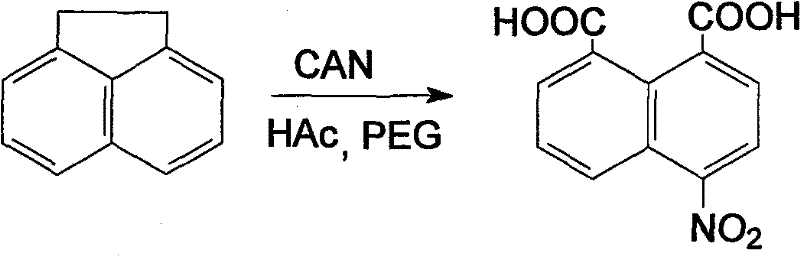
Advanced One-Step Synthesis of 4-Nitro-1,8-Naphthalic Acid Using Cerium Ammonium Nitrate for Commercial Scale Production
The chemical industry is currently witnessing a paradigm shift towards greener, more efficient synthetic routes for critical organic intermediates, particularly those serving the pharmaceutical and dye sectors. Patent CN101323576B, published in late 2011, introduces a groundbreaking methodology for the preparation of 4-nitro-1,8-naphthalic acid, a vital building block for fluorescent dyes and potential pharmaceutical scaffolds. This innovation departs radically from traditional multi-step protocols by employing cerium ammonium nitrate (CAN) as a dual-function reagent that serves simultaneously as both the nitrating and oxidizing agent. By utilizing acetic acid as the solvent and polyethylene glycol-400 (PEG-400) as a phase transfer catalyst, this process achieves the transformation of acenaphthene directly into the target dicarboxylic acid in a single operational step. For R&D directors and process chemists, this represents a significant leap forward in atom economy and operational simplicity, effectively bypassing the hazardous and corrosive conditions associated with historical methods involving strong mineral acids and hexavalent chromium salts.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 4-nitro-1,8-naphthalic acid has been plagued by severe inefficiencies and environmental liabilities inherent to legacy chemical processes. The conventional route typically necessitates a two-step sequence wherein acenaphthene is first subjected to nitration using concentrated nitric acid, followed by a separate oxidation step utilizing sodium dichromate. This bifurcated approach not only extends the total processing time but also introduces substantial risks regarding equipment corrosion due to the aggressive nature of the reagents employed. Furthermore, the reliance on sodium dichromate generates large volumes of toxic chromium-containing wastewater, imposing heavy burdens on waste treatment facilities and escalating the overall cost of compliance with environmental regulations. The isolation of intermediates between these two distinct stages further complicates the workflow, leading to cumulative yield losses and increased consumption of solvents and energy, which collectively diminish the economic viability of the traditional manufacturing pathway.
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent data streamlines the entire synthesis into a concise, one-pot reaction that dramatically reduces the chemical footprint and operational complexity. By leveraging the unique redox properties of cerium ammonium nitrate, the process accomplishes both the introduction of the nitro group and the oxidative cleavage of the ethylene bridge in acenaphthene concurrently. The inclusion of PEG-400 as a phase transfer catalyst facilitates the interaction between the organic substrate and the inorganic oxidant within the acetic acid medium, ensuring high selectivity and minimizing the formation of unwanted by-products. This integrated approach not only shortens the reaction timeline to merely 1.3 to 2.0 hours under mild thermal conditions but also eliminates the generation of carcinogenic chromium waste, aligning perfectly with modern principles of green chemistry and sustainable manufacturing practices desired by forward-thinking procurement teams.
Mechanistic Insights into CAN-Mediated Oxidative Nitration
The core of this technological advancement lies in the sophisticated mechanistic interplay between the cerium(IV) species and the acenaphthene substrate, which allows for a concerted oxidative nitration pathway. Cerium ammonium nitrate acts as a potent single-electron oxidant that initiates the reaction by generating radical cation intermediates from the electron-rich aromatic system of acenaphthene. In the presence of nitrate ions provided by the CAN reagent, these reactive intermediates undergo electrophilic attack to install the nitro functionality at the 4-position, while the powerful oxidizing capacity of Ce(IV) simultaneously drives the oxidative cleavage of the five-membered ring to form the carboxylic acid groups. This dual functionality obviates the need for separate reagents, thereby reducing the stoichiometric burden and simplifying the reaction matrix. The precise control of the molar ratio between acenaphthene, CAN, and the PEG-400 catalyst is critical, as it dictates the balance between complete oxidation and over-nitration or degradation of the naphthalene core.
Furthermore, the role of polyethylene glycol-400 cannot be overstated in terms of impurity control and yield optimization. As a phase transfer catalyst, PEG-400 forms complexes with the metal cations, effectively solubilizing the inorganic oxidant in the organic acetic acid phase and creating a homogeneous reaction environment that enhances mass transfer rates. This homogeneity ensures that the oxidation proceeds uniformly across the substrate molecules, preventing localized hotspots of high oxidant concentration that could lead to over-oxidation or ring destruction. Consequently, the impurity profile of the crude product is significantly cleaner compared to the heterogeneous mixtures typical of dichromate oxidations, facilitating easier downstream purification via simple recrystallization from ethanol. This mechanistic elegance translates directly into higher purity specifications for the final API intermediate, meeting the rigorous demands of pharmaceutical quality control.
