Advanced Catalytic Synthesis of 5,6-Dihydrobenzofuranone Derivatives for Commercial Pharmaceutical Applications
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to access bioactive scaffolds, particularly those found in natural products with significant therapeutic potential. Patent CN115043799A introduces a groundbreaking methodology for the preparation of 5,6-dihydrobenzofuranone derivatives, a core structural motif prevalent in biologically active molecules such as Atractylenolide I. This sesquiterpenoid compound is renowned for its anti-inflammatory and anti-tumor properties, as well as its ability to regulate gastrointestinal function. The disclosed technology represents a paradigm shift from traditional multi-step syntheses to a direct, catalytic condensation strategy. By leveraging alpha-keto acid compounds and aliphatic ketones as primary building blocks, this invention achieves high atom economy and operational simplicity. The core transformation involves a Lewis acid-catalyzed cyclization that constructs the fused heterocyclic system in a single pot, drastically reducing the number of unit operations required compared to legacy processes.
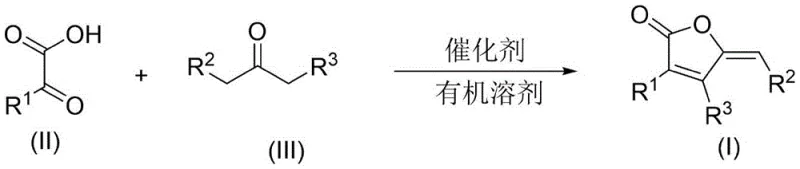
For R&D directors evaluating process feasibility, the mechanistic elegance of this route offers substantial benefits regarding impurity control and yield optimization. The reaction proceeds through a coordinated activation of the carbonyl species, facilitating a smooth cascade of nucleophilic attacks and dehydration steps. This direct approach minimizes the formation of intermediate by-products that typically plague stepwise constructions of fused ring systems. Furthermore, the broad substrate scope allows for the rapid generation of diverse analog libraries, enabling medicinal chemists to explore structure-activity relationships (SAR) with unprecedented speed. The compatibility with various aromatic and heteroaromatic substituents ensures that this platform technology can be adapted for a wide array of drug discovery programs targeting inflammation, cancer, and metabolic disorders.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 5,6-dihydrobenzofuranone skeleton has been a challenging endeavor in organic synthesis, often relying on convoluted pathways that hinder commercial viability. Conventional strategies frequently employ cyclic aliphatic ketones reacting with specialized reagents such as 2-chloro-2-methoxyacetic acid methyl ester or 1,1-dimethoxyacetone. A critical bottleneck in these traditional methods is the availability of these key reagents; they are not commodity chemicals but rather require their own multi-step synthesis from basic feedstocks. This dependency introduces significant supply chain vulnerabilities and inflates the cost of goods sold (COGS). Additionally, these older routes are often plagued by low overall yields due to the accumulation of losses at each synthetic step. The use of halogenated intermediates also raises environmental concerns regarding waste disposal and the need for rigorous purification to remove toxic residues, which complicates the regulatory approval process for final drug substances.
The Novel Approach
In stark contrast, the methodology described in patent CN115043799A dismantles these barriers by utilizing commercially abundant alpha-keto acids and simple aliphatic ketones as starting materials. This strategic pivot eliminates the need for pre-functionalized, hard-to-source reagents, thereby streamlining the raw material procurement process. The reaction is catalyzed by inexpensive Lewis acids, such as boron trifluoride etherate, which are widely available in bulk quantities. The operational simplicity is further enhanced by the fact that the reaction can be conducted under air conditions without the need for stringent exclusion of oxygen or moisture, a feature that significantly reduces capital expenditure on specialized reactor infrastructure. This robustness translates directly into a more reliable manufacturing process with reduced risk of batch failures, ensuring consistent supply for downstream pharmaceutical applications.
