Advanced Dual-Catalytic Synthesis of Cyano-Substituted Enamines for Commercial Pharmaceutical Manufacturing
Advanced Dual-Catalytic Synthesis of Cyano-Substituted Enamines for Commercial Pharmaceutical Manufacturing
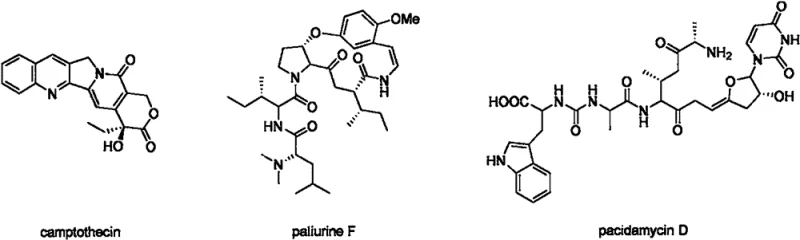
The landscape of organic synthesis is constantly evolving to meet the rigorous demands of modern drug discovery, particularly in the construction of complex nitrogen-containing scaffolds. Patent CN115490614A introduces a groundbreaking methodology for the synthesis of cyano-substituted enamines, a structural motif prevalent in numerous bioactive natural products and pharmaceutical agents. As illustrated in the structural diversity of compounds like camptothecin and pacidamycin D, the enamine fragment serves as a pivotal pharmacophore in many therapeutic classes. This novel approach leverages a sophisticated dual-catalytic system involving ruthenium and nickel complexes under visible light irradiation, addressing long-standing challenges in selectivity and efficiency. By enabling the direct coupling of enamines with cyclic ketoximes, this technology provides a robust pathway for generating high-value intermediates that were previously difficult to access with high fidelity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the functionalization of enamines has relied heavily on beta-hydrogen activation strategies, which often suffer from significant limitations regarding regioselectivity and functional group tolerance. Conventional thermal methods frequently require elevated temperatures and stoichiometric amounts of harsh oxidants or strong bases, leading to the degradation of sensitive moieties and the formation of complex impurity profiles that are costly to remove. Furthermore, the specific installation of a terminal cyano group onto an enamine scaffold has remained a formidable challenge, with existing protocols often yielding mixtures of geometric isomers or suffering from low conversion rates. These inefficiencies create substantial bottlenecks in the supply chain for pharmaceutical intermediates, where purity and consistency are paramount for regulatory approval. The reliance on precious metal catalysts in traditional cross-coupling reactions also exacerbates cost concerns, necessitating extensive purification steps to reduce residual metal content to acceptable parts-per-million levels.
The Novel Approach
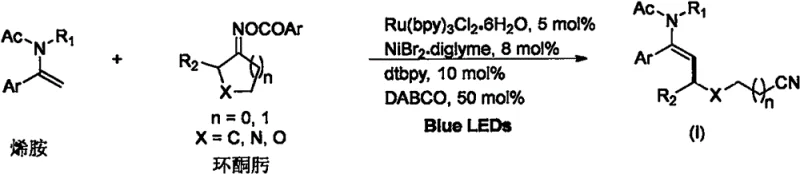
In stark contrast to these legacy methods, the technology disclosed in CN115490614A utilizes a synergistic photoredox and nickel catalysis cycle driven by benign visible light energy. This innovative strategy employs a dual-catalyst system comprising tris(2,2'-bipyridyl)ruthenium(II) chloride hexahydrate and a nickel bromide diglyme complex, facilitated by a bulky bipyridine ligand to control steric environment. The reaction proceeds under exceptionally mild conditions, specifically at room temperature in N,N-dimethylacetamide (DMAc) solvent, thereby preserving the integrity of delicate functional groups present on the substrate. As depicted in the general reaction scheme, this method achieves high efficiency and excellent E-selectivity, directly converting readily available enamines and cyclic ketoximes into the desired cyano-substituted products. This paradigm shift not only simplifies the operational complexity but also significantly enhances the safety profile of the manufacturing process by eliminating the need for hazardous thermal inputs.
