Advanced Synthesis of C10 High Carbon Sugars for Antiviral Drug Development
Advanced Synthesis of C10 High Carbon Sugars for Antiviral Drug Development
The pharmaceutical industry's relentless pursuit of novel antiviral agents has placed significant emphasis on the development of high-carbon sugar intermediates, particularly those mimicking natural structures like KDO. Patent CN1244588C introduces a groundbreaking methodology for synthesizing C10 high carbon sugars and their derivatives, utilizing a novel oxidative dimerization strategy that fundamentally shifts the paradigm from traditional carbon-chain elongation techniques. This technology leverages the abundant and cost-effective feedstock of alpha-D-xylose, transforming it through a sophisticated sequence of protection, PDC oxidation, and stereospecific reduction. For R&D directors and process chemists, this represents a critical advancement in accessing complex chiral synthons required for next-generation nucleoside analogs with anti-hepatitis B and anti-AIDS activity. The robustness of this pathway, evidenced by consistent high yields across multiple solvent systems, offers a compelling value proposition for commercial scale-up.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of high-carbon sugars containing seven or more consecutive carbon atoms has been fraught with significant chemical and economic challenges. Traditional methodologies predominantly rely on Wittig reactions or similar olefination strategies to extend the carbon chain, processes that are notoriously sensitive to reaction conditions and often require expensive, specialized phosphorus ylides. These conventional routes frequently suffer from poor atom economy, generating substantial stoichiometric waste that complicates downstream purification and increases environmental disposal costs. Furthermore, the starting materials for these Wittig-based approaches are often difficult to source in bulk quantities, creating supply chain bottlenecks that can delay critical drug development timelines. The operational complexity associated with controlling stereochemistry during these chain-elongation steps often results in mixtures of diastereomers, necessitating resource-intensive chromatographic separations that drastically reduce overall process efficiency and final product purity.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN1244588C employs a streamlined oxidative dimerization strategy that directly couples two C5 sugar units to form the desired C10 skeleton. By utilizing 1,2,5-protected alpha-D-xylose as the starting material, the process capitalizes on the inherent chirality of the natural feedstock, ensuring high stereochemical fidelity without the need for extensive resolution steps. The core innovation lies in the use of Pyridinium Dichromate (PDC) to facilitate a unique C5-C5' carbon-carbon bond formation under reflux conditions, effectively doubling the carbon chain length in a single transformative step. 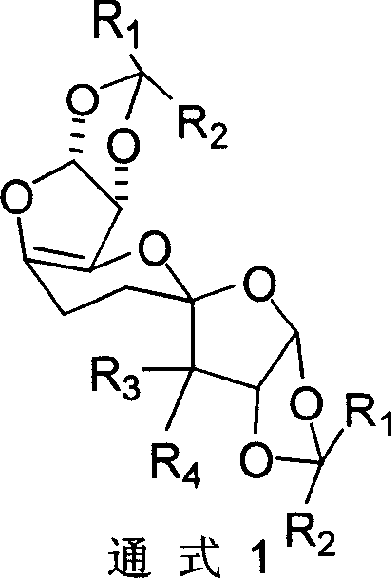 This approach not only simplifies the synthetic route by eliminating the need for complex organometallic reagents but also significantly enhances the overall yield, with experimental data demonstrating conversion rates exceeding 80% in optimized solvent systems. The resulting unsaturated C10 high carbon sugar serves as a versatile platform for further functionalization, enabling the rapid generation of diverse libraries of bioactive molecules.
This approach not only simplifies the synthetic route by eliminating the need for complex organometallic reagents but also significantly enhances the overall yield, with experimental data demonstrating conversion rates exceeding 80% in optimized solvent systems. The resulting unsaturated C10 high carbon sugar serves as a versatile platform for further functionalization, enabling the rapid generation of diverse libraries of bioactive molecules.
