Advanced Ruthenium Catalysis for Scalable Benzofuran-4-Carboxylic Acid Production
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to access complex heterocyclic scaffolds, particularly those found in bioactive natural products. Patent CN113754619B discloses a groundbreaking ruthenium-catalyzed method for preparing multi-substituted benzofuran-4-carboxylic acid compounds, addressing critical limitations in current synthetic methodologies. This innovation leverages a tandem carbon-hydrogen bond alkenylation and oxidative cyclization strategy, utilizing readily available m-hydroxybenzoic acids and diaryl alkynes as starting materials. By employing air or oxygen as a green oxidant, the process eliminates the need for hazardous stoichiometric oxidants, aligning perfectly with modern green chemistry principles. The versatility of this catalytic system allows for the efficient construction of the 2,3-diarylbenzofuran core, which serves as a pivotal intermediate for synthesizing potent immunosuppressants and anticancer agents like diptoindonesin G. 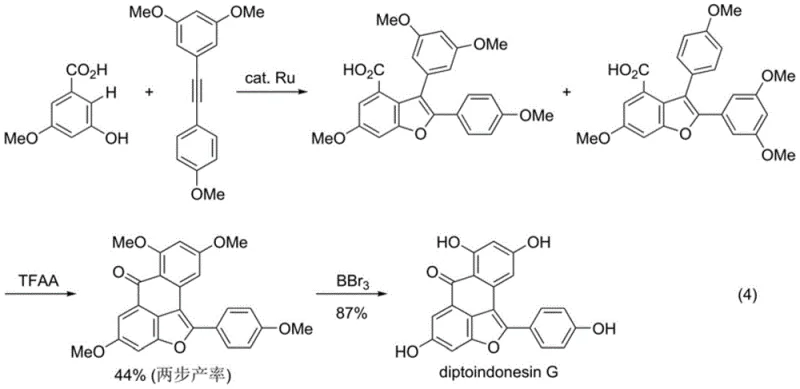
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzofuran derivatives has relied heavily on palladium, copper, gold, or zinc chloride-promoted systems, which present significant drawbacks for industrial application. Existing literature indicates that when hydroxybenzoic acids are used as substrates in these traditional transition-metal catalyzed reactions, the carboxyl group often interferes, leading to the formation of unwanted hydroxyisocoumarins or naphthol byproducts rather than the desired benzofuran ring. Furthermore, many of these conventional protocols require the use of toxic, dangerous, or prohibitively expensive chemical oxidants to drive the oxidative cyclization step, creating substantial waste disposal challenges and safety hazards in a manufacturing environment. The operational complexity is further compounded by the frequent need for rigorous exclusion of moisture and oxygen, necessitating specialized equipment and inert atmosphere techniques that increase capital expenditure. Additionally, the substrate scope of previous methods is often narrow, failing to accommodate diverse functional groups without compromising yield or selectivity. These cumulative inefficiencies result in higher production costs and longer lead times, making the reliable supply of high-purity benzofuran intermediates a persistent bottleneck for drug development pipelines.
The Novel Approach
The disclosed invention overcomes these hurdles by introducing a highly active ruthenium catalyst system that uniquely directs the reaction towards benzofuran-4-carboxylic acid formation with exceptional chemoselectivity. This novel approach utilizes the carboxyl group not as a hindrance, but as a directing group to assist the ruthenium in activating the specific carbon-hydrogen bond required for cyclization, thereby preventing the formation of isocoumarin side products. A defining feature of this methodology is its ability to operate under air or oxygen atmospheres, replacing hazardous chemical oxidants with benign molecular oxygen and generating water as the sole byproduct. The reaction conditions are remarkably mild and robust, tolerating water and proceeding effectively in polar aprotic solvents like gamma-valerolactone (GVL), which reduces the environmental footprint of the manufacturing process. Operational simplicity is a key advantage, as the procedure does not require special experimental equipment for deoxygenation or drying, facilitating easier technology transfer from laboratory to pilot plant. This robustness ensures consistent quality and yield across various substituted substrates, providing a dependable solution for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Ruthenium-Catalyzed Cyclization
The core of this technological advancement lies in the unique mechanistic pathway facilitated by the ruthenium catalyst, which orchestrates a sequential C-H activation and oxidative annulation. Initially, the carboxyl group on the m-hydroxybenzoic acid substrate coordinates with the ruthenium center, acting as a powerful directing group to position the metal proximal to the target C-H bond between the carboxyl and hydroxyl functionalities. This coordination enables the selective cleavage of the carbon-hydrogen bond, forming a stable ruthenacycle intermediate that is primed for subsequent transformation. The diaryl alkyne then undergoes insertion into the ruthenium-carbon bond, extending the molecular framework and setting the stage for ring closure. 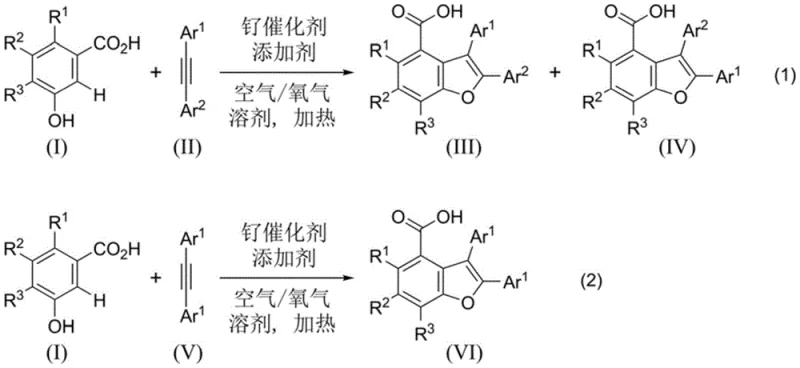
Following the alkenylation step, the intermediate undergoes an oxidative cyclization where the phenolic hydroxyl group attacks the activated alkyne moiety to construct the furan ring. The presence of specific additives, such as magnesium acetate tetrahydrate, plays a crucial role in promoting this cyclization and stabilizing the catalytic cycle, ensuring high turnover numbers. The use of air or oxygen serves to re-oxidize the reduced ruthenium species back to its active state, closing the catalytic loop without consuming stoichiometric amounts of metal oxidants. This mechanism effectively suppresses common side reactions associated with free radical pathways or non-directed metalation, resulting in a cleaner impurity profile. Understanding this precise mechanistic interplay allows process chemists to fine-tune reaction parameters, such as temperature and additive loading, to maximize efficiency and minimize the formation of regioisomers, which is critical for meeting stringent purity specifications in API synthesis.