Mechanistic Insights into Ru/Ni Dual-Catalyzed Photoredox Coupling
The mechanistic elegance of this transformation lies in the intricate interplay between the photoredox cycle and the nickel catalytic cycle, which work in concert to activate the inert bonds of the starting materials. Upon irradiation with 465 nm blue LEDs, the ruthenium photocatalyst enters an excited state capable of engaging in single-electron transfer (SET) processes with the cyclic ketoxime ester. This interaction generates a reactive iminyl radical species alongside a cyanoalkyl radical fragment, which is subsequently captured by the nickel center. The nickel catalyst, stabilized by the dtbpy ligand, undergoes oxidative addition with the enamine substrate, forming a key organometallic intermediate that facilitates the carbon-carbon bond formation. This radical-polar crossover mechanism ensures that the reaction proceeds with high chemoselectivity, avoiding the uncontrolled polymerization or decomposition pathways often observed in free-radical chemistry. The precise tuning of the redox potentials allows for the regeneration of the active catalytic species, sustaining the cycle with minimal catalyst loading.
From an impurity control perspective, the high stereoselectivity for the E-isomer is a critical advantage that streamlines downstream processing. The steric bulk of the dtbpy ligand plays a decisive role in directing the geometry of the final double bond, effectively suppressing the formation of the Z-isomer which would otherwise constitute a difficult-to-remove impurity. Additionally, the mild reaction conditions prevent the hydrolysis of the nitrile group or the isomerization of the enamine double bond, common side reactions in acidic or basic thermal environments. The use of DABCO as a mild organic base further contributes to a clean reaction profile by neutralizing acidic byproducts without promoting elimination reactions. For a reliable pharmaceutical intermediate supplier, this level of control translates directly into higher yields and reduced waste, ensuring that the final product meets the stringent purity specifications required for clinical applications without the need for recrystallization or chiral separation.
How to Synthesize Cyano-Substituted Enamines Efficiently
The practical implementation of this synthesis protocol is designed for ease of execution while maintaining rigorous control over reaction parameters to ensure reproducibility. The process begins with the careful assembly of the catalytic system in a sealed vessel, where the precise molar ratios of the ruthenium and nickel catalysts are critical for maximizing turnover numbers. Operators must ensure that the reaction environment is properly degassed to prevent oxygen quenching of the excited photocatalyst, although the system demonstrates remarkable robustness. Following the reaction period under controlled LED irradiation, the workup procedure involves simple solvent evaporation followed by standard silica gel chromatography, utilizing a petroleum ether and ethyl acetate gradient to isolate the pure product. This straightforward isolation protocol underscores the practicality of the method for both laboratory-scale optimization and larger-scale production campaigns.
- Prepare the reaction vessel by adding the photocatalyst Ru(bpy)3Cl2·6H2O (5 mol%), nickel catalyst NiBr2·diglyme (8 mol%), ligand dtbpy (10 mol%), and base DABCO (50 mol%) along with the cyclic ketoxime substrate.
- Purge the sealed reaction tube with argon gas three times to ensure an inert atmosphere, then add the enamine substrate and DMAc solvent under stirring.
- Irradiate the reaction mixture with 465 nm blue LEDs at room temperature for 12 hours, followed by solvent removal and silica gel column purification to isolate the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this photoredox-mediated synthesis offers transformative benefits that extend beyond mere chemical novelty. The ability to synthesize complex cyano-enamine scaffolds under ambient conditions drastically reduces the energy consumption associated with heating and cooling cycles, contributing to a lower overall carbon footprint for the manufacturing facility. Moreover, the use of earth-abundant nickel in conjunction with a recyclable ruthenium photocatalyst mitigates the supply risk and cost volatility associated with exclusive reliance on scarce precious metals like palladium or rhodium. This diversification of the catalyst portfolio enhances supply chain resilience, ensuring that production schedules are not disrupted by fluctuations in the global market for rare earth elements. The high functional group tolerance means that a single standardized protocol can be applied to a wide array of substrates, reducing the need for custom process development for each new derivative.