Mechanistic Insights into PDC-Catalyzed Oxidative Dimerization
The mechanistic elegance of this synthesis lies in the specific interaction between the protected xylose substrate and the PDC oxidant, which facilitates a radical-mediated or ion-pair coupling mechanism at the C5 position. Under reflux conditions in solvents such as dichloromethane or acetonitrile, the PDC activates the hemiacetal or adjacent hydroxyl equivalents, promoting the formation of a new carbon-carbon bond between two sugar molecules. This oxidative coupling is highly regioselective, preferentially occurring at the C5 position due to the electronic and steric environment created by the 1,2-acetonide and 5-benzoyl protecting groups. The reaction proceeds through a transition state that preserves the stereochemical integrity of the existing chiral centers while establishing the new linkage with high fidelity. Following the dimerization, the resulting unsaturated intermediate undergoes a stereospecific reduction, typically via catalytic hydrogenation, which saturates the double bond and establishes the final configuration of the C10 backbone. 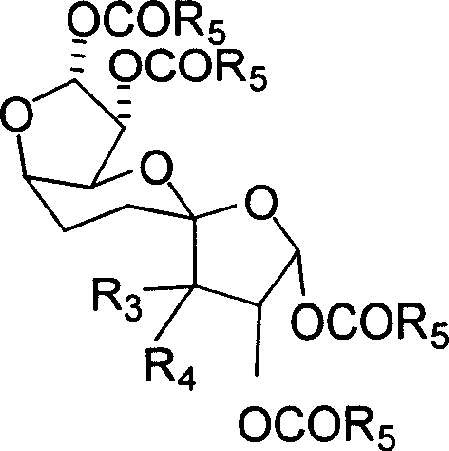 This two-stage sequence ensures that the final products, whether saturated derivatives or acylated variants, maintain the precise three-dimensional architecture required for biological recognition in antiviral applications.
This two-stage sequence ensures that the final products, whether saturated derivatives or acylated variants, maintain the precise three-dimensional architecture required for biological recognition in antiviral applications.
Impurity control is inherently built into this mechanism through the use of robust protecting group strategies and the specificity of the PDC oxidation. The 1,2-O-isopropylidene and 5-O-benzoyl groups not only direct the regioselectivity of the coupling but also protect sensitive hydroxyl functionalities from over-oxidation or side reactions. The subsequent workup procedures, involving washing with saturated sodium bicarbonate and recrystallization from ethyl acetate or ether, effectively remove chromium byproducts and unreacted starting materials. This results in a crude product of high purity, often requiring minimal further purification before proceeding to the next synthetic step. The ability to tune the R3 and R4 substituents through selective hydrolysis and acylation allows for the precise modulation of the molecule's physicochemical properties, facilitating the optimization of solubility and metabolic stability for downstream drug candidates.
How to Synthesize C10 High Carbon Sugar Efficiently
The practical implementation of this synthesis route is designed for scalability and operational simplicity, making it highly attractive for industrial manufacturing environments. The process begins with the preparation of the protected xylose starting material, followed by the critical PDC oxidation step which establishes the C10 framework. Detailed protocols for solvent selection, reaction times, and workup procedures are essential to maximizing yield and minimizing impurity profiles. The subsequent hydrogenation and acylation steps are equally straightforward, utilizing standard equipment and reagents commonly available in fine chemical facilities. For a comprehensive understanding of the specific operational parameters and safety considerations, please refer to the standardized synthesis guide below.
- Oxidative Dimerization: React 1,2,5-protected alpha-D-xylose with PDC in an organic solvent (e.g., dichloromethane) under reflux to form the unsaturated C10 high carbon sugar skeleton.
- Stereospecific Reduction: Dissolve the unsaturated C10 sugar in ethanol and perform catalytic hydrogenation at room temperature and 30KPa to obtain the saturated derivative.
- Acylation: Hydrolyze the saturated compound in acetic acid, then react with acid anhydride and pyridine at room temperature to yield the final acylated C10 sugar derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented synthesis route offers profound advantages in terms of cost structure, supply chain resilience, and environmental compliance. By shifting the raw material base from exotic, low-volume precursors to commodity-grade alpha-D-xylose, manufacturers can achieve significant reductions in direct material costs. The simplified reaction sequence reduces the number of unit operations required, thereby lowering labor costs, energy consumption, and equipment occupancy time. Furthermore, the high yields observed in the key dimerization and reduction steps minimize material loss, contributing to a more efficient overall mass balance. These factors collectively enhance the economic viability of producing high-value antiviral intermediates, allowing for more competitive pricing in the global marketplace.