Mechanistic Insights into BF3-Catalyzed Cyclization
The success of this synthetic route hinges on the precise activation of the carbonyl electrophile by the Lewis acid catalyst. In the presence of BF3·Et2O, the alpha-keto acid undergoes coordination at the carbonyl oxygen, increasing the electrophilicity of the adjacent carbon center. This activation facilitates the nucleophilic attack by the enol form of the aliphatic ketone, which is generated in situ under the acidic conditions. The resulting intermediate undergoes a series of proton transfers and dehydration events, ultimately leading to the formation of the furanone ring fused to the carbocyclic framework. This mechanism is highly efficient because it avoids the formation of stable off-cycle intermediates that could stall the reaction progress. The choice of solvent, typically toluene or similar aromatic hydrocarbons, plays a crucial role in solubilizing the organic substrates while maintaining the activity of the catalyst at elevated temperatures around 70°C.
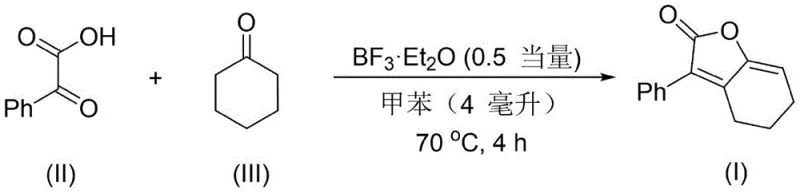
From an impurity control perspective, the mechanism favors the desired cyclization pathway over competing side reactions such as polymerization or oligomerization of the ketone. The specific stoichiometry, often employing a slight excess of the ketone (1:2 molar ratio), drives the equilibrium towards product formation while suppressing self-condensation of the alpha-keto acid. The mild acidity of the catalyst system ensures that sensitive functional groups on the aromatic ring, such as halogens or alkyl substituents, remain intact throughout the process. This chemoselectivity is paramount for producing high-purity intermediates that meet the stringent specifications required for active pharmaceutical ingredients (APIs). The final purification via standard silica gel chromatography effectively removes any trace catalyst or unreacted starting materials, yielding a product with a clean impurity profile suitable for subsequent biological testing or formulation.
How to Synthesize 5,6-Dihydrobenzofuranone Derivatives Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific operational parameters to maximize yield and safety. The process begins with the careful selection of high-purity starting materials to minimize the introduction of foreign impurities. The reaction is typically performed in a sealed vessel to prevent solvent loss at the operating temperature of 70°C. Monitoring the reaction progress via TLC or HPLC is recommended to determine the optimal endpoint, although the patent indicates that 4 hours is generally sufficient for complete conversion. Upon completion, the workup procedure involves a straightforward liquid-liquid extraction using ethyl acetate and brine, followed by drying and concentration. The crude product is then purified using column chromatography with a petroleum ether and ethyl acetate gradient, a standard technique familiar to most process chemistry teams. For detailed standardized operating procedures and safety guidelines, please refer to the technical steps outlined below.
- Charge a reaction vessel with an organic solvent such as toluene, followed by the sequential addition of the alpha-keto acid compound and the aliphatic ketone compound.
- Add the Lewis acid catalyst, specifically Boron Trifluoride Etherate (BF3·Et2O), maintaining a molar ratio of approximately 1: 0.5 relative to the acid substrate.
