Advanced Palladium-Catalyzed Synthesis of E-3-Aryl-1-Fluoro-1,3-Butadiene Phosphonates for Pharmaceutical Applications
Advanced Palladium-Catalyzed Synthesis of E-3-Aryl-1-Fluoro-1,3-Butadiene Phosphonates for Pharmaceutical Applications
The landscape of modern medicinal chemistry is increasingly defined by the strategic incorporation of fluorine atoms into organic scaffolds to modulate metabolic stability and binding affinity. A pivotal advancement in this domain is detailed in patent CN109503658B, which discloses a highly efficient synthesis method for (E)-3-aryl-1-fluoro-1,3-butadiene phosphonate compounds. These molecules represent a critical class of bioisosteres, where the monofluoro alkenyl structure serves as a robust mimic of the amido bond, resisting enzymatic hydrolysis while maintaining essential spatial geometry. As illustrated in the structural comparisons below, the transition from standard amides to fluorinated olefins and phosphonates allows for the precise tuning of biological activity, particularly in the realm of enzyme inhibition.
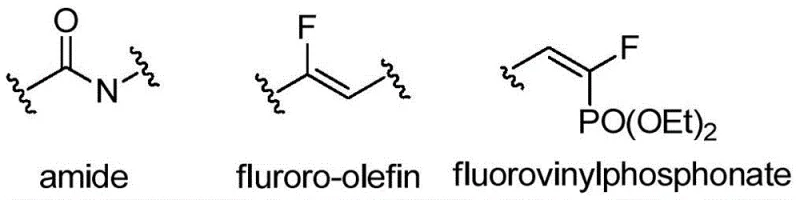
The patented technology addresses a significant gap in the availability of high-purity fluorovinylphosphonates, which are essential intermediates for developing next-generation antitumor agents. By leveraging a metal palladium-catalyzed coupling defluorination strategy, the inventors have achieved exceptional control over both regional and stereochemical outcomes. This breakthrough not only simplifies the synthetic pathway but also ensures the exclusive formation of the biologically active E-configuration, a feat that has historically challenged organic chemists working with complex fluorinated allenes. For R&D directors seeking reliable sources of specialized fluorine-containing building blocks, this methodology represents a paradigm shift towards more predictable and scalable manufacturing processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of monofluoroalkenyl phosphonates was fraught with substantial technical hurdles that impeded their widespread adoption in drug discovery pipelines. Traditional routes often suffered from poor regioselectivity, resulting in complex mixtures of Z and E isomers that required tedious and yield-depleting separation techniques. Furthermore, existing methodologies frequently necessitated harsh reaction conditions, including extreme temperatures or the use of hazardous reagents, which posed significant safety risks and environmental burdens during scale-up. The low overall yields associated with these legacy processes meant that the cost of goods for the final active pharmaceutical ingredient (API) was prohibitively high, limiting the commercial viability of potential drug candidates relying on this scaffold. Additionally, the lack of substrate generality in older methods restricted the chemical space that could be explored, preventing medicinal chemists from fully optimizing the pharmacokinetic profiles of their lead compounds.
The Novel Approach
In stark contrast, the novel approach described in the patent utilizes a sophisticated palladium-catalyzed cross-coupling reaction that seamlessly integrates a defluorination step to construct the target carbon-carbon double bond with precision. The core transformation involves the reaction of an alpha,alpha-difluoromethylene-beta-allene phosphonate with an aryl boronic acid in the presence of a palladium catalyst and a phosphine ligand. As depicted in the general reaction scheme, this process selectively targets the 2,3-position of the allene system, triggering a cascade that eliminates one fluorine atom and installs the aryl group with perfect stereocontrol. The reaction proceeds under remarkably mild conditions, typically at 50°C in toluene with potassium hydroxide as the base, eliminating the need for cryogenic cooling or high-pressure equipment. This streamlined protocol not only drastically reduces energy consumption but also simplifies the downstream purification process, allowing for the isolation of the desired E-isomer in excellent yields ranging from 80% to 94%.
