Advanced C-H Activation Strategy for Scalable Nitrogen Heterocycle Intermediate Production
Advanced C-H Activation Strategy for Scalable Nitrogen Heterocycle Intermediate Production
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to access complex nitrogen-containing heterocycles, which serve as the backbone for countless active pharmaceutical ingredients (APIs) ranging from analgesics like morphine derivatives to antibiotics like quinolones. Patent CN112679426A introduces a groundbreaking universal method for synthesizing these critical scaffolds through a high-efficiency carbon-hydrogen bond nitrogen-oxygen double functionalization strategy. This technology represents a significant leap forward from conventional step-wise syntheses, offering a streamlined route to a versatile intermediate compound, designated as Formula (I), which can be subsequently derivatized into multiple distinct heterocyclic families. By leveraging direct C-H activation, this process minimizes waste and maximizes atomic economy, addressing the growing demand for sustainable and cost-effective pharmaceutical intermediate manufacturing.
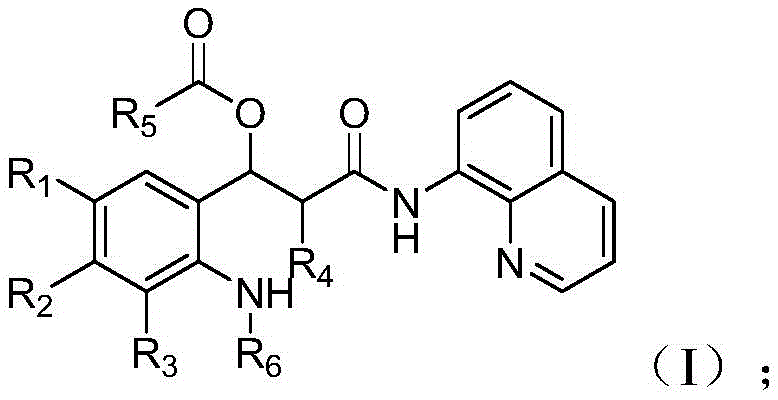
The core innovation lies in the structural flexibility of Intermediate Compound I, which acts as a pivotal junction point for divergent synthesis. As illustrated in the general formula, the molecule accommodates a wide array of substituents at positions R1 through R6, allowing chemists to tune the electronic and steric properties of the final product without altering the fundamental synthetic workflow. This adaptability is crucial for reliable pharmaceutical intermediate suppliers who must cater to diverse client specifications while maintaining a standardized production protocol. The ability to generate such complexity from simple starting materials underscores the strategic value of this patent in modern organic synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of nitrogen-containing heterocycles has been plagued by a lack of universality, where specific methodologies were often restricted to producing only one particular type of heterocyclic ring system. For instance, existing patents might disclose a method exclusively for thienopyridines or another solely for quaternary salts, forcing manufacturers to maintain separate, non-interchangeable production lines for different chemical entities. This fragmentation leads to significant inefficiencies in cost reduction in pharmaceutical intermediate manufacturing, as each new target molecule requires the development of a unique set of reaction conditions, catalysts, and purification protocols. Furthermore, traditional routes frequently rely on pre-functionalized starting materials, necessitating additional synthetic steps to install leaving groups or activating moieties prior to the cyclization event, which inherently lowers the overall yield and increases the environmental footprint of the process.
The Novel Approach
In stark contrast, the novel approach detailed in this patent utilizes a direct carbon-hydrogen bond bifunctional synthesis that bypasses the need for pre-functionalization, thereby collapsing multiple synthetic steps into a single, efficient transformation. By employing amides, azodicarboxylates, and carboxylic acids as readily available raw materials, the process constructs the complex skeleton of Intermediate I in one pot under the action of a palladium catalyst. This convergence not only simplifies the operational complexity but also enhances the commercial scale-up of complex pharmaceutical intermediates by reducing the number of isolation and purification stages required. The resulting intermediate serves as a universal precursor, capable of being converted into various nitrogen-containing heterocycles (Types A, B, and C) through straightforward downstream modifications, offering unparalleled versatility to R&D teams exploring new chemical space.
