Advanced Polymer-Supported Palladium Catalyst for Efficient Furfural Decarbonylation and Commercial Scale-Up
Advanced Polymer-Supported Palladium Catalyst for Efficient Furfural Decarbonylation and Commercial Scale-Up
The chemical industry is currently witnessing a paradigm shift towards sustainable biomass conversion, specifically targeting the efficient transformation of renewable carbohydrates into high-value fuels and chemicals. A pivotal advancement in this domain is detailed in patent CN108129424B, which discloses a novel method for the decarbonylation of furfural derivatives using a bidentate phosphine ligand polymer-supported palladium catalyst. This technology addresses the critical bottleneck of catalyst recovery in homogeneous systems by employing a vinyl-functionalized bidentate phosphine ligand that is polymerized to create a robust heterogeneous support. The resulting catalytic system not only exhibits exceptional thermal stability and resistance to strong acids and bases but also operates with significantly reduced palladium loading while maintaining high conversion rates. For R&D directors and process engineers, this represents a substantial leap forward in designing greener, more cost-effective pathways for producing furan derivatives from bio-based feedstocks like 5-hydroxymethylfurfural (HMF).

The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the decarbonylation of aldehydes, particularly biomass-derived furfurals, has relied heavily on homogeneous noble metal catalysts such as rhodium, ruthenium, or palladium complexes. While effective in laboratory settings, these homogeneous systems suffer from severe industrial drawbacks, primarily the difficulty in separating the expensive metal catalyst from the product mixture, leading to potential metal contamination and high operational costs. Previous attempts to heterogenize these catalysts, such as the use of SBA-15 mesoporous silica supports, have shown promise but are often hampered by complex and expensive carrier preparation procedures, narrow substrate adaptability, and the frequent requirement for large amounts of molecular sieves as additives to drive the reaction equilibrium. Furthermore, many conventional methods necessitate rigorous inert gas protection and long reaction times, which complicates the engineering controls required for large-scale manufacturing and increases the overall energy footprint of the process.
The Novel Approach
The innovative strategy presented in the patent data overcomes these legacy limitations by utilizing a specifically designed vinyl-functionalized bidentate phosphine ligand that undergoes polymerization to form an insoluble, cross-linked network known as POL-Xantphos. This polymer matrix effectively traps the palladium active sites, preventing leaching while allowing substrates to access the catalytic centers freely. Unlike silica-supported systems, this organic polymer support is synthesized through a straightforward radical polymerization of the functionalized ligand, eliminating the need for complex inorganic carrier synthesis. The resulting heterogeneous catalyst system demonstrates broad substrate applicability, short reaction times, and excellent selectivity without the need for excessive additives or stringent gas protection. This approach successfully bridges the gap between the high activity of homogeneous catalysis and the ease of separation associated with heterogeneous systems, offering a scalable solution for the bio-refinery sector.
Mechanistic Insights into Pd(OAc)2/POL-Xantphos Catalyzed Decarbonylation
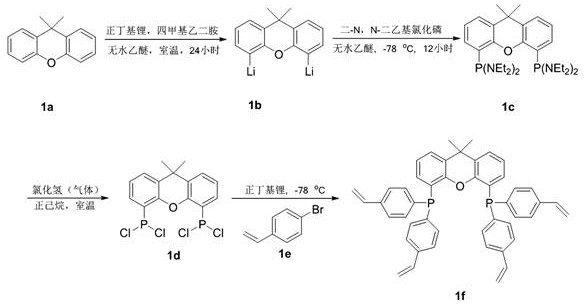
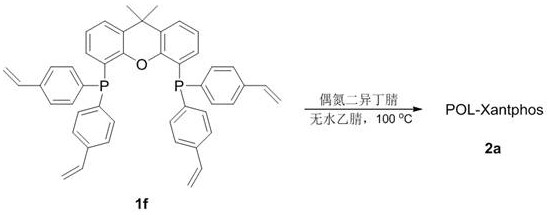
The core of this technological breakthrough lies in the precise molecular architecture of the ligand and its integration into the polymer backbone. The synthesis begins with the lithiation of 9,9-dimethylxanthene, followed by phosphorylation to introduce the phosphine groups, and finally coupling with p-bromostyrene to install the polymerizable vinyl handles. This creates a rigid, bulky Xantphos-type backbone which is renowned for stabilizing palladium centers and facilitating reductive elimination steps crucial for decarbonylation. By copolymerizing this monomer, the resulting POL-Xantphos provides a microenvironment that mimics homogeneous conditions locally while remaining macroscopically solid. The palladium acetate coordinates with the phosphine sites within the polymer matrix, creating active species that are sterically protected from aggregation into inactive palladium black, a common deactivation pathway in unsupported systems.
Furthermore, the impurity control mechanism is inherently superior in this heterogeneous setup. In homogeneous reactions, trace metal residues often persist in the final API or intermediate, requiring costly scavenging steps. Here, the insolubility of the POL-Xantphos support allows for physical separation via simple centrifugation or filtration post-reaction. The patent data indicates that the catalyst retains its structural integrity and activity over multiple cycles, suggesting that the covalent bonding of the phosphine ligands to the polymer chain prevents ligand dissociation and metal leaching. This stability is further enhanced by the thermal robustness of the xanthene backbone, which withstands the elevated reaction temperatures of 140°C without degradation. Consequently, the process yields furan derivatives with high purity, minimizing downstream purification burdens and ensuring compliance with stringent quality specifications required for pharmaceutical and fine chemical applications.