- Cost Reduction in Manufacturing: The elimination of extreme thermal conditions and the use of catalytic rather than stoichiometric reagents leads to substantial cost savings in utility consumption and raw material usage. By avoiding the need for cryogenic cooling or high-pressure reactors, capital expenditure for specialized equipment is minimized, allowing for the utilization of standard glass-lined reactors. The high selectivity of the reaction reduces the burden on purification teams, lowering the costs associated with solvent usage and waste disposal during chromatography. Furthermore, the improved atom economy of coupling enamines directly with ketoximes minimizes the generation of stoichiometric salt waste, aligning with green chemistry principles that are increasingly mandated by regulatory bodies.
- Enhanced Supply Chain Reliability: The starting materials, including various substituted enamines and cyclic ketoximes, are commercially available or easily synthesized from commodity chemicals, ensuring a stable and continuous supply of feedstock. The robustness of the catalytic system against moisture and air exposure simplifies the logistical requirements for raw material storage and handling, reducing the risk of spoilage or degradation prior to use. This reliability allows for more accurate forecasting and inventory management, crucial for maintaining just-in-time delivery schedules for downstream API manufacturers. The scalability of the photochemical process, potentially adaptable to continuous flow technology, further secures the supply chain by enabling rapid ramp-up of production volumes to meet surging demand without compromising quality.
- Scalability and Environmental Compliance: Operating at room temperature in polar aprotic solvents like DMAc simplifies the engineering controls required for scale-up, as there is no risk of thermal runaway associated with exothermic thermal reactions. The process generates minimal hazardous waste, primarily consisting of spent catalyst residues that can be sequestered and treated according to standard heavy metal waste protocols. This environmental compatibility facilitates easier permitting and compliance with increasingly strict environmental regulations in major pharmaceutical manufacturing hubs. The potential for solvent recycling and catalyst recovery further enhances the sustainability profile of the process, making it an attractive option for companies aiming to achieve carbon neutrality goals in their operations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this dual-catalytic synthesis platform. These insights are derived directly from the experimental data and scope evaluation presented in the patent documentation, providing a realistic overview of the technology's capabilities. Understanding these nuances is essential for R&D teams evaluating the feasibility of integrating this route into their existing process pipelines. The answers reflect the balance between high-performance chemistry and practical manufacturing constraints.
Q: What are the key advantages of this dual-catalytic method over traditional enamine functionalization?
A: This method utilizes a synergistic Ru/Ni photoredox system that operates under mild room temperature conditions with visible light, avoiding the harsh thermal conditions and poor selectivity often associated with conventional beta-H functionalization strategies.
Q: Is this synthetic route scalable for industrial production of pharmaceutical intermediates?
A: Yes, the process employs commercially available catalysts and operates at room temperature in standard solvents like DMAc, making it highly amenable to scale-up in flow reactors or large batch vessels without requiring specialized high-pressure equipment.
Q: What is the stereochemical outcome of the cyano-substituted enamine products?
A: The reaction demonstrates high stereoselectivity, consistently yielding the E-configuration isomer of the cyano-substituted enamine, which is critical for downstream biological activity and further synthetic transformations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cyano-Substituted Enamines Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of adopting cutting-edge synthetic methodologies to maintain a competitive edge in the global pharmaceutical market. Our team of expert chemists has thoroughly evaluated the dual-catalytic protocol described in CN115490614A and confirmed its viability for the commercial scale-up of complex pharmaceutical intermediates. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to industrial manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with advanced photochemical reactors and rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of cyano-substituted enamine delivered meets the highest international standards.
We invite you to collaborate with us to leverage this innovative technology for your next drug development project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline constraints. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions based on concrete performance metrics. Let us help you accelerate your pipeline with high-quality intermediates produced through sustainable and economically advantageous processes.
