Advanced N-Phosphonimide Reagents Enabling High-Enantioselectivity Strecker Synthesis for Pharmaceutical Intermediates
The landscape of asymmetric synthesis for chiral amino acids and their derivatives is undergoing a significant transformation driven by the innovations disclosed in patent CN102933589A. This pivotal intellectual property introduces a comprehensive suite of novel Strecker reagents, specifically focusing on electrophiles such as N-phosphoryl azides, N-phosphoramides, and N-phosphonimides, alongside their various derivatives. For R&D directors and process chemists, the significance lies in the ability to access high-purity chiral building blocks with unprecedented efficiency. The patent details a robust methodology where achiral diamines are converted into sophisticated phosphorus-containing electrophiles, which then participate in asymmetric Strecker reactions with exceptional stereocontrol. This technology addresses the longstanding challenge of balancing high enantioselectivity with practical synthetic feasibility, offering a pathway to produce complex pharmaceutical intermediates that were previously difficult to access with such high optical purity.
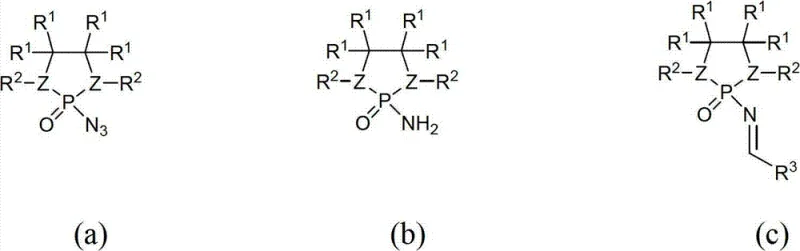
Traditional methods for synthesizing alpha-aminonitriles often rely on imine electrophiles that require harsh conditions or suffer from poor stereochemical outcomes, leading to costly downstream separation processes. Conventional N-protected imines frequently necessitate the use of expensive transition metal catalysts or stoichiometric chiral auxiliaries that generate substantial waste. Furthermore, the removal of protecting groups in older methodologies can be problematic, often requiring strong acids or bases that compromise the integrity of sensitive functional groups within the molecule. In contrast, the novel approach outlined in the patent utilizes N-phosphonimides which act as highly activated electrophiles. These reagents are synthesized through a streamlined sequence involving the reaction of diamines with phosphorus oxychloride followed by azide substitution and subsequent condensation. This new design not only enhances the reactivity of the imine carbon towards cyanide nucleophiles but also provides a steric environment that is crucial for inducing high levels of asymmetry during the bond-forming event.
The mechanistic insights into this improved Strecker reaction reveal a sophisticated interplay between the phosphorus-based electrophile and the aluminum-based nucleophile. The core of the innovation involves the generation of chiral aluminum-cyanide complexes in situ, derived from readily available chiral ligands such as amino acids, amino alcohols, or binaphthols. When these nucleophiles attack the N-phosphonimide, the bulky phosphoryl group directs the approach of the cyanide ion, ensuring that the addition occurs from a specific face of the planar imine bond. This steric guidance is further reinforced by the coordination of the aluminum center to the phosphoryl oxygen, creating a rigid transition state that locks the stereochemistry. The result is a reaction pathway that minimizes the formation of unwanted racemic byproducts. Additionally, the patent highlights that the N-CH2-naphthyl protecting group on the phosphorus moiety facilitates purification, as the resulting products can often be isolated simply by washing with hexane, a feature that drastically reduces solvent consumption and processing time compared to column chromatography.

How to Synthesize N-Phosphono Substituted Chiral Alpha-Aminonitriles Efficiently
The synthesis of these high-value intermediates follows a logical progression starting from simple diamine precursors. The initial step involves the conversion of the diamine into a phosphorochloridate using phosphorus oxychloride and triethylamine, typically in benzene or dichloromethane under reflux conditions to ensure quantitative conversion. This intermediate is then transformed into a phosphorazide using sodium azide, which is subsequently reduced to the corresponding phosphoramide via catalytic hydrogenation over palladium on carbon. The final activation step involves condensing this phosphoramide with an aldehyde in the presence of titanium(IV) chloride to generate the reactive N-phosphonimide. For the asymmetric Strecker reaction itself, the protocol requires the preparation of a chiral aluminum-cyanide species, often formed by mixing diethylaluminum cyanide with a chiral ligand like (S)-phenylglycine in toluene at low temperatures. The detailed standardized synthesis steps for executing this transformation are provided below.
- Preparation of Achiral N-Phosphorylazides: React achiral diamine with phosphorus oxychloride in the presence of triethylamine, followed by nucleophilic substitution with sodium azide to form the phosphorazide precursor.
