Advanced Nickel-Catalyzed Synthesis of Trimetazidine for Scalable Pharmaceutical Production
Advanced Nickel-Catalyzed Synthesis of Trimetazidine for Scalable Pharmaceutical Production
The pharmaceutical industry continuously seeks robust, cost-effective, and safe manufacturing routes for critical active pharmaceutical ingredients (APIs) and their intermediates. Patent CN101575321A, filed in 2009, presents a transformative approach to the production of Trimetazidine, a potent antianginal agent widely used to treat ischemic heart disease. This patent discloses a novel catalytic reductive amination process that utilizes 2,3,4-trimethoxybenzaldehyde and piperazine as primary starting materials, mediated by a cost-efficient nickel-type catalyst under hydrogen pressure. Unlike traditional methods that rely on expensive precious metals or hazardous stoichiometric reducing agents, this innovation leverages the unique activity of nickel catalysts to achieve high conversion rates and exceptional selectivity. For R&D directors and procurement managers, this technology represents a significant opportunity to optimize the supply chain for high-purity pharmaceutical intermediates while drastically reducing the environmental footprint and operational hazards associated with legacy synthesis routes.
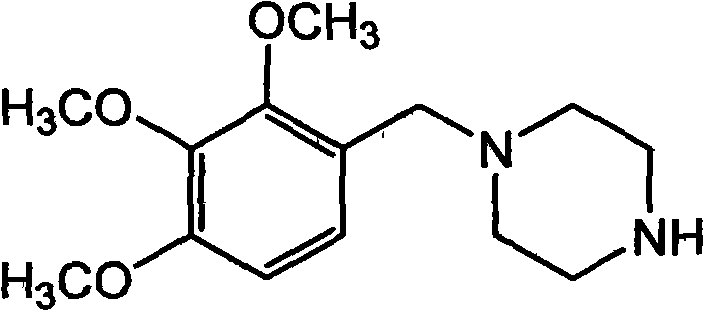
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Trimetazidine has been plagued by significant technical and economic inefficiencies that hinder large-scale commercial viability. Early methodologies, such as those described in FR 2493316, involved the reaction of 2,3,4-trimethoxybenzyl chloride with 2-piperazinone followed by reduction with lithium aluminum hydride (LiAlH4). This route is fundamentally flawed for modern manufacturing due to the extreme safety hazards posed by LiAlH4, which is pyrophoric and requires stringent anhydrous conditions, alongside the generation of substantial aluminum waste. Furthermore, alternative pathways utilizing sodium borohydride (NaBH4) for the reductive amination of the corresponding aldehyde, as seen in CN 1715275, still suffer from the high cost of stoichiometric hydride reagents and the complexity of quenching and disposing of boron-containing byproducts.
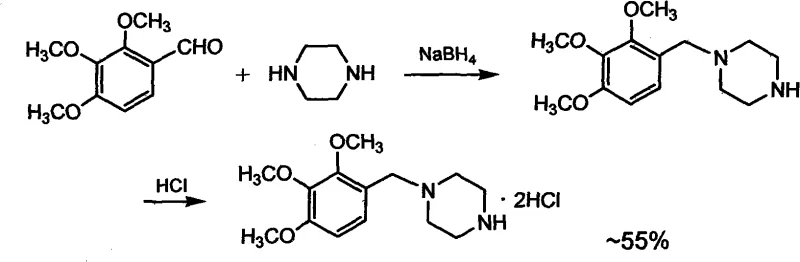
Another prevalent conventional method involves catalytic hydrogenation using palladium on carbon (Pd/C), as illustrated in US 5142053 and EP 453365. While this method avoids hydride reagents, it introduces a different set of supply chain vulnerabilities centered on the reliance on precious metal catalysts. Palladium is subject to extreme price volatility and geopolitical supply constraints, making long-term cost forecasting difficult for procurement teams. Additionally, the removal of trace palladium from the final API to meet strict regulatory limits (often in the ppm range) requires additional purification steps, such as specialized filtration or scavenging resins, which further erodes profit margins and extends production lead times. These cumulative factors render conventional methods suboptimal for the high-volume, cost-sensitive manufacturing required by the global generic pharmaceutical market.