Mechanistic Insights into Pd-Catalyzed C-H Bond Bifunctionalization
The mechanistic elegance of this transformation relies on the precise orchestration of a palladium-catalyzed cycle that simultaneously activates inert C-H bonds and installs nitrogen and oxygen functionalities. The reaction initiates with the coordination of the palladium catalyst, such as palladium acetate or palladium pivalate, to the amide substrate, facilitating the cleavage of a specific carbon-hydrogen bond adjacent to the aromatic ring. This activation generates a reactive organopalladium species that subsequently undergoes insertion reactions with the azodicarboxylate and carboxylic acid components. The choice of ligand and the specific oxidation state of the palladium center are critical in ensuring high regioselectivity, preventing the formation of unwanted isomers that could complicate downstream purification. Understanding this catalytic cycle is essential for reducing lead time for high-purity pharmaceutical intermediates, as it allows process chemists to fine-tune reaction parameters to maximize conversion and minimize impurity profiles.
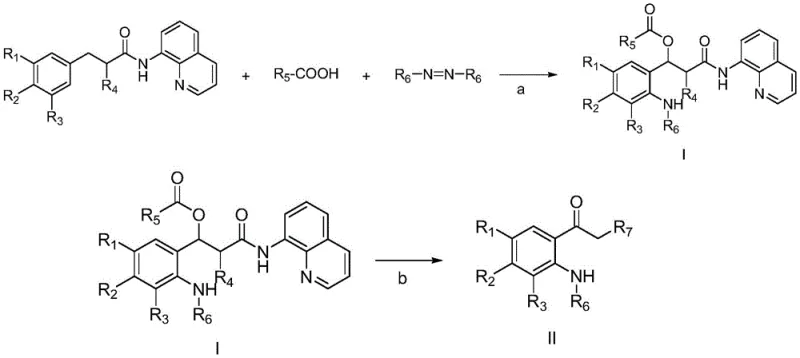
Following the formation of Intermediate I, the pathway diverges to yield distinct heterocyclic architectures based on the specific reagents and conditions applied in the second stage. For the synthesis of Type A heterocycles, such as 2-oxo-1,2,3,4-tetrahydroquinolines, the intermediate is treated with boron trifluoride diethyl etherate followed by thermal cyclization in tetrahydrofuran. Alternatively, Type B compounds, like 4-oxo-1,4-dihydroquinolines, are accessed via a sequence involving protection, base-mediated elimination, and subsequent cyclization using ferrous chloride and polymethylhydrosiloxane. This modularity ensures that a single batch of Intermediate I can potentially supply multiple product lines, drastically improving inventory management and supply chain resilience for manufacturers. The ability to control the outcome through simple reagent switches rather than complete process redesigns is a hallmark of robust industrial chemistry.
How to Synthesize Nitrogen-Containing Heterocycle Intermediates Efficiently
The practical execution of this synthesis involves a carefully controlled sequence of mixing, heating, and purification steps designed to ensure reproducibility and safety on a commercial scale. The process begins with the combination of the amide substrate, the carboxylic acid, and the azodicarboxylate in a suitable solvent such as toluene or 2-methyl-2-butanol, followed by the addition of the palladium catalyst. Detailed standard operating procedures regarding stoichiometry, temperature ramping, and workup protocols are critical for achieving the reported yields and purity levels. For a comprehensive guide on the specific molar ratios, reaction times, and purification techniques required to implement this technology in your facility, please refer to the standardized synthesis steps outlined below.
- Combine amide substrate, carboxylic acid, and azodicarboxylate with a palladium catalyst (e.g., palladium pivalate) in a solvent like toluene.
- Heat the reaction mixture to 110°C for approximately 4 hours to facilitate the C-H bond nitrogen-oxygen double functionalization, yielding Intermediate Compound I.
