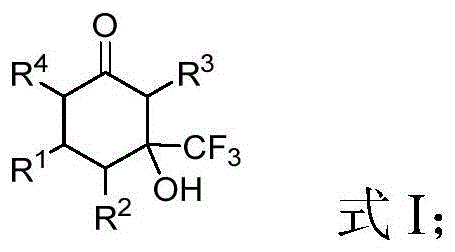
Scalable Copper-Catalyzed Synthesis of Beta-Trifluoromethyl Cyclohexanone Derivatives for Pharma
The pharmaceutical and agrochemical industries are increasingly reliant on fluorinated scaffolds to enhance the metabolic stability and bioactivity of active ingredients. Patent CN109608323B introduces a significant advancement in this domain by disclosing a robust synthesis method for beta-trifluoromethyl-beta-hydroxy substituted cyclohexanone derivatives. These compounds serve as critical building blocks for constructing complex molecular architectures found in modern therapeutics. The disclosed technology addresses the long-standing challenge of efficiently introducing trifluoromethyl groups into cyclic ketone systems without resorting to harsh conditions or expensive noble metal catalysts. By leveraging a cost-effective copper catalytic system, this innovation provides a streamlined pathway for generating high-value intermediates that were previously difficult to access with high purity and yield.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this invention, the construction of beta-trifluoromethyl-beta-hydroxy substituted cyclohexanone derivatives was fraught with significant technical hurdles that hindered widespread adoption in process chemistry. Existing literature, such as the work by Wang et al. in Organic Letters (2007), described cyclization reactions that were severely limited in scope, often restricted to acetone participation which yielded only a single specific compound. Furthermore, alternative approaches involving the cyclization of trifluoromethyl-modified 1,3-dicarbonyl compounds with alpha,beta-unsaturated ketones, as reported in the Russian Chemical Bulletin (1997), consistently delivered unsatisfactory results with isolated yields remaining stubbornly below 30 percent. These low efficiencies not only drove up material costs but also generated excessive chemical waste, creating substantial bottlenecks for supply chain managers aiming to secure reliable quantities of these key intermediates for drug development pipelines.
The Novel Approach
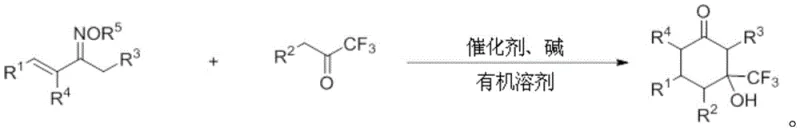
In stark contrast to these legacy methods, the novel approach detailed in CN109608323B utilizes a ligand-free copper catalytic system that dramatically improves both efficiency and versatility. The reaction proceeds by coupling alpha,beta-unsaturated oxime ester compounds with trifluoromethyl ketone compounds under mild thermal conditions, typically between 50°C and 90°C. This methodology eliminates the need for expensive phosphine ligands often required in palladium or rhodium catalysis, thereby simplifying the purification process and reducing the overall cost of goods sold. The protocol demonstrates exceptional functional group tolerance, successfully accommodating substrates with electron-withdrawing and electron-donating groups alike, which is crucial for medicinal chemists exploring structure-activity relationships. Moreover, the reaction has been proven scalable to gram-level production with high yields, signaling a clear path toward kilogram and tonne-scale manufacturing for commercial applications.
Mechanistic Insights into Copper-Catalyzed Cyclization
The core of this technological breakthrough lies in the efficient activation of the carbon-carbon double bond and the subsequent intramolecular cyclization facilitated by the copper species. In this ligand-free environment, the copper catalyst likely coordinates with the oxime ester moiety, promoting a radical or ionic cascade that triggers the nucleophilic attack on the trifluoromethyl ketone carbonyl or the activated alkene system. This mechanism avoids the formation of stable metal-complex intermediates that often plague transition metal catalysis, allowing for easier catalyst removal during workup. The absence of bulky ligands reduces steric hindrance around the active site, enabling the reaction to proceed with a broader range of sterically demanding substrates. This mechanistic simplicity is a key driver for the observed high yields and operational safety, as it minimizes the risk of exothermic runaway reactions associated with more complex catalytic cycles.
From an impurity control perspective, the specificity of this copper-catalyzed route ensures a cleaner crude reaction profile compared to traditional base-mediated condensations. The mild reaction conditions prevent the decomposition of sensitive functional groups such as esters, nitriles, and halides, which are frequently present in advanced pharmaceutical intermediates. By maintaining a nitrogen atmosphere and utilizing controlled temperatures, the process suppresses side reactions like polymerization or over-oxidation that could compromise the purity of the final beta-trifluoromethyl-beta-hydroxy cyclohexanone product. This high level of chemoselectivity reduces the burden on downstream purification steps, such as column chromatography or recrystallization, ultimately leading to a more sustainable and economically viable manufacturing process for high-purity fine chemicals.