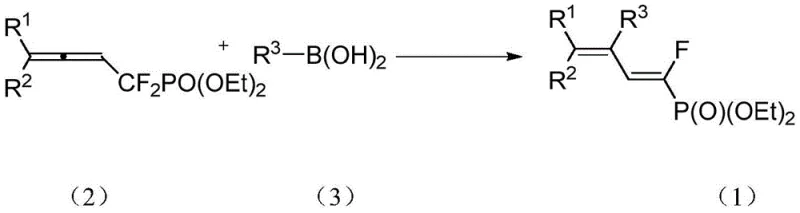
Mechanistic Insights into Palladium-Catalyzed Coupling Defluorination
The success of this synthetic route hinges on the intricate interplay between the palladium catalyst and the unique electronic properties of the fluorinated allene substrate. The catalytic cycle likely initiates with the oxidative addition of the low-valent palladium species into the carbon-fluorine bond or the activation of the allene system, facilitated by the electron-withdrawing nature of the phosphonate group. This activation renders the adjacent carbon susceptible to nucleophilic attack or transmetallation with the aryl boronic acid species, which is activated by the hydroxide base to form a reactive boronate complex. The subsequent reductive elimination step is the stereochemistry-determining event, where the steric bulk of the ligands and the thermodynamic stability of the E-alkene drive the formation of the single observed isomer. The use of triphenylphosphine (PPh3) as a ligand provides the optimal balance of electronic donation and steric hindrance to promote this selective transformation while suppressing side reactions such as homocoupling of the boronic acid or polymerization of the allene.
From an impurity control perspective, the high selectivity of this reaction is a major advantage for pharmaceutical manufacturing, as it minimizes the generation of difficult-to-remove geometric isomers. The mechanism inherently favors the formation of the thermodynamically more stable E-configuration, ensuring that the crude reaction mixture is already enriched with the target product before purification. This reduces the burden on chromatographic columns and crystallization steps, which are often the bottlenecks in process chemistry. Furthermore, the tolerance of the catalytic system to various functional groups on the aryl boronic acid, such as methoxy or fluoro substituents, demonstrates the robustness of the method. For instance, the synthesis of diethyl (E)-(1-fluoro-4-methyl-3-phenylpenta-1,3-dien-1-yl)phosphonate proceeds with a remarkable 94% yield, showcasing the efficiency of the catalyst system in handling sterically demanding substrates without compromising purity.
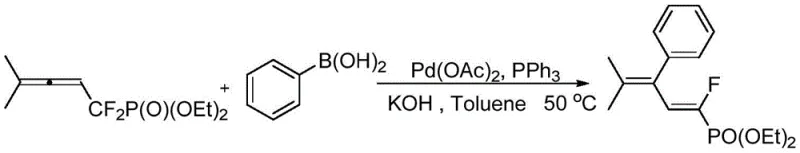
How to Synthesize (E)-3-Aryl-1-Fluoro-1,3-Butadiene Phosphonate Efficiently
The practical implementation of this patented methodology offers a straightforward path for process chemists to access these valuable intermediates with minimal operational complexity. The procedure involves charging a reaction vessel with the difluoroallene precursor, the appropriate aryl boronic acid, and the catalytic system consisting of palladium acetate and triphenylphosphine. The reaction is conducted in toluene with potassium hydroxide as the base, heated to a moderate 50°C, and monitored via thin-layer chromatography to ensure complete conversion. Following the reaction, a simple workup involving filtration and solvent removal yields the crude product, which can be further purified by standard silica gel chromatography. For a detailed, step-by-step guide on executing this synthesis with precise stoichiometric ratios and safety precautions, please refer to the standardized protocol below.
- Combine alpha,alpha-difluoromethylene-beta-allene phosphonate, aryl boronic acid, Pd(OAc)2 catalyst, PPh3 ligand, and KOH base in toluene solvent.
- Heat the reaction mixture to 50°C under air or inert atmosphere and stir until TLC monitoring indicates complete consumption of the starting allene.
- Filter the mixture, concentrate the filtrate, and purify the crude residue via column chromatography using a petroleum ether-ethyl acetate system to isolate the pure E-isomer.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route translates into tangible strategic benefits that extend beyond mere chemical elegance. The shift towards a milder, higher-yielding process directly impacts the cost structure of the supply chain by reducing raw material waste and energy expenditure. Unlike legacy methods that might require expensive cryogenic infrastructure or exotic reagents, this protocol utilizes commodity chemicals like toluene and KOH, which are readily available in the global market at stable prices. The high atom economy and selectivity mean that less starting material is required to produce the same amount of API intermediate, effectively lowering the cost per kilogram of the final product. This efficiency is crucial for maintaining competitive pricing in the generic pharmaceutical market, where margin compression is a constant challenge.
