Advanced Metal-Free Synthesis of 5-Trifluoromethyl-4H-Imidazoline-4-Ketone Derivatives for Pharma
Advanced Metal-Free Synthesis of 5-Trifluoromethyl-4H-Imidazoline-4-Ketone Derivatives for Pharma
In the rapidly evolving landscape of pharmaceutical and agrochemical development, the efficient incorporation of fluorine atoms into heterocyclic scaffolds remains a critical challenge for process chemists. Patent CN108976170B introduces a groundbreaking methodology for the synthesis of 5-trifluoromethyl-4H-imidazoline-4-ketone derivatives, addressing the urgent need for robust, scalable routes to these valuable building blocks. These fluorinated heterocycles are increasingly recognized for their ability to enhance metabolic stability and bioavailability in drug candidates, yet traditional synthetic pathways have often been plagued by harsh conditions or reliance on scarce resources. The disclosed invention provides a streamlined approach that utilizes readily available amidine salts and trifluoromethyl ketones, reacting them under basic conditions to form the target imidazoline core with exceptional efficiency. This technological leap not only fills a significant gap in the current literature regarding fluorinated imidazolines but also offers a practical solution for the commercial scale-up of complex fluorinated heterocycles required in modern medicinal chemistry programs.
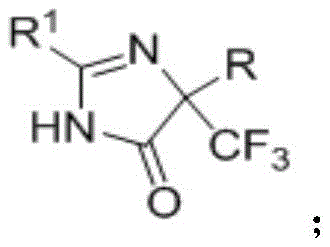
The structural versatility of the compounds produced via this method is evident in the general Formula I, where diverse substituents can be introduced at multiple positions to fine-tune physicochemical properties. By enabling the systematic variation of R1 and R groups, this synthesis empowers research teams to rapidly generate libraries of analogues for structure-activity relationship (SAR) studies. As a reliable pharmaceutical intermediate supplier, understanding the nuances of such flexible synthetic platforms is essential for meeting the dynamic demands of global R&D pipelines. The ability to access these scaffolds without proprietary bottlenecks ensures a steady supply chain for downstream API manufacturing, positioning this technology as a cornerstone for future drug discovery initiatives targeting resistant pathogens or metabolic disorders.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of fluorinated imidazoline rings has relied heavily on multistep sequences that often necessitate the use of expensive transition metal catalysts, such as palladium or copper complexes, to facilitate cyclization or cross-coupling events. These conventional protocols frequently suffer from significant drawbacks, including the requirement for stringent inert atmospheres, high energy consumption due to elevated temperatures, and the generation of toxic heavy metal waste that complicates environmental compliance. Furthermore, the sensitivity of fluorine-containing intermediates to harsh acidic or basic conditions in traditional routes often leads to defluorination side reactions, resulting in poor yields and difficult-to-remove impurities that compromise the purity profile of the final active ingredient. For procurement managers, these inefficiencies translate into inflated raw material costs and extended lead times, as specialized reagents and rigorous purification steps become unavoidable necessities in the production workflow.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in CN108976170B leverages a direct condensation strategy that eliminates the need for transition metal catalysis entirely, relying instead on simple, cost-effective bases like potassium tert-butoxide or potassium carbonate. This metal-free paradigm shift allows the reaction to proceed smoothly under ambient air or oxygen atmospheres at moderate temperatures ranging from 50°C to 90°C, drastically reducing the operational complexity and energy footprint of the manufacturing process. The reaction exhibits remarkable tolerance to a wide array of functional groups, including electron-withdrawing nitro and cyano groups as well as electron-donating methoxy and amino moieties, ensuring that diverse chemical spaces can be explored without protecting group manipulations. By simplifying the reaction setup and workup procedures, this method facilitates cost reduction in API manufacturing while simultaneously enhancing the safety profile of the operation by avoiding pyrophoric reagents or high-pressure equipment.

Mechanistic Insights into Base-Catalyzed Cyclization
The mechanistic pathway of this transformation involves the nucleophilic attack of the amidine nitrogen on the electrophilic carbonyl carbon of the trifluoromethyl ketone, initiated by the deprotonation of the amidine salt by the base. This initial addition forms a tetrahedral intermediate which subsequently undergoes intramolecular cyclization, driven by the elimination of water or alcohol depending on the specific leaving group dynamics of the salt form. The presence of the strongly electron-withdrawing trifluoromethyl group adjacent to the carbonyl enhances the electrophilicity of the ketone, thereby accelerating the rate of nucleophilic attack and allowing the reaction to proceed efficiently even with sterically hindered substrates. Crucially, the choice of base plays a pivotal role in regulating the reaction kinetics; stronger bases like potassium tert-butoxide generally afford higher yields compared to weaker carbonates, suggesting that the complete generation of the free amidine species is a key determinant of success in this catalytic cycle.
From an impurity control perspective, the absence of transition metals inherently removes the risk of metal leaching, a common regulatory hurdle in pharmaceutical production that often requires additional scavenging steps to meet strict ppm limits. The high chemoselectivity of this base-mediated process minimizes the formation of oligomeric byproducts or over-alkylated species, which are frequently observed in metal-catalyzed variants involving radical intermediates. Detailed analysis of the reaction mixtures indicates that the primary diastereoisomer is formed with high selectivity, likely due to thermodynamic control during the ring-closing step, which simplifies the downstream purification burden. For quality assurance teams, this inherent purity profile means that standard crystallization or chromatographic techniques are sufficient to achieve high-purity pharmaceutical intermediates suitable for subsequent coupling reactions, thereby streamlining the overall quality control workflow and reducing analytical testing overhead.