- Heat the reaction mixture to 70°C and stir for 4 hours under sealed conditions, followed by standard aqueous workup and silica gel column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers compelling economic and logistical advantages that extend beyond simple yield improvements. The primary driver of cost reduction lies in the raw material strategy; by shifting from specialized, multi-step reagents to commodity alpha-keto acids and ketones, the volatility of raw material pricing is significantly mitigated. These starting materials are produced on a massive global scale for various industries, ensuring a stable and competitive supply market. Furthermore, the elimination of complex precursor synthesis steps reduces the overall manufacturing lead time, allowing for faster response to market demand fluctuations. The simplified process flow also means fewer reactors and less manpower are required per kilogram of output, directly lowering the conversion costs associated with production.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound due to the drastic simplification of the synthetic sequence. By removing the need to synthesize expensive reagents like 2-chloro-2-methoxyacetic acid methyl ester in-house or purchase them at a premium, the direct material costs are substantially lowered. Additionally, the use of cheap Lewis acid catalysts instead of precious metal complexes further decreases the catalyst cost burden. The high atom economy of the reaction means that a greater proportion of the raw material mass ends up in the final product, reducing waste disposal costs and maximizing the value derived from every kilogram of input. This efficiency translates into a more competitive pricing structure for the final API intermediate, enhancing the margin potential for downstream drug manufacturers.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the reliance on widely available commodity chemicals. Unlike niche reagents that may have single-source suppliers or long lead times, alpha-keto acids and cyclic ketones are stocked by numerous global chemical distributors. This diversity of supply sources reduces the risk of production stoppages due to raw material shortages. Moreover, the robustness of the reaction conditions, which do not require cryogenic temperatures or ultra-high vacuum, means that the process can be easily transferred between different manufacturing sites or contract manufacturing organizations (CMOs) without extensive re-validation. This flexibility ensures continuity of supply even in the face of regional disruptions or geopolitical instability.
- Scalability and Environmental Compliance: Scaling this process from gram to tonnage levels is straightforward due to the absence of hazardous reagents and extreme conditions. The reaction exotherm is manageable, and the use of common solvents like toluene simplifies solvent recovery and recycling protocols. From an environmental standpoint, the high atom economy and reduced waste generation align perfectly with green chemistry principles, helping companies meet increasingly stringent regulatory requirements regarding carbon footprint and waste discharge. The avoidance of halogenated intermediates in the main synthetic path also simplifies the treatment of effluent streams, reducing the environmental compliance burden and associated costs for wastewater treatment facilities.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the adoption of this technology, we have compiled answers to common questions regarding the process specifics and its applicability to various drug development stages. These insights are derived directly from the experimental data and technical disclosures within the patent literature, ensuring accuracy and relevance for process development scientists. Understanding these nuances is critical for making informed decisions about route selection and resource allocation in your R&D pipeline.
Q: What are the primary advantages of this new synthetic route over conventional methods?
A: Unlike traditional methods requiring multi-step preparation of specialized reagents like 2-chloro-2-methoxyacetic acid methyl ester, this novel approach utilizes readily available alpha-keto acids and aliphatic ketones directly. It eliminates complex precursor synthesis, significantly improving atom economy and reducing overall process complexity.
Q: Is the catalyst system compatible with large-scale industrial production?
A: Yes, the process utilizes inexpensive and commercially abundant Lewis acid catalysts such as BF3·Et2O. The reaction operates under mild conditions (70°C) and is insensitive to oxygen, making it highly suitable for scale-up without requiring specialized inert atmosphere equipment for every step.
Q: What is the substrate scope for this 5,6-dihydrobenzofuranone synthesis?
A: The method demonstrates excellent functional group tolerance. It accommodates various R1 groups including phenyl, substituted phenyl, naphthyl, and thienyl moieties. Furthermore, it works effectively with both cyclic ketones like cyclohexanone and acyclic ketones like acetone and 3-pentanone.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5,6-Dihydrobenzofuranone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this patented synthetic route for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess the technical expertise and infrastructure to rapidly translate this laboratory-scale innovation into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We maintain stringent purity specifications across all our manufacturing lines, supported by rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify the identity and quality of every batch. Our commitment to excellence ensures that the 5,6-dihydrobenzofuranone derivatives produced meet the highest standards required for clinical and commercial applications.
We invite you to collaborate with us to leverage this advanced technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact us to request specific COA data for our reference standards and comprehensive route feasibility assessments. By partnering with NINGBO INNO PHARMCHEM, you gain access to a reliable supply chain partner dedicated to driving efficiency and innovation in your pharmaceutical manufacturing operations.
