Advanced One-Pot Synthesis of N-Aryl Naphthofuranone Imines for High-Performance Dye Manufacturing
The landscape of organic dye intermediate manufacturing is undergoing a significant transformation driven by the urgent need for greener, more efficient synthetic pathways. Patent CN108947948B introduces a groundbreaking methodology for the synthesis of N-aryl naphthofuranone imine compounds, a critical structural motif found in high-performance dyes and bioactive materials. Unlike traditional multi-step processes that rely on hazardous reagents, this invention leverages a direct one-pot oxidative cyclization strategy. By utilizing molecular oxygen as the terminal oxidant and avoiding toxic solvents like benzene, this technology offers a compelling solution for modern chemical production. For R&D directors and procurement specialists alike, this represents a pivotal shift towards sustainable chemistry that does not compromise on yield or purity, establishing a new benchmark for the reliable dye intermediate supplier market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzo- and naphthofuranone derivatives has been plagued by severe operational and environmental drawbacks. Early methodologies, such as those described by Hafez and Taghrid, necessitated the use of benzene, a known carcinogen, as the primary reaction solvent, posing significant health risks and regulatory hurdles for large-scale operations. Furthermore, alternative routes proposed by researchers like Feiler required the use of oxalyl chloride and aluminum trichloride, demanding strictly anhydrous conditions and generating substantial amounts of corrosive hydrogen chloride gas. These legacy processes often involved multi-step sequences with intermediate isolation, leading to accumulated material losses and complex waste streams. The reliance on glacial acetic acid for dehydration steps further complicated the purification process, requiring additional neutralization and washing stages that inflated production costs and extended lead times for high-purity dye intermediates.
The Novel Approach
In stark contrast, the method disclosed in CN108947948B streamlines the entire synthetic sequence into a single, efficient operation. This innovative route combines aromatic amines, 2-naphthol derivatives, and glyoxylates in the presence of tert-valeric acid and molecular sieves under an oxygen atmosphere. The elimination of oxalyl chloride removes the need for rigorous moisture control and hazardous gas scrubbing systems. Moreover, replacing benzene with safer alternatives like toluene or chlorobenzene significantly enhances workplace safety and environmental compliance. The reaction proceeds smoothly at moderate temperatures between 100-120°C, demonstrating remarkable functional group tolerance that allows for the incorporation of diverse substituents without protecting group strategies. This simplification not only accelerates the development timeline but also drastically reduces the overall cost reduction in organic dye manufacturing by minimizing unit operations and waste treatment requirements.
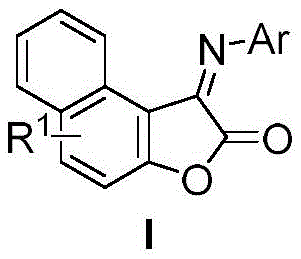
Mechanistic Insights into Oxygen-Mediated Oxidative Cyclization
The core of this technological advancement lies in its elegant mechanistic pathway, which facilitates the construction of the fused furanone-imine skeleton through a concerted oxidative process. The reaction initiates with the condensation of the aromatic amine and glyoxylate to form an imine intermediate, which subsequently undergoes electrophilic attack on the electron-rich 2-naphthol ring. Molecular oxygen serves as the critical oxidant, regenerating the catalytic species and driving the aromatization of the furanone ring system. This mechanism avoids the formation of unstable acyl chloride intermediates typical of Friedel-Crafts approaches, thereby reducing the potential for side reactions such as polymerization or over-oxidation. The presence of tert-valeric acid likely acts as a proton shuttle or weak acid catalyst, optimizing the pH environment for cyclization while maintaining the stability of the sensitive imine linkage. Understanding this mechanism is vital for process chemists aiming to optimize reaction parameters for commercial scale-up of complex dye intermediates.
From an impurity control perspective, this oxidative protocol offers superior selectivity compared to traditional acid-catalyzed dehydrations. The mild reaction conditions prevent the degradation of sensitive functional groups such as esters, halides, and trifluoromethyl moieties, which are often prone to hydrolysis or displacement under harsh Lewis acidic conditions. The use of molecular sieves effectively sequesters water produced during the condensation steps, shifting the equilibrium towards product formation without the need for azeotropic distillation. This results in a cleaner crude reaction profile, simplifying downstream purification and ensuring that the final high-purity OLED material or dye precursor meets stringent specifications. The robustness of this catalytic system against various electronic environments on the aryl ring ensures consistent quality across a broad library of analogues, a key requirement for pharmaceutical and agrochemical applications.
