Advanced Rhodium-Catalyzed Synthesis of Cyclohexanone Fused Pyrones for Commercial Pharmaceutical Applications
Advanced Rhodium-Catalyzed Synthesis of Cyclohexanone Fused Pyrones for Commercial Pharmaceutical Applications
The pharmaceutical industry constantly seeks efficient pathways to access complex heterocyclic scaffolds that serve as critical building blocks for novel therapeutics. Patent CN111978281A introduces a groundbreaking method for preparing cyclohexanone and chromenopyrone compounds using a highly selective rhodium-catalyzed system. This technology represents a significant leap forward in organic synthesis, offering a streamlined route to structures that have demonstrated potent inhibitory activity against various cancer cell lines, including MCF-7 and A549. By leveraging Cp*Rh(III) catalysis, this process achieves high stereoselectivity and regioselectivity under remarkably mild conditions, addressing long-standing challenges in the manufacturing of high-purity pharmaceutical intermediates. The ability to construct these fused ring systems in a single step from readily available starting materials positions this technology as a vital asset for R&D teams aiming to accelerate drug discovery pipelines while maintaining rigorous quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes to cyclohexanone fused pyrone derivatives often involve multi-step sequences that are both time-consuming and resource-intensive. Conventional methodologies frequently require harsh reaction conditions, including extreme temperatures or the use of hazardous reagents that complicate waste management and increase operational risks. Furthermore, achieving the necessary stereochemical control in these classical approaches often demands extensive protection and deprotection strategies, which inevitably lower the overall atom economy and yield. The reliance on inert atmospheres and strictly anhydrous conditions in many legacy processes adds another layer of complexity, requiring specialized equipment and highly trained personnel to prevent reaction failure. These cumulative inefficiencies result in prolonged lead times and escalated production costs, creating significant bottlenecks for procurement managers tasked with securing reliable supplies of complex intermediates for clinical and commercial programs.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a rhodium-catalyzed annulation strategy that dramatically simplifies the synthetic landscape. By reacting alpha,beta-unsaturated carboxylic acids with cyclic dione-phenyl iodides in the presence of a Cp*Rh catalyst, the process achieves direct C-H activation and cyclization in a single operational step. This method operates under mild heating conditions, typically between 60-100°C, and notably proceeds efficiently in air without the need for inert gas protection, thereby reducing equipment overhead. The use of hexafluoroisopropanol (HFIP) as a solvent further enhances reaction efficiency and selectivity, enabling the formation of the target fused ring systems with exceptional precision. This streamlined workflow not only accelerates the synthesis timeline but also minimizes the generation of chemical waste, aligning perfectly with modern green chemistry principles and sustainability goals.
Mechanistic Insights into Rhodium-Catalyzed C-H Activation and Cyclization
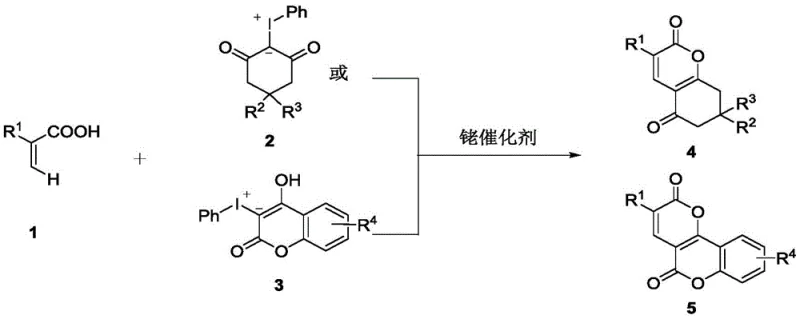
The core of this technological advancement lies in the sophisticated catalytic cycle driven by the Cp*Rh(III) complex, which facilitates the activation of inert C-H bonds with remarkable specificity. The mechanism initiates with the coordination of the rhodium catalyst to the unsaturated carboxylic acid, followed by a concerted metalation-deprotonation step that generates a key rhodacycle intermediate. This active species then undergoes oxidative addition with the hypervalent iodine reagent, setting the stage for the subsequent migratory insertion and reductive elimination steps that forge the new carbon-carbon bonds. The precise control over the oxidation state of the rhodium center throughout this cycle ensures that side reactions are minimized, leading to the high purity profiles required for pharmaceutical applications. Understanding this mechanistic pathway is crucial for R&D directors, as it highlights the robustness of the catalyst system and its tolerance to various functional groups present on the substrate molecules.
