed globally, ensuring a consistent and reliable flow of raw materials. This abundance translates to shorter lead times for catalyst procurement and greater flexibility in sourcing. For supply chain heads, this means improved continuity of operations and the ability to scale production volumes rapidly in response to market demand without being constrained by the availability of specialized noble metal catalysts.
- Scalability and Environmental Compliance: The process design inherently supports scalability, utilizing simple reaction conditions and common solvents like toluene that are easily managed in large-scale reactors. The avoidance of stoichiometric oxidants and heavy metal waste simplifies the effluent treatment process, reducing the environmental footprint of the manufacturing site. This alignment with stricter environmental regulations minimizes compliance risks and potential fines, while the simplified workup procedure enhances overall throughput efficiency, allowing facilities to maximize their production capacity with existing infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this nickel-catalyzed synthesis method. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on substrate scope, reaction conditions, and product quality. Understanding these details is crucial for R&D teams evaluating the feasibility of integrating this technology into their existing process workflows.
Q: What is the primary advantage of using nickel catalysts over palladium for this synthesis?
A: The primary advantage is cost reduction and resource availability. Nickel acetate tetrahydrate is significantly cheaper and more abundant than noble metals like palladium or rhodium, which reduces the overall production cost of the pharmaceutical intermediates without compromising selectivity.
Q: What types of substrates are compatible with this nickel-catalyzed method?
A: The method demonstrates broad substrate scope, accommodating various tertiary cyclobutanols with naphthyl or substituted phenyl groups, and aryl bromides including naphthyl, methyl-substituted, ethyl-substituted, and phenyl-substituted benzene rings.
Q: How does this method address the challenge of gamma-position functionalization?
A: Traditional methods often struggle with remote functionalization. This protocol utilizes the ring strain of tertiary cyclobutanols to drive a beta-carbon elimination process, selectively installing the aryl group at the remote gamma-position relative to the carbonyl group.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Gamma-Aryl Ketones Supplier
As the demand for efficient and cost-effective synthetic routes for complex organic molecules grows, partnering with an experienced CDMO becomes essential for bringing these innovations to market. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial plant is seamless and robust. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs, guaranteeing that every batch of gamma-aryl ketones meets the exacting standards required by the global pharmaceutical industry.
We invite you to collaborate with us to leverage this advanced nickel-catalyzed technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this method can optimize your budget. Please contact us today to request specific COA data and route feasibility assessments, and let us help you secure a reliable supply of high-purity intermediates for your next generation of therapeutic products.
