Advanced Copper-Catalyzed Synthesis of 1,1-Difluoro-2-Propylene Hydrocarbon Compounds for Commercial Scale-Up
Introduction to Next-Generation Difluoroalkene Synthesis
The strategic incorporation of fluorine atoms into organic molecules has revolutionized modern medicinal chemistry, particularly in the design of bioisosteres that enhance metabolic stability and membrane permeability. Patent CN113173830A discloses a groundbreaking synthesis method for 1,1-difluoro-2-propylene hydrocarbon compounds, addressing a critical gap in the availability of these valuable structural motifs. Traditionally, accessing difluoromethyl-alkenes has been challenging due to the inertness of C-F bonds and the lack of efficient activation strategies. This innovation leverages a selective C-F bond activation strategy on trifluoromethyl alkyne precursors, utilizing a robust copper catalytic system to achieve high selectivity and efficiency. The method represents a significant leap forward for reliable pharmaceutical intermediate suppliers seeking to diversify their portfolios with high-value fluorinated building blocks.
By transforming readily available trifluoromethyl alkynes into versatile 1,1-difluoro-2-propylene derivatives, this technology opens new avenues for the development of active pharmaceutical ingredients (APIs) and agrochemicals. The process is characterized by its green chemistry profile, operating under relatively mild conditions with minimal byproduct formation, which is essential for reducing downstream purification burdens. For R&D teams focused on novel drug discovery, access to such diverse fluorinated scaffolds is invaluable for structure-activity relationship (SAR) studies, enabling the rapid exploration of chemical space around the difluoromethyl pharmacophore.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art in the synthesis of 1,1-difluoro-2-propenes has been notably constrained by severe operational limitations and substrate restrictions. For instance, earlier methodologies reported by Hartwig and colleagues relied on the use of specialized difluoromethylating agents like TMSCHF2 in conjunction with cuprous iodide catalysts. These conventional routes often necessitated excessively high reaction temperatures and prolonged reaction times, leading to energy inefficiencies and potential thermal degradation of sensitive substrates. Furthermore, the scope of these traditional methods was frequently limited to a narrow range of simple substrates, failing to accommodate the complex molecular architectures often required in late-stage functionalization of drug candidates. The reliance on unstable or difficult-to-handle reagents also posed significant safety hazards and supply chain vulnerabilities for large-scale manufacturing operations.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a highly efficient copper-catalyzed borylation and defluorination sequence that operates under significantly milder conditions. By employing trifluoromethyl alkynes as the starting material, the method bypasses the need for pre-functionalized difluoromethyl sources, thereby simplifying the synthetic route and reducing raw material costs. The reaction proceeds smoothly at 90°C in an inert atmosphere, utilizing a combination of cuprous iodide, a phosphine ligand, and a strong base to facilitate the selective cleavage of the C-F bond. This streamlined process not only enhances safety but also dramatically improves the overall atom economy. As illustrated in the reaction scheme below, the transformation is direct and high-yielding, making it an ideal candidate for cost reduction in fine chemical manufacturing.
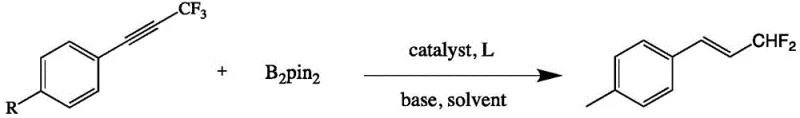
Mechanistic Insights into Copper-Catalyzed C-F Bond Activation
The core of this synthetic breakthrough lies in the intricate interplay between the copper catalyst, the phosphine ligand, and the boron reagent. Mechanistically, the reaction is believed to initiate with the oxidative addition of the copper species into the C-F bond of the trifluoromethyl group, a step that is notoriously difficult due to the high bond dissociation energy of C-F bonds. The presence of tricyclohexylphosphine (PCy3) as a ligand is critical, as screening data indicated it provides superior steric and electronic properties compared to other ligands like phenanthroline or triphenylphosphine, stabilizing the active copper intermediate and preventing catalyst deactivation. Subsequent transmetallation with the diboron reagent (B2pin2) introduces the boron moiety, which eventually serves as a leaving group or participates in the protonation step to finalize the alkene geometry.
Impurity control is another hallmark of this mechanistic pathway. The high selectivity observed across a wide range of substrates suggests that the catalytic cycle effectively suppresses competing side reactions such as homocoupling or over-reduction. The use of potassium tert-butoxide as the base ensures rapid deprotonation steps without promoting elimination reactions that could lead to alkyne polymerization. For high-purity pharmaceutical intermediates, this level of chemoselectivity is paramount, as it minimizes the formation of structurally similar impurities that are difficult to separate. The tolerance for various functional groups, including halides, esters, and ethers, further underscores the robustness of the catalytic system, allowing for the synthesis of complex molecules without the need for extensive protecting group strategies.