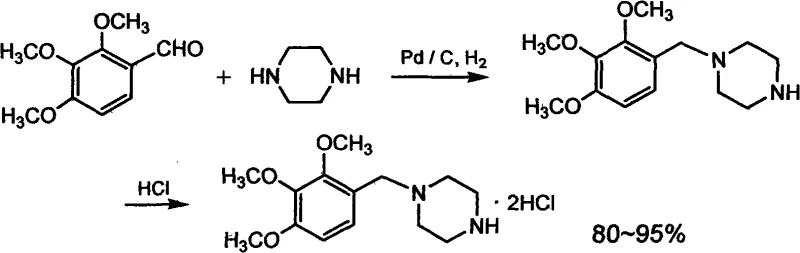
The Novel Approach
The methodology disclosed in patent CN101575321A effectively circumvents these historical bottlenecks by substituting expensive palladium catalysts with economical nickel-based alternatives. The process initiates with the direct condensation of 2,3,4-trimethoxybenzaldehyde and anhydrous piperazine in a suitable solvent system, followed by catalytic hydrogenation using nickel catalysts such as Raney nickel, nickel on diatomaceous earth, or nickel boride. This strategic shift allows the reaction to proceed under moderate hydrogen pressures (0.7-2.0 MPa) and temperatures (50-95°C), achieving yields of 82-90% for the free base. By eliminating the need for stoichiometric hydride reagents and precious metals, this novel approach not only enhances the intrinsic safety profile of the operation but also stabilizes the bill of materials (BOM) costs, providing a predictable and scalable foundation for commercial production.
Mechanistic Insights into Nickel-Catalyzed Reductive Amination
The core of this technological advancement lies in the efficient mechanism of nickel-catalyzed reductive amination, which proceeds through a well-defined sequence of imine formation and subsequent hydrogenation. Initially, the carbonyl group of 2,3,4-trimethoxybenzaldehyde undergoes nucleophilic attack by the secondary amine of piperazine to form an iminium ion or Schiff base intermediate in situ. The nickel catalyst, possessing high surface area and active sites, facilitates the heterolytic cleavage of molecular hydrogen, generating active nickel-hydride species on the catalyst surface. These species then transfer hydrogen to the electrophilic carbon of the iminium intermediate, reducing it to the stable tertiary amine bond found in Trimetazidine. The choice of nickel over other transition metals is critical, as it offers a balanced activity that promotes the desired reduction without causing excessive hydrogenolysis of the methoxy groups or the benzyl-nitrogen bond, which are common side reactions that degrade product purity.
Furthermore, the workup procedure described in the patent is meticulously designed to control the impurity profile and ensure high product quality. Following the hydrogenation, the reaction mixture is treated with acid to adjust the pH to 3-4, which protonates the amine product and allows for the separation of non-basic organic impurities into the organic phase. Subsequent basification of the aqueous phase to pH 12 regenerates the free base, which is then extracted into an aromatic solvent. This acid-base extraction strategy is highly effective at removing unreacted aldehyde, piperazine, and potential over-reduction byproducts, ensuring that the final isolated Trimetazidine meets the stringent purity specifications required for downstream salt formation. The ability to achieve such high purity through simple liquid-liquid extraction rather than complex chromatography is a key factor in the process economic viability.