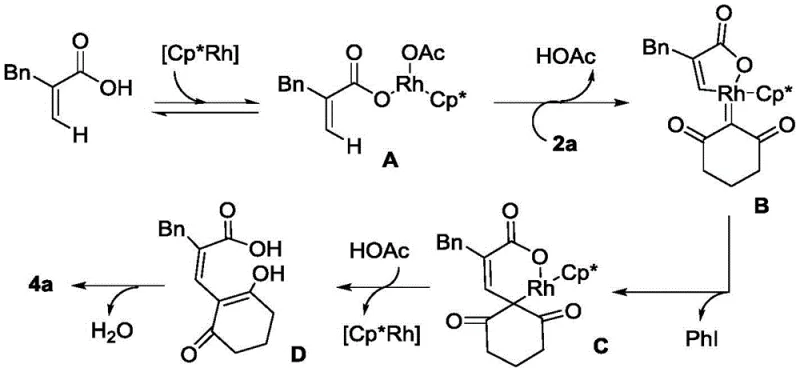
Impurity control is a paramount concern in the synthesis of active pharmaceutical ingredients, and this rhodium-catalyzed method offers distinct advantages in managing byproduct formation. The high regioselectivity of the C-H activation step ensures that the cyclization occurs exclusively at the desired position, preventing the formation of isomeric impurities that are difficult to separate. Additionally, the mild reaction conditions prevent the degradation of sensitive functional groups that might otherwise decompose under harsher thermal or acidic conditions typical of traditional methods. The simplicity of the post-treatment process, which often involves only solvent removal and standard column chromatography, further contributes to the overall purity of the final product. For quality control teams, this means a more consistent impurity profile and reduced risk of encountering genotoxic impurities or heavy metal residues, provided that appropriate catalyst removal protocols are implemented during the workup phase.
How to Synthesize Cyclohexanone Fused Pyrones Efficiently
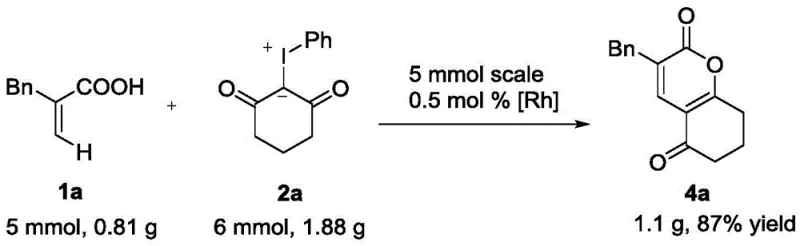
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to the specific reaction parameters outlined in the patent data to ensure optimal results. The process begins with the precise weighing of the alpha,beta-unsaturated carboxylic acid and the cyclic dione-phenyl iodide substrate, maintaining a molar ratio that favors complete conversion of the limiting reagent. The rhodium catalyst, typically generated in situ from [Cp*RhCl2]2 and sodium acetate, is added along with the HFIP solvent, and the mixture is heated to the specified temperature range. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during operation.
- Mix alpha,beta-unsaturated carboxylic acid and cyclic dione-phenyl iodide with Cp*Rh catalyst and sodium acetate in HFIP solvent.
- Heat the reaction mixture to 60-100 degrees Celsius under air atmosphere without inert gas protection for 9 to 12 hours.