- Cost Reduction in Manufacturing: The elimination of complex separation steps for Z/E isomers significantly lowers processing costs, as the reaction inherently produces the desired E-isomer with high fidelity. By avoiding the need for extensive recycling of unwanted isomers or the use of chiral resolving agents, manufacturers can achieve substantial cost savings in both labor and materials. The use of a robust palladium catalyst system that operates at low loading further contributes to cost efficiency, as precious metal recovery becomes more manageable and the overall catalyst consumption is minimized. Additionally, the simplified workup procedure reduces the volume of solvents and consumables required for purification, leading to a leaner and more cost-effective manufacturing operation that aligns with strict budgetary constraints.
- Enhanced Supply Chain Reliability: The reliance on stable, commercially available starting materials such as aryl boronic acids and difluoroallenes ensures a resilient supply chain that is less susceptible to disruptions. Since the reaction conditions are mild and do not require specialized high-pressure reactors or extreme temperature control, the process can be easily transferred between different manufacturing sites or contract manufacturing organizations (CMOs) without significant re-validation efforts. This flexibility allows supply chain managers to diversify their supplier base and mitigate risks associated with single-source dependencies. The robustness of the chemistry also means that batch-to-batch variability is minimized, ensuring a consistent supply of high-quality intermediates that meet the rigorous specifications required for downstream drug substance production.
- Scalability and Environmental Compliance: The scalability of this process is evidenced by its ability to maintain high yields and selectivity even as the reaction scale increases, making it suitable for commercial production ranging from pilot plant to multi-ton manufacturing. The use of toluene as a solvent, while requiring proper handling, is well-established in the industry with mature recovery and recycling protocols, facilitating compliance with environmental regulations regarding volatile organic compound (VOC) emissions. Furthermore, the high selectivity of the reaction reduces the generation of hazardous waste streams associated with byproduct formation, supporting the industry's move towards greener chemistry practices. This alignment with sustainability goals not only satisfies regulatory requirements but also enhances the corporate social responsibility profile of the manufacturing entity, appealing to environmentally conscious stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these fluorinated phosphonate compounds. The answers are derived directly from the experimental data and technical disclosures within the patent documentation, providing a reliable basis for decision-making. Understanding these details is essential for evaluating the feasibility of integrating this technology into existing production workflows or new drug development programs.
Q: What are the primary advantages of this palladium-catalyzed method over conventional synthesis routes?
A: This method offers superior regioselectivity and stereoselectivity, specifically producing the E-configuration isomer with yields ranging from 80% to 94%, while operating under mild conditions (50°C) compared to harsher traditional protocols.
Q: What are the biological applications of these fluorovinylphosphonate compounds?
A: These compounds serve as potent enzyme inhibitors and phosphate ester mimics, showing significant potential in the development of antitumor medicaments and polypeptide mimetics due to their resistance to enzymatic hydrolysis.
Q: Is this synthesis protocol suitable for large-scale commercial manufacturing?
A: Yes, the process utilizes readily available reagents like KOH and toluene, avoids extreme temperatures or pressures, and employs a simple workup procedure, making it highly amenable to scale-up for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluorovinylphosphonate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality fluorinated intermediates play in the development of life-saving medications. Our team of expert process chemists has extensively evaluated the methodology described in CN109503658B and possesses the technical capability to execute this palladium-catalyzed coupling defluorination with precision and consistency. We offer comprehensive CDMO services tailored to the unique needs of the pharmaceutical industry, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to ensure that every batch of fluorovinylphosphonate meets stringent purity specifications, guaranteeing the safety and efficacy of your final drug product.
We invite you to collaborate with us to optimize your supply chain and accelerate your drug development timelines. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis that identifies opportunities to reduce manufacturing expenses without compromising quality. We encourage you to contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary molecules. Let us be your trusted partner in navigating the complexities of fluorine chemistry and delivering value to your organization through innovation and reliability.