How to Synthesize Benzofuran-4-Carboxylic Acid Efficiently
Implementing this synthesis route requires careful attention to reagent ratios and reaction conditions to ensure optimal conversion and selectivity. The patent outlines a straightforward protocol where m-hydroxybenzoic acid derivatives and diaryl alkynes are combined with a ruthenium catalyst precursor and a magnesium-based additive in a green solvent system. The reaction is typically conducted at elevated temperatures between 90°C and 120°C for a duration of 6 to 30 hours, depending on the electronic nature of the substrates. Detailed standardized synthesis steps see the guide below.
- Combine m-hydroxybenzoic acid compound, diaryl alkyne, ruthenium catalyst (e.g., p-cymene ruthenium dichloride dimer), additive (e.g., magnesium acetate), and solvent (e.g., GVL) in a reaction vessel.
- Heat the mixture to 90-120°C under an air or oxygen atmosphere and stir for 6-30 hours to facilitate C-H activation and cyclization.
- Cool the reaction, dilute with ethyl acetate, filter through diatomaceous earth, extract with saturated salt solution, dry the organic phase, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, this patented technology offers tangible benefits that directly impact the bottom line and operational resilience. By shifting from precious metals like palladium or gold to ruthenium, which is relatively cheaper among noble metal catalysts, the direct material costs associated with catalysis are significantly optimized. The elimination of expensive and hazardous chemical oxidants not only reduces raw material expenses but also simplifies waste treatment protocols, leading to substantial cost savings in environmental compliance and disposal. Furthermore, the use of commercially available and structurally simple starting materials ensures a stable supply chain, reducing the risk of shortages that often plague complex custom synthetic building blocks. The robustness of the reaction against moisture and oxygen means that manufacturing can proceed with less stringent environmental controls, lowering facility overheads and energy consumption associated with inert gas generation. These factors combine to create a highly scalable process that enhances supply chain reliability while maintaining high standards of product quality.
- Cost Reduction in Manufacturing: The substitution of costly oxidants with air or oxygen drastically lowers the variable costs per kilogram of produced intermediate. Additionally, the use of a ruthenium catalyst, which is more economical than palladium or gold alternatives, contributes to a leaner cost structure for the overall synthesis. The simplified workup procedure, which avoids complex quenching of reactive oxidants, further reduces labor and processing time, enhancing overall manufacturing efficiency. These cumulative effects result in a more competitive pricing model for high-purity pharmaceutical intermediates without compromising on quality or yield.
- Enhanced Supply Chain Reliability: The reliance on widely available commodity chemicals like m-hydroxybenzoic acids and diaryl alkynes mitigates the risk of supply disruptions common with exotic reagents. The tolerance of the reaction to ambient conditions reduces the dependency on specialized infrastructure, allowing for more flexible production scheduling and faster turnaround times. This flexibility is crucial for responding to fluctuating market demands and ensures a continuous flow of materials for downstream drug synthesis. Consequently, partners can expect reduced lead times for high-purity intermediates and greater confidence in long-term supply agreements.
- Scalability and Environmental Compliance: The process has been validated at the gram scale with indications of smooth translation to larger batches, supported by the use of high-boiling, recyclable solvents like GVL. The generation of water as the primary byproduct aligns with increasingly strict global environmental regulations, minimizing the ecological footprint of the manufacturing process. This green chemistry profile facilitates easier regulatory approval and reduces the burden of hazardous waste management, making it an ideal candidate for sustainable commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this ruthenium-catalyzed synthesis platform. These answers are derived directly from the experimental data and comparative examples provided in the patent documentation to ensure accuracy and relevance. Understanding these details helps stakeholders evaluate the feasibility of integrating this technology into their existing production workflows. We encourage technical teams to review these points when assessing process viability.
Q: Why is this ruthenium method superior to traditional palladium catalysis for benzofuran synthesis?
A: Traditional palladium systems often fail with hydroxybenzoic acid substrates, yielding hydroxyisocoumarins instead of benzofurans. This ruthenium method specifically targets the formation of benzofuran-4-carboxylic acids with high selectivity.
Q: What oxidants are required for this reaction?
A: Unlike conventional methods requiring toxic or expensive chemical oxidants, this process utilizes air or molecular oxygen as a green oxidant, producing only water as a byproduct.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the patent demonstrates successful gram-scale synthesis with simple operation and no need for strict inert gas protection, indicating strong potential for industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzofuran-4-Carboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this ruthenium-catalyzed methodology for accelerating the development of next-generation therapeutics. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications required for clinical and commercial API production. We are committed to leveraging this advanced catalytic technology to deliver high-quality intermediates that support your drug development timelines.
We invite you to contact our technical procurement team to discuss how we can tailor this synthesis route to your specific needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this greener, more efficient process. We are ready to provide specific COA data and route feasibility assessments to demonstrate our capability as your trusted partner in fine chemical manufacturing.