How to Synthesize 1,1-Difluoro-2-Propylene Compounds Efficiently
Implementing this synthesis route requires careful attention to reaction parameters to maximize yield and reproducibility. The standard protocol involves dissolving the trifluoromethyl alkyne substrate in anhydrous toluene under an argon atmosphere to prevent moisture interference. Key reagents including cuprous iodide, tricyclohexylphosphine, potassium tert-butoxide, and bis(pinacolato)diboron are added in specific stoichiometric ratios optimized through extensive screening. The mixture is then heated to 90°C for a duration of approximately 4 hours, after which the reaction is quenched and worked up using standard extraction and chromatographic techniques. Detailed standardized synthesis steps are provided in the guide below to ensure consistent results across different batches.
- Dissolve the trifluoromethyl alkyne substrate in toluene under argon protection and add cuprous iodide catalyst, tricyclohexylphosphine ligand, and potassium tert-butoxide base.
- Introduce the boron reagent B2pin2 to the reaction mixture and maintain the temperature at 90°C for approximately 4 hours to ensure complete conversion.
- Quench the reaction with water, extract with ethyl acetate, dry over anhydrous sodium sulfate, and purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this synthetic methodology offers transformative benefits that directly impact the bottom line and operational resilience. By shifting away from exotic, single-source reagents to commodity chemicals like trifluoromethyl alkynes and diboron compounds, manufacturers can significantly mitigate supply risks associated with specialized fluorinating agents. The simplified workflow reduces the number of unit operations required, which translates to lower capital expenditure on equipment and reduced labor costs per kilogram of product. Moreover, the mild reaction conditions decrease energy consumption, aligning with sustainability goals and reducing the carbon footprint of the manufacturing process. These factors collectively contribute to a more agile and cost-effective supply chain capable of responding rapidly to market demands.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous difluoromethylating reagents drastically lowers the raw material cost basis. Additionally, the high selectivity of the reaction minimizes waste generation and reduces the solvent volume required for purification, leading to substantial savings in waste disposal and solvent recovery costs. The use of earth-abundant copper catalysts instead of precious metals like palladium or rhodium further enhances the economic viability of the process for large-scale production.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis are widely available from multiple global suppliers, ensuring a stable and continuous supply of feedstock. This diversification of the supply base protects against disruptions caused by geopolitical issues or single-vendor bottlenecks. Furthermore, the robustness of the reaction conditions allows for flexible scheduling and batch processing, enabling manufacturers to maintain optimal inventory levels and reduce lead times for delivering critical intermediates to downstream customers.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work efficiently on laboratory scales with clear pathways for translation to pilot and commercial plants. The reduced generation of toxic byproducts and the use of less hazardous solvents simplify compliance with increasingly stringent environmental regulations. This 'green' profile not only avoids potential regulatory fines but also enhances the brand reputation of the manufacturer as a responsible partner in the global pharmaceutical supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and optimization studies presented in the patent documentation, providing a clear understanding of the process capabilities and limitations. Understanding these details is crucial for project managers and technical leads evaluating the feasibility of adopting this route for their specific product pipelines.
Q: What are the key advantages of this C-F activation method over traditional difluoromethylation?
A: Unlike traditional methods requiring harsh conditions or specialized reagents like TMSCHF2, this protocol utilizes readily available trifluoromethyl alkynes and operates under milder temperatures (90°C) with shorter reaction times, significantly improving operational safety and efficiency.
Q: Is this synthesis method compatible with sensitive functional groups?
A: Yes, the method demonstrates excellent functional group tolerance, successfully accommodating substrates with electron-donating groups like methoxy and tert-butyl, as well as electron-withdrawing groups such as esters, ketones, and halides, without significant side reactions.
Q: What is the expected yield range for commercial production of these intermediates?
A: While laboratory scale results show yields ranging from 50% to 79% depending on the substrate complexity, optimized commercial processes typically aim to maximize throughput and purity through refined purification protocols rather than focusing solely on isolated yield metrics.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,1-Difluoro-2-Propylene Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing advanced synthetic technologies to drive innovation in drug development. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries are seamlessly translated into reliable industrial realities. We are committed to delivering stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 1,1-difluoro-2-propylene compounds meets the highest international standards for pharmaceutical applications. Our state-of-the-art facilities are equipped to handle the specific requirements of fluorine chemistry, including specialized containment and waste treatment systems.
We invite you to collaborate with us to leverage this cutting-edge synthesis method for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to help you accelerate your development timelines and secure a competitive advantage in the marketplace. Let us be your trusted partner in navigating the complexities of fluorinated intermediate manufacturing.