- Synthesis of N-Phosphonimide Electrophile: Reduce the phosphorazide to phosphoramide via catalytic hydrogenation, then condense with an aldehyde using titanium(IV) chloride to generate the active N-phosphonimide.
- Asymmetric Strecker Reaction: Combine the N-phosphonimide with a chiral aluminum-cyanide complex (derived from amino acids or diols) at -78°C in toluene to yield the chiral alpha-aminonitrile with high enantioselectivity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology offers tangible benefits regarding cost structure and operational reliability. The primary advantage stems from the significant simplification of the purification workflow. Because the novel protecting groups allow for product isolation via simple hexane washing rather than extensive chromatographic separation, the consumption of silica gel and high-purity solvents is drastically reduced. This directly translates to lower material costs and reduced hazardous waste disposal fees, contributing to a more sustainable and economically viable manufacturing process. Furthermore, the use of non-toxic and inexpensive diethylaluminum cyanide as the cyanide source eliminates the need for more hazardous or costly cyanating agents, enhancing workplace safety and reducing regulatory compliance burdens associated with handling toxic reagents.
- Cost Reduction in Manufacturing: The elimination of complex purification steps and the use of recoverable ligands lead to substantial cost savings. The patent explicitly notes that the N,N'-bis(naphthalen-1-methyl)ethane-1,2-diamine ligand can be quantitatively recovered under mild conditions, allowing for its reuse in subsequent batches. This closed-loop usage of chiral ligands significantly lowers the cost of goods sold (COGS) for high-value chiral intermediates, making the process economically attractive for large-scale production without compromising on quality.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable starting materials such as diamines, aldehydes, and diethylaluminum cyanide ensures a robust supply chain. Unlike processes dependent on exotic or custom-synthesized catalysts that may have long lead times, the reagents for this Strecker variation are accessible from multiple global suppliers. This diversification of the supply base mitigates the risk of production delays caused by raw material shortages, ensuring consistent delivery schedules for downstream pharmaceutical customers who rely on just-in-time inventory models.
- Scalability and Environmental Compliance: The reaction conditions, while requiring low temperatures (-78°C), are standard in industrial cryogenic reactors and do not necessitate specialized high-pressure equipment. The high yields (>97%) and enantioselectivities (>99% ee) mean that less raw material is wasted to produce the same amount of active pharmaceutical ingredient (API) intermediate. This atom economy, combined with the ability to recycle ligands and simplify workups, aligns well with green chemistry principles, facilitating easier environmental permitting and reducing the overall carbon footprint of the manufacturing facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented Strecker reaction technology. These answers are derived directly from the experimental data and specifications provided in the patent documentation to ensure accuracy and relevance for potential partners. Understanding these details is crucial for evaluating the feasibility of integrating this chemistry into existing production lines or new drug development pipelines.
Q: What are the primary advantages of using N-phosphonimides in Strecker reactions compared to traditional imines?
A: N-phosphonimides offer superior control over stereochemistry and chemoselectivity. The phosphoryl group acts as a robust protecting group that can be easily cleaved under mild conditions, allowing for quantitative recovery of the diamine ligand and simplifying the purification process significantly compared to traditional N-protected imines.
Q: What level of enantioselectivity can be achieved with this novel catalytic system?
A: The patented asymmetric catalytic Strecker reaction utilizing these novel chiral N-phosphonimides has demonstrated excellent enantioselectivities, achieving up to greater than 99% ee (enantiomeric excess) alongside high chemical yields exceeding 97%, making it highly suitable for producing single-enantiomer pharmaceutical ingredients.
Q: Is the purification process scalable for industrial manufacturing?
A: Yes, the process is designed for scalability. A key feature is the use of an N-CH2-naphthyl protecting group which allows for simple purification of the product merely by washing the semi-finished product with hexane, eliminating the need for complex chromatography steps often required in fine chemical manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Phosphonimide Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating advanced academic research into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high enantioselectivity observed in the lab is maintained at the plant scale. Our facility is equipped with stringent purity specifications and rigorous QC labs capable of verifying the >99% ee targets set by this technology. We understand that consistency is key for pharmaceutical clients, and our robust quality management systems guarantee that every batch of N-phosphonimide reagents meets the highest international standards for impurity profiles and optical purity.
We invite you to collaborate with us to leverage this cutting-edge chemistry for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and target molecules. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. By partnering with us, you gain access not just to a chemical supplier, but to a strategic partner dedicated to optimizing your supply chain for cost, quality, and speed in the competitive pharmaceutical intermediates market.