How to Synthesize Beta-Trifluoromethyl Cyclohexanone Efficiently
The synthesis protocol outlined in the patent provides a standardized framework for producing these valuable derivatives with consistent quality. The process begins with the precise stoichiometric mixing of the alpha,beta-unsaturated oxime ester and the trifluoromethyl ketone in a polar aprotic solvent such as acetonitrile or dimethyl sulfoxide. A copper salt, such as cuprous chloride or cupric acetate, is introduced alongside a mild inorganic base like cesium carbonate or potassium carbonate to initiate the transformation. The reaction mixture is then heated under an inert nitrogen atmosphere to ensure optimal conversion while preventing oxidative degradation of the reagents. Following the completion of the reaction, a straightforward aqueous workup and extraction procedure isolates the crude product, which can be further purified to meet stringent pharmaceutical specifications.
- Mix alpha,beta-unsaturated oxime ester and trifluoromethyl ketone with a copper catalyst and base in an organic solvent.
- Heat the reaction mixture under nitrogen atmosphere at 50-90°C for 6-24 hours to facilitate cyclization.
- Cool, extract with organic solvent, remove solvent via rotary evaporation, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this copper-catalyzed synthesis route offers tangible strategic benefits that extend beyond mere technical feasibility. The elimination of expensive ligands and the use of abundant copper salts directly translate into a reduction in raw material costs, making the final intermediate more price-competitive in the global market. Furthermore, the mild reaction conditions reduce energy consumption requirements for heating and cooling, contributing to lower utility costs and a smaller carbon footprint for the manufacturing facility. The robustness of the method across diverse substrates means that a single production line can be adapted to manufacture a wide library of analogues, enhancing asset utilization and flexibility in responding to changing market demands for specialized fluorinated compounds.
- Cost Reduction in Manufacturing: The ligand-free nature of this catalytic system removes a significant cost driver associated with precious metal complexes and specialized organic ligands. By relying on inexpensive copper salts and common inorganic bases, the direct material cost per kilogram of product is substantially lowered. Additionally, the simplified post-treatment process, which avoids complex metal scavenging steps often required for palladium residues, reduces the consumption of purification media and solvents. This streamlining of the workflow leads to significant operational savings and improved overall process economics for large-scale production campaigns.
- Enhanced Supply Chain Reliability: The use of commercially available and stable starting materials ensures a secure supply chain that is less vulnerable to geopolitical disruptions or shortages of exotic reagents. The demonstrated scalability of the reaction from milligram to gram scales in the patent data suggests a low technical risk for scaling up to multi-kilogram or tonne quantities. This reliability allows procurement teams to negotiate longer-term contracts with greater confidence, knowing that the manufacturing process is robust enough to handle increased volume requirements without compromising on delivery timelines or product quality.
- Scalability and Environmental Compliance: The reaction operates under relatively mild thermal conditions and uses standard organic solvents that are easily recovered and recycled, aligning with modern green chemistry principles. The high atom economy and reduced waste generation simplify waste treatment protocols, ensuring compliance with increasingly stringent environmental regulations. The ability to scale this process safely without the need for high-pressure equipment or cryogenic conditions makes it an attractive option for contract development and manufacturing organizations looking to expand their capacity for fluorinated intermediate production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the fit of this technology within their existing R&D and manufacturing frameworks.
Q: What are the advantages of this copper-catalyzed method over prior art?
A: Unlike previous methods which suffered from low yields below 30% and limited substrate scope, this patent utilizes a ligand-free copper catalyst system that achieves significantly higher yields and accommodates a wide range of functional groups.
Q: Is this synthesis method suitable for industrial scale-up?
A: Yes, the patent explicitly demonstrates successful gram-scale production with mild reaction conditions and simple post-treatment procedures, indicating strong potential for commercial scale-up in pharmaceutical manufacturing.
Q: What types of substituents are tolerated in this reaction?
A: The method shows excellent adaptability to various substituents including halogens, alkyl groups, methoxy, cyano, and heterocycles like thiophene and furan, making it versatile for diverse drug discovery programs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Trifluoromethyl Cyclohexanone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality fluorinated intermediates play in the development of next-generation pharmaceuticals and agrochemicals. Our team of expert chemists has extensively evaluated the copper-catalyzed cyclization route described in CN109608323B and confirmed its viability for industrial application. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition smoothly from the laboratory bench to full-scale manufacturing. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of beta-trifluoromethyl-beta-hydroxy cyclohexanone derivative meets the exacting standards required by global regulatory bodies.
We invite you to collaborate with us to optimize your supply chain for these essential building blocks. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. By partnering with us, you gain access to specific COA data and comprehensive route feasibility assessments that will empower you to make informed sourcing decisions. Contact us today to discuss how we can support your R&D initiatives and commercial production goals with reliable, cost-effective, and high-quality chemical solutions.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