How to Synthesize 5-Trifluoromethyl-4H-Imidazoline-4-Ketone Efficiently
To implement this synthesis in a laboratory or pilot plant setting, operators should follow a standardized protocol that emphasizes precise stoichiometric control and temperature management to maximize yield and reproducibility. The process begins with the dissolution of the amidine hydrochloride salt and the trifluoromethyl ketone in a polar aprotic solvent such as N,N-dimethylformamide (DMF) or 1,4-dioxane, ensuring complete solubility before the addition of the base. Upon adding the base, typically in a molar ratio of 1:1 to 4:1 relative to the amidine, the reaction mixture is heated to between 50°C and 90°C and stirred for a period of 12 to 24 hours, during which time the progress can be monitored via TLC or HPLC to determine the optimal endpoint. Following the reaction, the mixture is cooled to room temperature, quenched with water, and extracted with an organic solvent like ethyl acetate, after which the crude product is isolated via rotary evaporation and purified using column chromatography with a petroleum ether and ethyl acetate gradient.
- Combine amidine hydrochloride salt and trifluoromethyl ketone compound in an organic solvent such as DMF or dioxane.
- Add a base such as potassium tert-butoxide or potassium carbonate and stir the mixture at 50-90°C for 12-24 hours under air.
- Cool the reaction, extract with ethyl acetate, remove solvent via rotary evaporation, and purify the crude product using column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement specialists, the adoption of this synthetic route offers tangible strategic benefits that extend beyond mere chemical elegance, directly impacting the bottom line through operational efficiencies. The elimination of precious metal catalysts removes a significant variable cost component and mitigates supply chain risks associated with the volatility of metal prices and geopolitical sourcing constraints. Furthermore, the use of commodity chemicals such as potassium carbonate and common organic solvents ensures that raw material availability remains stable, preventing production delays caused by specialty reagent shortages. This robustness in the supply chain is critical for maintaining continuous manufacturing operations, especially when scaling from clinical trial materials to commercial volumes where consistency is paramount.
- Cost Reduction in Manufacturing: The economic advantage of this method is primarily derived from the removal of expensive ligand-catalyst systems and the associated purification resins required to strip metal residues from the final product. By utilizing inexpensive inorganic bases and operating under air rather than inert gas, the process significantly lowers utility costs and capital expenditure on specialized reactor hardware. Additionally, the high yields reported across various substrates reduce the amount of starting material wasted per batch, improving the overall atom economy and lowering the cost of goods sold (COGS) for the final intermediate. These cumulative savings allow for more competitive pricing strategies in the global market while preserving healthy margins for manufacturers.
- Enhanced Supply Chain Reliability: The reliance on broadly available starting materials, such as substituted benzamidines and trifluoroacetophenones, ensures that the supply chain is not vulnerable to single-source bottlenecks. Since these precursors are produced by multiple vendors globally, procurement teams can easily qualify alternative suppliers to mitigate risk, ensuring uninterrupted production schedules. The simplicity of the reaction workup, which involves standard extraction and evaporation techniques, also means that the process can be transferred easily between different manufacturing sites without requiring extensive re-validation or specialized training for operators, further enhancing supply continuity.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, the absence of heavy metals simplifies waste stream management, reducing the cost and complexity of effluent treatment and disposal. The reaction conditions are mild enough to be safely scaled from gram-scale laboratory experiments to multi-kilogram pilot runs without encountering exothermic runaway risks, facilitating a smoother technology transfer process. This scalability, combined with the reduced environmental footprint, aligns perfectly with the increasing regulatory pressure on pharmaceutical companies to adopt greener chemistry practices, making this route highly attractive for long-term sustainable manufacturing partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, providing clarity for stakeholders evaluating its potential integration into their existing workflows. These answers are derived directly from the experimental data and scope defined in the patent documentation, ensuring accuracy and relevance for decision-makers. Understanding these details is crucial for assessing the feasibility of adopting this method for specific project requirements and for anticipating any potential challenges during scale-up.
Q: Does this synthesis method require expensive transition metal catalysts?
A: No, the patented method (CN108976170B) specifically avoids the use of transition metal catalysts, utilizing inexpensive inorganic or organic bases instead, which significantly simplifies downstream purification.
Q: What is the substrate scope for this imidazoline ketone synthesis?
A: The method demonstrates excellent functional group tolerance, accommodating various substituents on both the amidine salt and the ketone, including halogens, methoxy groups, nitro groups, and heterocycles like pyridine.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the reaction operates under mild conditions (50-90°C) and air atmosphere without sensitive catalysts, making it highly amenable to scale-up from gram to multi-kilogram production levels.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-4H-Imidazoline-4-Ketone Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free synthesis technology in accelerating the development of next-generation fluorinated therapeutics. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 5-trifluoromethyl-4H-imidazoline-4-ketone derivative delivered meets the highest international standards for pharmaceutical intermediates. We are committed to leveraging our technical expertise to optimize this route further, tailoring the process to your specific volume and quality needs.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis can drive value in your upcoming projects. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this metal-free protocol for your specific target molecules. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our capability as your trusted partner in delivering high-quality, cost-effective chemical solutions for the global healthcare industry.
