Revolutionizing Para-Selective Arylation for Scalable Pharmaceutical Intermediate Production
The pharmaceutical and agrochemical industries constantly seek more efficient pathways to construct biaryl motifs, which are ubiquitous in active pharmaceutical ingredients (APIs) such as Losartan, Canagliflozin, and Celecoxib. Patent CN111187130B introduces a groundbreaking methodology for the preparation of para-substituted aryl compounds that addresses long-standing challenges in regioselectivity and step economy. This technology leverages an innovative strategy involving the in-situ construction of aryl sulfonium salts from simple monosubstituted arenes, followed by a palladium-catalyzed Suzuki-Miyaura coupling reaction. By bypassing the need for pre-installed halogen leaving groups or cumbersome directing groups, this approach offers a streamlined route to high-value diaryl structures. The method is characterized by its mild reaction conditions, exceptional para-selectivity, and broad compatibility with diverse functional groups, positioning it as a transformative tool for modern process chemistry.
For R&D directors evaluating new synthetic routes, the ability to control site selectivity in C-H functionalization is often the critical bottleneck. Traditional methods for constructing para-substituted biaryls typically rely on cross-coupling reactions that require the prior introduction of a halogen atom at the specific reaction site. This pre-functionalization step not only adds synthetic complexity and cost but also generates stoichiometric amounts of waste. Furthermore, achieving high para-selectivity on simple alkylbenzenes or alkoxybenzenes without directing groups has historically been difficult, often resulting in mixtures of ortho, meta, and para isomers that are costly to separate. Existing catalytic systems sometimes require large excesses of the arene substrate to drive selectivity, which is impractical for complex or expensive starting materials. These limitations hinder the rapid development of late-stage functionalization strategies essential for drug discovery and optimization pipelines.
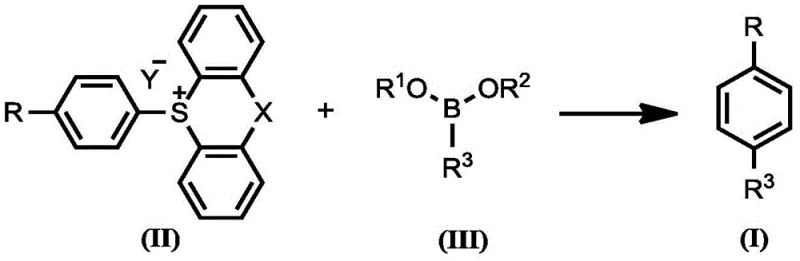
The novel approach disclosed in the patent fundamentally shifts the paradigm by utilizing thianthrene-S-oxide derivatives to generate aryl sulfonium salts with high intrinsic para-selectivity. This electrophilic activation strategy effectively masks the reactivity of the arene, directing the subsequent nucleophilic attack exclusively to the para position. Once the sulfonium salt is formed in situ, it serves as a highly reactive electrophile in a palladium-catalyzed cross-coupling with various boronic acids or esters. This two-step one-pot sequence eliminates the need for isolating unstable intermediates and avoids the use of harsh halogenating agents. The result is a robust protocol that delivers para-substituted products with impressive purity profiles, significantly reducing the burden on downstream purification processes and enabling the synthesis of complex molecules that were previously inaccessible via direct C-H activation.
Mechanistic Insights into Aryl Sulfonium Salt Mediated C-H Activation
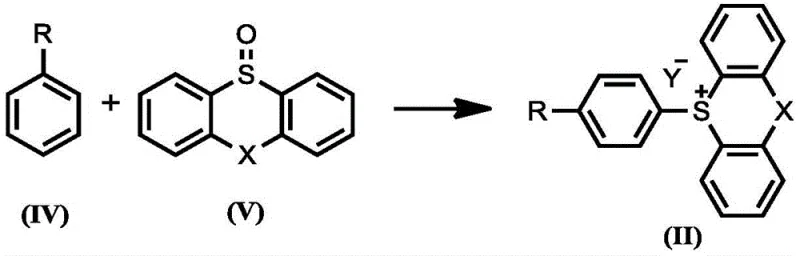
The core of this technology lies in the unique reactivity of the aryl sulfonium salt intermediate. The process begins with the reaction of a monosubstituted arene with a sulfoxide reagent, such as thianthrene-S-oxide, in the presence of an activating agent like trifluoromethanesulfonic anhydride (Tf2O). This step generates a highly electrophilic sulfonium species where the positive charge on the sulfur atom activates the attached aromatic ring towards nucleophilic substitution while simultaneously blocking ortho positions sterically. The electronic properties of the sulfonium group strongly favor attack at the para position relative to the existing substituent on the arene ring. This inherent selectivity is a major advantage over traditional electrophilic aromatic substitution, which often yields mixed isomers. The stability of these sulfonium salts allows them to be generated in situ and carried forward directly into the coupling step without isolation, minimizing handling risks and processing time.
Following the formation of the sulfonium salt, the second phase involves a palladium-catalyzed Suzuki-Miyaura coupling. The sulfonium salt acts as an electrophilic partner, undergoing oxidative addition with the palladium catalyst more readily than traditional aryl halides in certain contexts. The use of specialized ligands, such as tri-tert-butylphosphine, enhances the catalytic activity and stability of the palladium species, facilitating the transmetallation with the boronic acid derivative. The choice of base, such as sodium bicarbonate or potassium carbonate, plays a crucial role in activating the boron species and neutralizing the acidic byproducts generated during the cycle. This mechanistic pathway ensures that the carbon-carbon bond formation occurs efficiently under mild thermal conditions, often at room temperature or slightly elevated temperatures. The tolerance of this system towards sensitive functional groups, including esters, nitriles, and heterocycles, underscores its versatility for synthesizing complex pharmaceutical intermediates without extensive protecting group manipulation.
