Scalable Synthesis of Bepidoic Acid: A High-Purity Organozinc Route for Commercial API Production
Scalable Synthesis of Bepidoic Acid: A High-Purity Organozinc Route for Commercial API Production
The pharmaceutical industry is constantly seeking robust synthetic pathways for novel therapeutic agents, particularly for metabolic disorders where ATP-citrate lyase inhibitors like Bepidoic acid show immense promise. A recent breakthrough detailed in patent CN115108904A introduces a transformative synthesis method that addresses long-standing challenges in yield and purity associated with this complex molecule. This innovative approach leverages the unique chemoselectivity of organozinc reagents to construct the carbon backbone, bypassing the limitations of traditional Grignard chemistry. By shifting the paradigm from highly reactive magnesium-based nucleophiles to milder zinc-based counterparts, the process achieves a remarkable purity of 99.93% and a yield of 95%, setting a new benchmark for the commercial production of high-purity pharmaceutical intermediates. This technical advancement not only streamlines the manufacturing workflow but also ensures that the final active pharmaceutical ingredient meets the rigorous quality standards required for global regulatory approval.
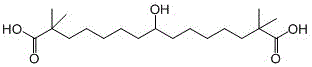
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies, such as those disclosed in patent CN111825546A, rely heavily on the formation of Grignard reagents to establish key carbon-carbon bonds. While Grignard chemistry is a staple in organic synthesis, it presents significant drawbacks when applied to substrates containing sensitive functional groups like esters or nitriles. In the conventional route, the Grignard reagent inevitably attacks the ester moieties present within the intermediate molecules, leading to a cascade of unwanted side reactions and the formation of complex impurity profiles. This lack of chemoselectivity results in a suboptimal total yield of approximately 74% and a product purity that struggles to reach 99.0%, which is often insufficient for direct use as a pharmaceutical raw material without extensive and costly purification steps. Furthermore, the harsh conditions required for Grignard reactions necessitate stringent safety protocols and specialized equipment, complicating the scale-up process for industrial manufacturing.
The Novel Approach
In stark contrast, the novel methodology outlined in CN115108904A utilizes an organozinc reagent (Compound 1) coupled with a specifically designed aldehyde (Compound 2) to construct the target framework. Organozinc species are renowned for their functional group tolerance, allowing them to react selectively with aldehydes in the presence of esters without triggering nucleophilic acyl substitution. This fundamental shift in reactivity eliminates the major source of byproducts seen in the Grignard route, thereby drastically simplifying the downstream purification process. The reaction proceeds smoothly in aprotic solvents like tetrahydrofuran or dichloromethane, generating the intermediate ester (Compound 3) with high fidelity. Subsequent hydrolysis under alkaline conditions followed by acidification yields the final Bepidoic acid with exceptional efficiency. This route not only enhances the economic viability of the process but also aligns perfectly with the principles of green chemistry by reducing waste and energy consumption.
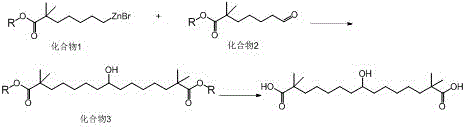
Mechanistic Insights into Organozinc-Mediated Addition
The core of this technological leap lies in the distinct electronic properties of the carbon-zinc bond compared to the carbon-magnesium bond. In the mechanistic cycle, the organozinc reagent acts as a soft nucleophile that preferentially attacks the electrophilic carbonyl carbon of the aldehyde component. Unlike the hard, highly basic Grignard reagents, organozinc compounds do not aggressively deprotonate alpha-hydrogens or attack the carbonyl carbons of the distal ester groups within the same molecule. This chemoselectivity is crucial for maintaining the integrity of the ester functionalities that are essential for the final hydrolysis step. The reaction likely proceeds through a six-membered transition state where the zinc atom coordinates with the aldehyde oxygen, facilitating the transfer of the alkyl group. This controlled addition ensures that the stereochemistry and structural fidelity of the growing chain are preserved, minimizing the formation of diastereomers or structural isomers that would otherwise complicate the isolation of the pure drug substance.
Furthermore, the preparation of the organozinc reagent itself is optimized to ensure stability and reactivity. The process involves the insertion of activated zinc powder into a bromo-ester precursor, a transformation catalyzed by small amounts of iodine or cuprous iodide. This activation step is critical for overcoming the passivation layer on the zinc surface, ensuring rapid and complete formation of the organometallic species. The use of inert solvents such as N,N-dimethylformamide or ethers provides a stable medium that prevents premature decomposition of the sensitive organozinc intermediate. By carefully controlling the stoichiometry, typically maintaining a molar ratio of the organozinc reagent to the aldehyde between 1:1.0 and 1:1.5, the process maximizes the conversion of the limiting reagent while minimizing the presence of unreacted starting materials in the final crude mixture. This precise control over reaction parameters is what enables the achievement of the reported 99.93% purity.
How to Synthesize Bepidoic Acid Efficiently
The synthesis of Bepidoic acid via this organozinc pathway involves a sequence of well-defined transformations that can be readily adapted for pilot and commercial scale operations. The process begins with the preparation of the two key building blocks: the organozinc nucleophile and the aldehyde electrophile. Both precursors are derived from readily available starting materials such as alkyl isobutyrate and brominated alkanes or alkenes, ensuring a secure supply chain for raw materials. The coupling reaction is the pivotal step, requiring careful temperature management to balance reaction rate and selectivity. Following the coupling, the intermediate ester undergoes saponification, a standard unit operation in fine chemical manufacturing, to reveal the free carboxylic acid groups. For a detailed breakdown of the specific operational parameters, reagent grades, and workup procedures, please refer to the standardized synthesis guide below.
