Revolutionizing Besifloxacin Intermediate Production: A Technical Deep Dive into Scalable Synthesis
The pharmaceutical industry continuously seeks robust and economically viable pathways for synthesizing critical antibiotic intermediates, particularly for next-generation fluoroquinolones like Besifloxacin. Patent CN102718711A introduces a groundbreaking methodology for the preparation of (R)-alpha-amino caprolactam hydrochloride, a pivotal chiral building block in the synthesis of this broad-spectrum antimicrobial agent. This technical disclosure addresses the longstanding challenges associated with low yields, excessive reaction times, and prohibitive costs that have historically plagued the manufacturing of this specific lactam derivative. By leveraging a direct cyclization strategy using readily available D-Lysine derivatives, the patent outlines a process that not only enhances chemical efficiency but also aligns perfectly with the rigorous demands of modern Good Manufacturing Practice (GMP) standards. For R&D directors and procurement specialists alike, understanding the nuances of this innovation is crucial for optimizing supply chains and reducing the overall cost of goods sold (COGS) in ophthalmic drug production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of (R)-alpha-amino caprolactam hydrochloride has been fraught with significant inefficiencies that hinder large-scale commercial viability. One prominent prior art method, described in US20060014945A1, relies on D-Methionine monohydrochloride as a starting material, utilizing hexamethyldisilazane (HMDS) as a catalyst in toluene. This approach is critically flawed due to its excessively long reaction duration, requiring up to one week of reflux to achieve completion, which results in a dismal yield of merely 15%. Such poor throughput makes it entirely unsuitable for industrial application where time and resource optimization are paramount. Furthermore, another established route disclosed in WO20050042489A1 employs a multi-step sequence involving N-alpha-tert-butyloxycarbonyl protected D-Lysine.
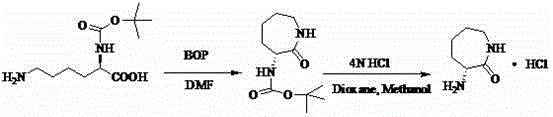
This conventional pathway necessitates the use of expensive coupling reagents like BOP in DMF solvent, followed by a separate deprotection step using strong acid. While this method avoids racemization, it suffers from high raw material costs, complex post-treatment procedures, and lower overall yields due to the cumulative losses inherent in multi-step syntheses. The reliance on specialized reagents and the generation of substantial chemical waste further exacerbate the environmental and economic burdens, creating a compelling case for the adoption of more streamlined technologies in pharmaceutical intermediates manufacturing.
The Novel Approach
In stark contrast to these cumbersome legacy processes, the novel method disclosed in CN102718711A offers a remarkably direct and efficient solution. This innovative route utilizes D-Lysine monohydrochloride or D-Lysine as the primary feedstock, reacting it directly with sodium hydroxide in an alcoholic solvent system. The process operates under heating reflux conditions, typically between 120°C and 185°C, to facilitate an intramolecular cyclization that forms the seven-membered caprolactam ring in a single pot. By eliminating the need for protecting groups and expensive coupling agents, this approach drastically simplifies the operational workflow. The use of common alcohols such as n-hexyl alcohol or 1,2-butanediol as solvents not only reduces material costs but also facilitates easier solvent recovery and recycling.

Experimental data from the patent demonstrates that this method can achieve yields as high as 80% when optimized with n-hexyl alcohol, representing a massive improvement over the 15% yield of older methods. The post-treatment is equally elegant, involving simple filtration to remove sodium chloride byproducts followed by acid extraction and crystallization. This simplicity translates directly into reduced processing time and lower energy consumption, positioning this technology as a superior choice for cost reduction in pharmaceutical intermediates manufacturing. The ability to produce high-purity material with minimal impurities makes it an attractive option for companies seeking a reliable pharmaceutical intermediates supplier capable of meeting stringent quality specifications.
Mechanistic Insights into Base-Catalyzed Intramolecular Amidation
The core chemical transformation in this novel process is a base-catalyzed intramolecular amidation, often referred to as lactamization. Mechanistically, the reaction begins with the neutralization of the ammonium salt of D-Lysine monohydrochloride by sodium hydroxide, liberating the free amine nucleophile. Under the elevated thermal conditions provided by the high-boiling alcohol solvent, the epsilon-amino group performs a nucleophilic attack on the carbonyl carbon of the carboxylic acid moiety. This cyclization event is driven by the entropy gain of releasing a water molecule and the stability of the resulting seven-membered ring structure. The choice of solvent plays a critical role here; high-boiling alcohols like n-hexyl alcohol allow the reaction to proceed at temperatures sufficient to overcome the activation energy barrier for amide bond formation without requiring exotic catalysts.