How to Synthesize Para-Substituted Aryl Compounds Efficiently
Implementing this synthesis route requires careful control of reaction parameters to maximize yield and selectivity. The process generally involves charging the monosubstituted arene and the sulfoxide reagent into a reactor under an inert atmosphere, followed by cooling to low temperatures (e.g., -40°C) before the slow addition of the activating anhydride. After the sulfonium salt is formed, the reaction mixture is warmed, and the coupling partners are introduced along with the catalyst and base. The flexibility of the solvent system, which can include mixtures of dichloromethane, acetone, or DMF, allows for optimization based on the solubility of specific substrates. This adaptability makes the method scalable and suitable for various production environments, ensuring consistent quality across different batches of high-purity intermediates.
- Generate aryl sulfonium salt in situ by reacting monosubstituted arene with thianthrene-S-oxide and triflic anhydride at low temperature.
- Perform Suzuki-Miyaura coupling using the crude sulfonium salt, a boronic acid derivative, palladium catalyst, and base in a suitable solvent.
- Quench the reaction, filter, and purify the resulting para-substituted aryl compound via chromatography or crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this technology offers substantial strategic benefits by simplifying the raw material landscape. Traditional routes to para-substituted biaryls often depend on specialized halogenated starting materials, which can be subject to price volatility and supply constraints due to their multi-step manufacturing requirements. In contrast, this method utilizes simple, commodity-grade monosubstituted arenes such as toluene or anisole derivatives, which are abundantly available and cost-effective. By eliminating the pre-functionalization step, manufacturers can significantly reduce the overall number of unit operations required, leading to lower capital expenditure on equipment and reduced consumption of utilities. The streamlined process flow also minimizes the inventory of hazardous intermediates, enhancing workplace safety and reducing regulatory compliance burdens associated with storing reactive halogenated compounds.
The enhanced supply chain reliability stems from the robustness of the reaction conditions and the broad substrate scope. Because the method tolerates a wide range of functional groups and heterocycles, it reduces the risk of batch failures due to substrate incompatibility, a common issue in complex molecule synthesis. This reliability translates into more predictable lead times for delivering critical intermediates to downstream API manufacturers. Furthermore, the high selectivity of the reaction minimizes the formation of difficult-to-remove isomeric impurities, which often necessitate expensive and time-consuming chromatographic separations. By producing cleaner crude products, the technology facilitates simpler workup procedures, such as crystallization or filtration, thereby accelerating the overall production timeline and improving throughput capacity without compromising on stringent purity specifications required by global regulatory agencies.
Scalability and environmental compliance are also key drivers for adopting this methodology. The reaction operates under relatively mild conditions, avoiding the extreme temperatures or pressures that can pose engineering challenges during scale-up. The use of common organic solvents and inorganic bases simplifies waste management and solvent recovery processes. Additionally, the atom economy is improved by avoiding the generation of stoichiometric halide waste associated with traditional cross-coupling precursors. This alignment with green chemistry principles supports corporate sustainability goals and helps manufacturers meet increasingly strict environmental regulations. The ability to synthesize complex drug molecules like Ibuprofen and Bifonazole derivatives efficiently demonstrates the practical viability of this approach for commercial-scale manufacturing, offering a competitive edge in the production of high-value fine chemicals.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis method is crucial for stakeholders evaluating its integration into existing workflows. The following questions address common inquiries regarding the operational feasibility and scope of the technology. These insights are derived directly from the experimental data and embodiments provided in the patent documentation, ensuring that the information reflects the actual capabilities and limitations of the described chemical process.
Q: What is the primary advantage of this para-arylation method over traditional cross-coupling?
A: The primary advantage is the ability to achieve high para-selectivity directly from simple monosubstituted arenes without requiring pre-functionalization with halogens or directing groups, significantly simplifying the synthetic route.
Q: Does this method tolerate heterocyclic substrates?
A: Yes, the patent data demonstrates wide tolerance for heterocyclic coupling substrates, making it highly suitable for synthesizing complex pharmaceutical intermediates containing nitrogen, oxygen, or sulfur heterocycles.
Q: What are the typical reaction conditions for the coupling step?
A: The coupling typically proceeds under mild conditions, often at room temperature or slightly elevated temperatures (e.g., 50°C), using common solvents like acetone, DMF, or DCM mixtures with bases such as sodium bicarbonate.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Para-Substituted Aryl Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced C-H activation technologies in reshaping the landscape of pharmaceutical intermediate manufacturing. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. We are committed to delivering high-purity para-substituted aryl compounds that meet the rigorous quality standards demanded by the global pharmaceutical industry. Our state-of-the-art facilities are equipped with rigorous QC labs capable of performing detailed impurity profiling and structural confirmation, guaranteeing that every batch adheres to stringent purity specifications.
We invite you to collaborate with us to leverage this cutting-edge synthesis technology for your next project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific molecule, exploring how this streamlined route can optimize your supply chain. We encourage you to reach out for specific COA data and route feasibility assessments to determine the best path forward for your commercial needs. Let us help you accelerate your development timeline and achieve superior economic efficiency through our advanced manufacturing capabilities.