How to Synthesize POL-Xantphos Efficiently
The synthesis of the core catalytic material involves a multi-step sequence that transforms readily available starting materials into a high-performance heterogeneous catalyst. The process initiates with the functionalization of the xanthene scaffold to create the active ligand monomer, followed by a controlled radical polymerization to generate the solid support. Finally, the palladium source is loaded onto the polymer to activate the system for decarbonylation reactions. This modular approach allows for precise tuning of the catalyst properties and ensures batch-to-batch consistency, which is critical for industrial adoption. The detailed standardized synthesis steps for producing this advanced catalyst and executing the decarbonylation reaction are outlined in the guide below.
- Synthesize the vinyl-functionalized bidentate phosphine ligand monomer (4,5-bis(4-vinylphenyl)phosphino-9,9-dimethylxanthene) via lithiation and phosphorylation of 9,9-dimethylxanthene followed by coupling with p-bromostyrene.
- Polymerize the ligand monomer using azobisisobutyronitrile (AIBN) in acetonitrile at 100°C to form the insoluble POL-Xantphos polymer support, then load with palladium acetate.
- Perform the decarbonylation reaction by heating the furfural derivative with potassium carbonate and the Pd/POL-Xantphos catalyst in 1,4-dioxane at 140°C for 6 hours.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this polymer-supported catalytic system offers tangible strategic benefits that directly impact the bottom line and operational resilience. The primary advantage stems from the drastic reduction in the consumption of expensive palladium resources; because the catalyst is heterogeneous and recyclable, the effective cost per kilogram of product is significantly lowered compared to single-use homogeneous systems. Additionally, the elimination of complex separation units and metal scavengers simplifies the manufacturing workflow, reducing both capital expenditure on equipment and operational expenditure on consumables. The robustness of the catalyst also means fewer production stoppages for catalyst replacement or regeneration, thereby enhancing overall plant throughput and reliability.
- Cost Reduction in Manufacturing: The implementation of this technology leads to substantial cost savings by minimizing the usage of precious palladium metals, as the catalyst can be recovered and reused multiple times without significant loss of activity. The simplified work-up procedure, which avoids the need for expensive chromatographic purification or metal scavenging resins typically required for homogeneous catalysts, further drives down the variable costs associated with production. Moreover, the high reaction rates and short cycle times increase the asset utilization rate of the reactors, allowing for higher production volumes within the same timeframe and infrastructure.
- Enhanced Supply Chain Reliability: The raw materials required for synthesizing the catalyst, such as 9,9-dimethylxanthene and p-bromostyrene, are commercially available and do not rely on obscure or geopolitically sensitive supply chains. The operational simplicity of the reaction, which proceeds without the need for specialized inert gas protection or extreme pressure conditions, reduces the dependency on complex utility systems and specialized safety protocols. This ease of operation translates to a more resilient supply chain that is less prone to disruptions caused by equipment failure or logistical bottlenecks associated with handling hazardous gases or sensitive reagents.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this heterogeneous catalytic system aligns perfectly with green chemistry principles by reducing waste generation and solvent usage. The ability to recycle the catalyst ten times or more significantly decreases the volume of heavy metal waste that requires disposal, simplifying compliance with increasingly strict environmental regulations. The process is inherently scalable, moving seamlessly from gram-scale laboratory optimization to multi-ton commercial production without the engineering challenges often associated with scaling up homogeneous catalytic processes, ensuring a smooth path to market for new bio-based products.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this polymer-supported palladium catalyst technology. These insights are derived directly from the experimental data and performance metrics reported in the patent literature, providing a clear picture of the system's capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this method into existing production lines or developing new synthetic routes for furan derivatives.
Q: Can the polymer-supported palladium catalyst be recycled without losing activity?
A: Yes, the patent data demonstrates that the Pd(OAc)2/POL-Xantphos catalyst can be reused at least ten times via simple centrifugation and washing without significant loss of catalytic activity or yield.
Q: What are the advantages of this heterogeneous system over traditional homogeneous catalysts?
A: Unlike homogeneous systems which require complex separation processes, this polymer-supported system allows for easy filtration or centrifugation, significantly reducing metal contamination in the final product and lowering operational costs.
Q: Does this method require inert gas protection during the reaction?
A: No, one of the key operational advantages highlighted in the technology is that the reaction proceeds efficiently at 140°C without the need for stringent inert gas protection, simplifying the industrial setup.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable POL-Xantphos Supplier
As the global demand for sustainable chemical intermediates grows, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM ensures that your transition to advanced catalytic technologies is seamless and successful. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, leveraging our state-of-the-art facilities to deliver high-purity intermediates that meet the most rigorous industry standards. Our team of expert chemists is well-versed in handling complex organometallic systems and polymer-supported catalysts, ensuring that every batch is produced with stringent purity specifications and validated through our rigorous QC labs to guarantee consistency and performance.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through the adoption of this cutting-edge decarbonylation technology. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific production needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can accelerate your project timelines and enhance your competitive advantage in the market.