- Preparation of Organozinc Reagent: React alkyl isobutyrate with 1,5-dibromopentane to form a bromo-ester intermediate, then treat with zinc powder and a catalyst like iodine or cuprous iodide in an inert solvent.
- Preparation of Aldehyde Component: Condense alkyl isobutyrate with 1-bromopentene to form a vinyl ester, followed by ozonolysis and reduction to generate the corresponding aldehyde.
- Coupling and Hydrolysis: Perform the addition reaction between the organozinc reagent and the aldehyde in an aprotic solvent to form the ester intermediate, followed by alkaline hydrolysis and acidification to yield Bepidoic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers tangible benefits that extend beyond mere technical superiority. The primary advantage lies in the substantial improvement in process efficiency, which directly translates to cost reduction in pharmaceutical intermediate manufacturing. By eliminating the formation of difficult-to-remove impurities associated with Grignard side reactions, the new method significantly reduces the burden on purification units such as chromatography columns or crystallization tanks. This reduction in processing steps lowers the consumption of solvents, adsorbents, and energy, resulting in a leaner and more cost-effective production cycle. Additionally, the higher overall yield means that less raw material is required to produce the same amount of final API, optimizing the utilization of expensive starting reagents and maximizing the return on investment for every batch produced.
- Cost Reduction in Manufacturing: The transition to an organozinc-based strategy fundamentally alters the cost structure of Bepidoic acid production. Because the reaction is highly chemoselective, there is no need for complex protection-deprotection strategies or extensive scavenging of byproducts, which are common cost drivers in traditional routes. The simplified workflow allows for shorter cycle times and higher throughput in existing reactor trains. Moreover, the use of zinc, a relatively abundant and inexpensive metal compared to some specialized catalysts used in alternative cross-coupling methods, keeps the bill of materials low. The qualitative improvement in crude purity means that recrystallization becomes a highly effective purification tool, avoiding the need for expensive preparative HPLC or multiple distillation steps that erode profit margins.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of commodity chemicals as starting materials. Alkyl isobutyrate, dibromopentane, and bromopentene are widely available from multiple global suppliers, reducing the risk of single-source bottlenecks. The robustness of the organozinc reaction conditions also implies a wider operating window, making the process less susceptible to minor fluctuations in utility supplies or environmental conditions that might stall more sensitive reactions. This reliability ensures consistent delivery schedules for downstream formulation partners. Furthermore, the scalability of the reaction has been demonstrated to be straightforward, allowing manufacturers to ramp up production volumes rapidly in response to market demand without the need for bespoke equipment or hazardous high-pressure setups.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route offers a cleaner profile. The reduction in byproduct formation inherently lowers the volume of chemical waste generated per kilogram of product, simplifying waste treatment and disposal compliance. The avoidance of highly pyrophoric reagents often associated with large-scale Grignard preparations enhances plant safety, potentially lowering insurance premiums and reducing the need for specialized containment infrastructure. The process aligns with modern sustainability goals by improving atom economy and reducing the E-factor (mass of waste per mass of product). These factors make the technology attractive not just for its economic returns but also for its alignment with corporate social responsibility targets regarding green manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, offering a clear picture of the operational realities. Understanding these nuances is essential for R&D teams evaluating technology transfer and for procurement officers assessing supplier capabilities. The answers reflect the consensus on best practices for handling organozinc chemistry and managing the specific purification requirements of Bepidoic acid to ensure it meets pharmacopeial standards.
Q: Why is the organozinc route superior to the Grignard method for Bepidoic acid synthesis?
A: The organozinc route avoids the compatibility issues found in Grignard reactions where the reagent attacks ester or cyano groups present in the substrate. This chemoselectivity significantly reduces byproducts, leading to higher yields (up to 95%) and purity (99.93%) compared to the 74% yield and 99.0% purity of conventional methods.
Q: What are the critical reaction conditions for the organozinc coupling step?
A: The coupling reaction between the organozinc reagent and the aldehyde should be conducted in an aprotic solvent such as tetrahydrofuran or dichloromethane at low temperatures, typically below 5°C, to control exothermicity and ensure high selectivity before warming to complete the reaction.
Q: How does this synthesis method impact industrial scalability?
A: By eliminating the need for strict temperature controls required to suppress Grignard side reactions and simplifying the purification process due to fewer impurities, this method offers a more robust pathway for commercial scale-up, reducing overall processing time and waste generation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bepidoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of novel therapeutics depends on the reliability and quality of the supply chain. Our team of expert process chemists has thoroughly evaluated the organozinc route described in CN115108904A and confirmed its potential for large-scale manufacturing. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to pilot plant is seamless. Our facilities are equipped with state-of-the-art reactors capable of handling moisture-sensitive organometallic reactions under strictly controlled inert atmospheres. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Bepidoic acid we deliver meets the highest international standards for API intermediates.
We invite you to collaborate with us to optimize your supply chain for this critical metabolic inhibitor. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. By partnering with us, you gain access to our deep reservoir of chemical expertise and our commitment to continuous process improvement. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a stable, high-quality supply of Bepidoic acid that supports your clinical and commercial goals without compromise.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