How to Synthesize N-Aryl Naphthofuranone Imine Efficiently
The practical implementation of this synthesis involves a straightforward procedure accessible to standard laboratory and pilot plant facilities. Operators simply charge the reactor with the requisite amine, naphthol, and glyoxylate components along with the acid promoter and desiccant. The system is then pressurized with oxygen or operated under an oxygen balloon, heated to the specified range, and monitored until conversion is complete. Workup involves basic aqueous washes to remove acidic residues followed by organic extraction, yielding the target compound in high purity after chromatographic separation.
- Mix aromatic amine, 2-naphthol, glyoxylic ester, tert-valeric acid, molecular sieves, and organic solvent in an oxygen-filled vessel.
- Heat the reaction mixture to 100-120°C and stir for 12-24 hours to facilitate oxidative cyclization.
- Cool, wash with water/brine, extract with dichloromethane, dry, remove solvent, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology translates into tangible strategic benefits beyond mere technical novelty. The shift away from controlled substances like benzene and highly reactive reagents like oxalyl chloride simplifies logistics and storage requirements, reducing the regulatory burden associated with hazardous material handling. The one-pot nature of the reaction significantly shortens the manufacturing cycle time, allowing for faster response to market demands and reduced inventory holding costs. Furthermore, the use of commodity chemicals such as toluene and oxygen ensures a stable and resilient supply chain, mitigating the risks associated with specialty reagent shortages. These factors collectively contribute to a more robust and cost-effective sourcing strategy for critical dye and pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The elimination of expensive acylating agents and the reduction of reaction steps directly lower the bill of materials. By avoiding the need for strict anhydrous conditions and specialized corrosion-resistant equipment required for hydrogen chloride management, capital expenditure and maintenance costs are substantially decreased. The simplified workup procedure reduces solvent consumption and energy usage during distillation, leading to significant operational savings. Additionally, the high yields reported across various substrates minimize raw material waste, further enhancing the overall economic efficiency of the production process.
- Enhanced Supply Chain Reliability: Utilizing widely available starting materials such as substituted anilines and naphthols ensures consistent availability and price stability. The process tolerance to moisture and air (post-oxygenation) reduces the risk of batch failures due to environmental excursions, guaranteeing on-time delivery performance. The ability to synthesize a wide range of derivatives using a unified platform allows suppliers to offer a diversified portfolio without maintaining separate production lines for each analogue. This flexibility is crucial for meeting the dynamic needs of downstream customers in the textile and electronics sectors.
- Scalability and Environmental Compliance: The absence of toxic benzene and the use of oxygen as a green oxidant align perfectly with modern environmental, health, and safety (EHS) standards. This facilitates easier permitting for new production facilities and reduces the cost of waste disposal and emissions control. The reaction's exothermic profile is manageable, allowing for safe scale-up from kilogram to tonne quantities without complex engineering controls. Consequently, manufacturers can expand capacity rapidly to meet growing global demand while maintaining a strong sustainability profile.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and scope defined in the patent literature, providing clarity for potential partners and licensees.
Q: What are the primary advantages of this new synthesis method over conventional routes?
A: The novel method eliminates the need for carcinogenic benzene solvents and hazardous oxalyl chloride reagents. It utilizes molecular oxygen as a green oxidant and operates under simpler conditions with broader functional group tolerance.
Q: Which substrates are compatible with this oxidative cyclization protocol?
A: The process demonstrates exceptional compatibility with various substituents including alkyl, alkoxy, halogens, nitro, trifluoromethyl, ester, and heterocyclic groups on both the amine and naphthol components.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the one-pot nature, avoidance of strict anhydrous conditions, and use of common solvents like toluene make it highly scalable and economically viable for industrial dye intermediate manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Aryl Naphthofuranone Imine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this green synthesis route for the next generation of functional dyes and electronic materials. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless. Our state-of-the-art facilities are equipped to handle oxidative chemistries safely and efficiently, adhering to stringent purity specifications and rigorous QC labs to guarantee product consistency. We are committed to delivering high-quality intermediates that empower your innovation pipeline while respecting environmental boundaries.
We invite you to collaborate with us to leverage this advanced technology for your specific application needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and quality targets. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how we can become your trusted partner in sustainable chemical manufacturing.