- Monitor reaction completion via TLC, remove solvent, and purify the crude solid using column chromatography with petroleum ether and ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic methodology offers substantial benefits that directly impact the bottom line and supply chain resilience for chemical manufacturers. The elimination of inert gas protection and the ability to run reactions in air significantly reduce the capital expenditure required for specialized reactor setups, making the technology accessible for a wider range of production facilities. Moreover, the high yields reported in the patent examples suggest that raw material utilization is highly efficient, minimizing the cost per kilogram of the final intermediate. For procurement managers, this translates into a more predictable cost structure and the potential for significant cost savings when sourcing these complex building blocks at scale. The robustness of the reaction also implies fewer batch failures, ensuring a more reliable supply of critical materials for downstream drug manufacturing processes.
- Cost Reduction in Manufacturing: The process eliminates the need for expensive transition metal scavengers often required in palladium-catalyzed couplings, as the rhodium system is highly efficient at low loadings. By avoiding harsh reagents and multi-step protection strategies, the overall consumption of solvents and auxiliary chemicals is drastically reduced, leading to lower waste disposal costs. The simplified workup procedure further decreases labor hours and processing time, contributing to a leaner manufacturing operation. These qualitative improvements collectively drive down the cost of goods sold, allowing for more competitive pricing in the global market for pharmaceutical intermediates without compromising on quality standards.
- Enhanced Supply Chain Reliability: The starting materials for this reaction, such as alpha,beta-unsaturated carboxylic acids and cyclic diones, are generally commercially available and stable, reducing the risk of raw material shortages. The reaction's tolerance to air and moisture means that storage and handling requirements for intermediates are less stringent, simplifying logistics and warehousing. This robustness ensures that production schedules can be maintained even under less than ideal environmental conditions, providing a buffer against supply chain disruptions. For supply chain heads, this reliability is crucial for maintaining continuous manufacturing flows and meeting tight delivery deadlines for client projects.
- Scalability and Environmental Compliance: The patent demonstrates successful gram-scale experiments with reduced catalyst loading, indicating a clear path towards commercial scale-up of complex heterocycles. The use of HFIP, while a specialized solvent, allows for easy recovery and recycling, aligning with environmental regulations regarding volatile organic compound emissions. The high atom economy of the annulation reaction minimizes the generation of hazardous byproducts, simplifying compliance with increasingly strict environmental protection laws. This scalability ensures that the technology can grow with demand, supporting the transition from clinical trial materials to full commercial production without the need for process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this rhodium-catalyzed technology in industrial settings. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation to provide clarity for decision-makers. Understanding these details helps stakeholders evaluate the feasibility of adopting this method for their specific production needs and regulatory requirements.
Q: Does this rhodium-catalyzed reaction require inert gas protection?
A: No, the patent explicitly states that the reaction can be directly carried out in the air without the protection of inert gas, significantly simplifying operational requirements.
Q: What is the biological activity of the synthesized cyclohexanopyrones?
A: The compounds exhibit good activity in inhibiting various cancer cells, with specific derivatives showing an IC50 of 241 nM against MCF-7 breast cancer cells.
Q: Can this process be scaled up for industrial production?
A: Yes, the patent demonstrates successful gram-scale testing with reduced catalyst loading, indicating strong potential for commercial scale-up of complex intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cyclohexanone Chromenopyrone Supplier
The technical potential of this rhodium-catalyzed route is immense, offering a viable solution for the efficient production of bioactive heterocyclic intermediates used in anticancer drug development. NINGBO INNO PHARMCHEM, as a seasoned CDMO expert, possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that this innovative chemistry can be seamlessly transferred to large-scale manufacturing. Our facility is equipped with stringent purity specifications and rigorous QC labs capable of handling sensitive organometallic reactions, guaranteeing that every batch meets the highest international standards. We understand the critical nature of these intermediates in the drug development timeline and are committed to delivering consistent quality and performance.
We invite you to initiate a conversation about optimizing your supply chain for these valuable compounds by requesting a Customized Cost-Saving Analysis tailored to your specific project requirements. Our technical procurement team is ready to provide specific COA data and route feasibility assessments to help you make informed decisions about your sourcing strategy. By partnering with us, you gain access to not just a supplier, but a strategic ally dedicated to advancing your pharmaceutical projects through superior chemical manufacturing solutions. Contact us today to discuss how we can support your next breakthrough.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