Furthermore, the reaction conditions are meticulously designed to suppress potential side reactions that could compromise optical purity. Unlike harsh acidic or metal-catalyzed conditions which might induce racemization at the alpha-carbon, this mild basic environment preserves the chiral integrity of the D-Lysine backbone. The continuous removal of water, either through azeotropic distillation with the solvent or via a water trap as described in the embodiments, shifts the equilibrium towards the product side according to Le Chatelier's principle. This mechanistic elegance ensures that the final product, (R)-alpha-amino caprolactam hydrochloride, is obtained with high enantiomeric excess, a critical parameter for the efficacy of the final Besifloxacin API. Understanding these mechanistic details allows process chemists to fine-tune parameters for commercial scale-up of complex pharmaceutical intermediates, ensuring consistent batch-to-batch quality.
How to Synthesize (R)-alpha-amino caprolactam hydrochloride Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing simplicity and safety. The procedure involves charging the amino acid salt and base into a reactor equipped with a reflux condenser and water separator, followed by heating to the specified temperature range. Monitoring the reaction progress via Thin Layer Chromatography (TLC) ensures that the cyclization is complete before proceeding to workup. The subsequent steps of filtration, acidification, and solvent swapping are standard unit operations that can be easily automated or scaled. For detailed operational parameters and safety guidelines, please refer to the standardized synthesis steps provided below.
- Charge D-Lysine monohydrochloride and sodium hydroxide into a reactor with a high-boiling alcohol solvent such as n-hexyl alcohol.
- Heat the mixture to reflux temperatures between 155°C and 175°C for approximately 6 hours while removing generated water.
- Filter off sodium chloride byproduct, extract the product into aqueous acid, and crystallize via methanol slurry to obtain high-purity solid.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this novel synthesis route offers transformative benefits for procurement managers and supply chain directors looking to optimize their sourcing strategies for ophthalmic antibiotic ingredients. The shift away from proprietary or expensive reagents towards commodity chemicals fundamentally alters the cost structure of the intermediate. By removing the dependency on coupling agents like BOP and protecting groups like Boc, manufacturers can significantly reduce the raw material bill of materials (BOM). Additionally, the simplification of the workflow from a multi-step sequence to a one-pot reaction reduces labor costs, equipment occupancy time, and utility consumption, leading to substantial overall cost savings in the production budget.
- Cost Reduction in Manufacturing: The elimination of expensive stoichiometric reagents and the reduction of reaction steps directly correlate to a lower cost per kilogram of the final product. The use of inexpensive sodium hydroxide and recyclable alcohols replaces costly specialty chemicals, allowing for a more competitive pricing model. Furthermore, the high yield of up to 80% minimizes waste of the valuable chiral starting material, D-Lysine, ensuring maximum atom economy and financial efficiency. This economic advantage is critical for maintaining margins in the highly competitive generic pharmaceutical market.
- Enhanced Supply Chain Reliability: Sourcing strategies benefit immensely from the use of widely available raw materials. D-Lysine monohydrochloride and sodium hydroxide are bulk commodities with stable global supply chains, reducing the risk of shortages that often plague specialty reagent markets. The robustness of the process means that production schedules are less likely to be disrupted by reagent lead times or quality variances. This reliability ensures a steady flow of high-purity pharmaceutical intermediates to downstream API manufacturers, supporting uninterrupted drug production timelines.
- Scalability and Environmental Compliance: The process is inherently scalable due to its reliance on simple thermal conditions and standard solvent systems. The absence of heavy metal catalysts or hazardous coupling byproducts simplifies waste treatment and regulatory compliance, aligning with green chemistry principles. The ability to recover and reuse the alcohol solvent further reduces the environmental footprint and operational costs. This scalability ensures that the technology can meet increasing market demand for Besifloxacin without requiring massive capital investment in specialized infrastructure, facilitating reducing lead time for high-purity pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing clarity on yield expectations, purity profiles, and operational feasibility. Understanding these aspects is essential for stakeholders evaluating the transition to this more efficient manufacturing platform.
Q: What are the primary advantages of this new synthesis route over traditional BOP-mediated methods?
A: The new route eliminates the need for expensive coupling reagents like BOP and protecting groups like Boc, significantly reducing raw material costs and simplifying the purification process by avoiding complex chromatography.
Q: How does the process ensure high optical purity for the final API intermediate?
A: By starting directly from chiral D-Lysine monohydrochloride and utilizing mild basic conditions without harsh racemization risks, the method preserves the stereochemical integrity, yielding a single configuration product suitable for ophthalmic applications.
Q: Is this manufacturing process scalable for commercial tonnage production?
A: Yes, the process uses common industrial solvents like n-hexyl alcohol and simple unit operations such as filtration and extraction, making it highly adaptable for large-scale commercial manufacturing without specialized equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (R)-alpha-amino caprolactam hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development and commercialization of life-saving antibiotics. Our team of expert process chemists has thoroughly analyzed the technology disclosed in CN102718711A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this efficient route to fruition. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, ensuring that every batch of (R)-alpha-amino caprolactam hydrochloride supports the efficacy and safety of your final pharmaceutical formulations.
We invite you to collaborate with us to leverage these technological advancements for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your project's success and drive value for your organization.