- Purify Intermediate I and subject it to specific downstream cyclization conditions (e.g., Lewis acid or base treatment) to generate target heterocycles A, B, or C.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this C-H activation technology offers substantial strategic benefits that extend beyond mere technical novelty. By consolidating the synthesis of multiple heterocyclic variants onto a single intermediate platform, organizations can significantly streamline their raw material sourcing strategies, reducing the complexity of managing diverse supplier relationships for specialized precursors. This consolidation directly translates to enhanced negotiating power and reduced administrative overhead, allowing procurement teams to focus on securing bulk quantities of the common starting materials like amides and azodicarboxylates. Furthermore, the robustness of the reaction conditions, which tolerate a wide range of substituents, ensures consistent output quality even when facing minor variations in feedstock quality, thereby mitigating supply chain risks associated with raw material volatility.
- Cost Reduction in Manufacturing: The elimination of pre-functionalization steps and the reduction in the total number of synthetic operations inherently lower the manufacturing costs associated with labor, energy, and solvent consumption. By avoiding the use of expensive and specialized reagents often required for traditional heterocycle construction, this process achieves significant cost savings through simplified material bills. Additionally, the high atom economy of the direct C-H functionalization minimizes waste generation, which reduces the financial burden associated with waste disposal and environmental compliance, contributing to a leaner and more profitable production model.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable starting materials such as carboxylic acids and azodicarboxylates ensures a steady and reliable supply of inputs, minimizing the risk of production stoppages due to material shortages. The versatility of the intermediate allows for flexible production scheduling, where a single stock of Intermediate I can be allocated to meet fluctuating demands for different downstream heterocycles, thus optimizing inventory turnover rates. This flexibility is crucial for maintaining continuity of supply in the face of market dynamics, ensuring that customers receive their orders on time without the need for excessive safety stock.
- Scalability and Environmental Compliance: The reaction conditions employed, specifically the use of standard solvents like toluene and moderate temperatures around 110°C, are fully compatible with existing industrial infrastructure, facilitating seamless scale-up from pilot plant to commercial production volumes. The process avoids the use of highly toxic or hazardous reagents that would require specialized containment systems, simplifying the regulatory approval process for new manufacturing sites. Moreover, the improved efficiency and reduced waste profile align with global sustainability goals, enhancing the corporate social responsibility profile of the manufacturing operation and ensuring long-term viability in an increasingly regulated chemical landscape.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis method, providing clarity on its operational feasibility and strategic value. These insights are derived directly from the experimental data and technical disclosures within the patent, offering a realistic assessment of what partners can expect when integrating this technology into their supply chains. Understanding these nuances is vital for making informed decisions about process adoption and resource allocation.
Q: What is the primary advantage of this C-H activation method over traditional heterocycle synthesis?
A: Unlike traditional methods that are often limited to synthesizing a single type of heterocyclic ring, this patent describes a universal platform. It allows for the generation of structural diversity (Compounds A, B, and C) from a common intermediate (Compound I) through simple variations in downstream processing, significantly reducing the need for multiple distinct synthetic routes.
Q: What are the typical reaction conditions for the key bifunctionalization step?
A: The core transformation utilizes mild to moderate thermal conditions, typically heating the reaction mixture to between 100°C and 120°C, with an optimal temperature of 110°C. The reaction proceeds efficiently in common organic solvents such as toluene or tetrahydrofuran over a period of 3 to 5 hours.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process demonstrates high scalability potential. It employs commercially available reagents like azodicarboxylates and carboxylic acids, avoids exotic catalysts beyond standard palladium salts, and operates at temperatures compatible with standard industrial reactor setups, ensuring robust supply chain continuity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nitrogen-Containing Heterocycle Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced C-H activation technologies in reshaping the landscape of fine chemical manufacturing. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in practical, large-volume applications. Our state-of-the-art facilities are equipped with rigorous QC labs and stringent purity specifications, guaranteeing that every batch of nitrogen-containing heterocycle intermediate meets the exacting standards required by the global pharmaceutical industry. We are committed to delivering not just chemicals, but reliable solutions that drive your drug development programs forward.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic advantages specific to your target molecules. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our capability to be your trusted partner in the efficient and sustainable production of high-value chemical intermediates.