How to Synthesize 4-Nitro-1,8-Naphthalic Acid Efficiently
Implementing this synthesis route requires careful attention to the stoichiometric ratios and thermal parameters outlined in the patent embodiments to ensure reproducibility and safety. The process begins with the dissolution of acenaphthene, cerium ammonium nitrate, and PEG-400 in glacial or dilute acetic acid, maintaining a specific molar ratio range of 1:1.0-3.0:0.14-2.8 to optimize the oxidant efficiency. The reaction mixture is then heated to a moderate temperature range of 30 to 70 degrees Celsius, or alternatively subjected to microwave irradiation for accelerated kinetics, and maintained for approximately 1.3 to 2.0 hours while monitoring conversion via thin-layer chromatography. Upon completion, the product is precipitated by pouring the reaction mass into ice water, filtered, washed to remove residual cerium salts, and finally recrystallized from ethanol to afford the high-purity yellow solid. The detailed standardized synthesis steps see the guide below.
- Dissolve acenaphthene, cerium ammonium nitrate (CAN), and polyethylene glycol-400 (PEG-400) in acetic acid solvent with a molar ratio of 1: 1.0-3.0:0.14-2.8.
- Heat the reaction mixture to 30-70°C (or use microwave irradiation at 40-65°C) and maintain for 1.3 to 2.0 hours while monitoring progress via TLC.
- Pour the reaction product into ice water to precipitate the yellow solid, filter, wash, and recrystallize from ethanol to obtain high-purity 4-nitro-1,8-naphthalic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this CAN-mediated synthesis offers profound strategic benefits that extend far beyond mere chemical curiosity. The elimination of sodium dichromate removes a heavily regulated hazardous material from the supply chain, thereby reducing the administrative and logistical overhead associated with the storage, handling, and disposal of toxic heavy metals. This shift significantly de-risks the manufacturing process, ensuring greater continuity of supply by avoiding potential shutdowns due to environmental compliance violations or waste treatment bottlenecks. Moreover, the consolidation of two distinct chemical transformations into a single reactor run drastically reduces the consumption of utilities such as steam and cooling water, while minimizing the floor space required for production equipment. These operational efficiencies culminate in a leaner, more agile manufacturing model that is better equipped to respond to fluctuating market demands for high-value dye and pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The transition to a one-step process inherently lowers the variable costs of production by reducing labor hours, solvent usage, and energy consumption per kilogram of output. By removing the expensive and environmentally costly chromium oxidation step, manufacturers can achieve substantial cost savings in waste disposal fees and effluent treatment, which are often hidden but significant components of the total cost of ownership. Additionally, the use of acetic acid as a solvent, which is cheaper and easier to recover than many chlorinated alternatives, further contributes to the economic attractiveness of this route. The qualitative improvement in process efficiency allows for a more competitive pricing structure without compromising on the quality or purity of the final 4-nitro-1,8-naphthalic acid product.
- Enhanced Supply Chain Reliability: Sourcing raw materials for this process is straightforward, as acenaphthene is a readily available commodity chemical derived from coal tar heavy oil, ensuring a stable and robust upstream supply base. The simplified reaction protocol reduces the number of unit operations and intermediate handling steps, which in turn minimizes the risk of mechanical failures or human errors that often cause delays in complex multi-step syntheses. This streamlined workflow enhances the predictability of production schedules, allowing supply chain planners to commit to tighter delivery windows with greater confidence. The reduced dependency on specialized, corrosion-resistant equipment for handling hot concentrated nitric acid or chromic acid also lowers the barrier to entry for contract manufacturing organizations, expanding the pool of qualified reliable pharma intermediates suppliers.
- Scalability and Environmental Compliance: The mild reaction conditions (30-70°C) and the absence of highly exothermic nitration spikes make this process inherently safer and easier to scale from pilot plant to commercial tonnage. The environmental profile is vastly superior, as the primary byproduct is cerium(III) salt, which is significantly less toxic and easier to manage than hexavalent chromium waste, facilitating smoother regulatory approvals in jurisdictions with strict environmental laws. This alignment with green chemistry principles not only future-proofs the manufacturing asset against tightening regulations but also enhances the brand reputation of the end-product users who are increasingly demanding sustainable sourcing. The ability to commercial scale-up of complex intermediates with minimal environmental impact is a decisive factor for long-term partnerships in the global chemical market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route, based on the specific data points and advantages highlighted in the patent literature. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this technology for their own production lines or for procurement specialists assessing the value proposition of suppliers utilizing this method. The answers provided reflect the consensus on the operational benefits and chemical realities of using cerium ammonium nitrate in oxidative transformations.
Q: What are the primary advantages of using Cerium Ammonium Nitrate (CAN) over traditional chromic acid oxidation?
A: The use of CAN eliminates the need for toxic sodium dichromate, significantly reducing heavy metal waste disposal costs and environmental hazards while combining nitration and oxidation into a single operational step.
Q: How does the addition of PEG-400 influence the reaction selectivity?
A: Polyethylene glycol-400 acts as an effective phase transfer catalyst that enhances the solubility of reactants in the acetic acid medium, thereby improving reaction selectivity and overall yield without requiring harsh conditions.
Q: Is this synthesis method scalable for industrial production of dye intermediates?
A: Yes, the process utilizes readily available raw materials like acenaphthene from coal tar and operates at moderate temperatures (30-70°C), making it highly suitable for commercial scale-up with simplified equipment requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Nitro-1,8-Naphthalic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced synthetic methodologies like the CAN-mediated oxidation described in patent CN101323576B to deliver superior value to our global clientele. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this green chemistry approach are fully realized in practical, large-scale manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs and stringent purity specifications that guarantee every batch of 4-nitro-1,8-naphthalic acid meets the highest international standards for pharmaceutical and specialty chemical applications. We are committed to leveraging such innovative processes to provide our partners with a consistent, high-quality supply of critical intermediates.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can specifically benefit your project requirements and cost structures. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this chromium-free methodology for your specific volume needs. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your development timeline, ensuring that your supply chain remains robust, compliant, and cost-effective in an evolving regulatory landscape.