- Cost Reduction in Manufacturing: The elimination of expensive organometallic reagents and complex purification steps translates directly into lower production costs. By utilizing PDC oxidation instead of Wittig chemistry, the process avoids the generation of phosphine oxide waste, which is costly to dispose of and difficult to separate. The use of common organic solvents like dichloromethane and ethanol further reduces procurement expenses and simplifies solvent recovery operations. Additionally, the high stereocontrol reduces the need for expensive chiral chromatography, streamlining the purification workflow and maximizing throughput.
- Enhanced Supply Chain Reliability: Sourcing alpha-D-xylose, a biomass-derived sugar, ensures a stable and sustainable supply chain that is less susceptible to the geopolitical volatility often associated with petrochemical-derived reagents. The robustness of the reaction conditions allows for flexible manufacturing schedules, as the process is tolerant to minor variations in temperature and pressure. This reliability enables suppliers to maintain consistent inventory levels and meet tight delivery deadlines, reducing the risk of production stoppages for downstream pharmaceutical clients. The modular nature of the synthesis also allows for easy scaling from pilot plant to commercial production without significant process re-engineering.
- Scalability and Environmental Compliance: The process is inherently greener due to its higher atom economy and reduced waste generation compared to traditional methods. The avoidance of heavy metal catalysts in the reduction step (using Pd/C which is recyclable) and the use of stoichiometric oxidants that can be managed effectively align with modern environmental regulations. The simplified workup procedures reduce the volume of aqueous waste streams, lowering wastewater treatment costs. This environmental profile not only ensures regulatory compliance but also enhances the corporate sustainability credentials of the manufacturing entity, a key factor for multinational pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these C10 high carbon sugar derivatives. The answers are derived directly from the experimental data and technical specifications outlined in the patent literature, providing accurate guidance for potential partners and licensees. Understanding these details is crucial for evaluating the feasibility of integrating this technology into existing drug development pipelines.
Q: What are the primary advantages of this C10 sugar synthesis method over traditional Wittig reactions?
A: Unlike traditional Wittig reactions which often require complex operations and difficult-to-obtain raw materials, this patented method utilizes abundant and inexpensive alpha-D-xylose. The process simplifies the formation of the carbon-carbon bond through direct oxidative dimerization, significantly reducing operational complexity and raw material costs while maintaining high stereocontrol.
Q: What is the typical yield range for the key oxidative dimerization step?
A: According to the experimental data in patent CN1244588C, the oxidative dimerization step using PDC in solvents like dichloromethane, acetonitrile, or benzene consistently achieves high yields ranging from approximately 76% to 82.5%, demonstrating robust process reliability for scale-up.
Q: Can this synthetic route be adapted for producing KDO analogs?
A: Yes, the resulting C10 high carbon sugar derivatives serve as critical chiral synthons and key intermediates for synthesizing KDO (3-deoxy-D-manno-2-octulosonic acid) analogs. The method allows for flexible substitution at the 3-position with various amines or ester groups, making it highly suitable for developing new antibiotics and glycoprotein components.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable C10 High Carbon Sugar Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis and contract manufacturing, possessing the technical expertise to translate complex laboratory pathways like the one described in CN1244588C into robust commercial processes. Our team of experienced chemists specializes in the scale-up of carbohydrate chemistry and chiral intermediates, ensuring that the transition from gram-scale research to multi-ton production is seamless and efficient. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, supported by state-of-the-art facilities equipped for handling sensitive oxidation and hydrogenation reactions. Our commitment to quality is unwavering, with stringent purity specifications enforced through rigorous QC labs that utilize advanced analytical techniques to verify identity and potency.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this innovative synthesis route for their antiviral drug programs. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this xylose-based platform. We encourage you to contact our technical procurement team to request specific COA data for our C10 sugar intermediates and to discuss detailed route feasibility assessments tailored to your specific project requirements. Let us help you accelerate your drug development timeline with reliable, high-quality intermediates.