How to Synthesize Trimetazidine Efficiently
The implementation of this nickel-catalyzed route requires precise control over reaction parameters to maximize yield and safety. The process begins by charging an autoclave with a solvent such as dimethoxyethane or ethanol, followed by the addition of 2,3,4-trimethoxybenzaldehyde and anhydrous piperazine in a molar ratio ranging from 1:1 to 1:3. A nickel-type catalyst, constituting 3-10% of the total reactant mass, is introduced before the system is purged with nitrogen to remove oxygen. Hydrogen is then introduced to maintain a pressure of 0.7-2.0 MPa while the temperature is raised to 50-95°C for a duration of 4-10 hours. Detailed standardized synthesis steps, including specific workup protocols and crystallization conditions for the hydrochloride salt, are provided in the technical guide below.
- Charge an autoclave with solvent, 2,3,4-trimethoxybenzaldehyde, anhydrous piperazine (molar ratio 1: 1 to 1:3), and a nickel-type catalyst (3-10% by weight).
- Purge with nitrogen, pressurize with hydrogen to 0.7-2.0 MPa, and maintain reaction temperature between 50-95°C for 4-10 hours until hydrogen absorption ceases.
- Filter the mixture, adjust pH to 3-4 to separate organic phases, then basify the aqueous phase to pH 12 and extract with aromatics to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this nickel-catalyzed synthesis route offers profound strategic advantages that extend beyond simple yield improvements. The primary benefit is the drastic reduction in raw material costs achieved by replacing volatile precious metal catalysts with abundant and inexpensive nickel variants. This substitution insulates the manufacturing process from the erratic pricing fluctuations of the platinum group metals market, allowing for more accurate long-term budgeting and contract negotiations. Moreover, the elimination of hazardous hydride reagents like LiAlH4 removes the need for specialized storage facilities and expensive waste disposal contracts associated with reactive metal wastes, thereby lowering the overall overhead costs of the production facility.
- Cost Reduction in Manufacturing: The transition from palladium or stoichiometric hydrides to nickel catalysts results in substantial cost savings by lowering the direct material expense per kilogram of product. Since nickel catalysts are significantly cheaper than palladium and can often be recycled or disposed of at a lower cost, the overall catalyst expenditure is minimized. Additionally, the simplified workup procedure, which relies on standard extraction techniques rather than complex purification columns, reduces solvent consumption and labor hours, further driving down the cost of goods sold (COGS) for this high-purity pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: Utilizing nickel catalysts and common solvents like ethanol or ethers ensures a robust and resilient supply chain that is less susceptible to geopolitical disruptions. Unlike palladium, which is sourced from a limited number of regions, nickel is globally available with a stable supply infrastructure. This availability guarantees consistent production schedules and reduces the risk of manufacturing stoppages due to catalyst shortages. Furthermore, the use of stable, non-pyrophoric reagents simplifies logistics and transportation, allowing for safer and more flexible inventory management strategies.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively in high-pressure autoclaves suitable for multi-ton production. From an environmental perspective, the avoidance of heavy metal waste (from palladium) and reactive metal sludge (from aluminum or boron hydrides) simplifies effluent treatment and helps facilities meet increasingly rigorous environmental regulations. The cleaner reaction profile reduces the burden on wastewater treatment plants and minimizes the generation of hazardous solid waste, aligning the manufacturing process with green chemistry principles and corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These answers are derived directly from the experimental data and beneficial effects reported in patent CN101575321A, providing a factual basis for evaluating the technology feasibility. Understanding these details is crucial for technical teams assessing the compatibility of this process with existing manufacturing infrastructure and quality standards.
Q: What are the primary advantages of using a nickel catalyst over palladium for trimetazidine synthesis?
A: The use of nickel catalysts significantly reduces raw material costs compared to precious metal palladium catalysts while maintaining comparable product yields of 82-90%. This substitution eliminates the economic volatility associated with precious metal markets and simplifies the supply chain for catalyst procurement.
Q: How does this method improve safety compared to traditional hydride reduction routes?
A: This catalytic hydrogenation method completely avoids the use of hazardous reducing agents such as lithium aluminum hydride (LiAlH4) or sodium borohydride (NaBH4). By utilizing hydrogen gas and a heterogeneous nickel catalyst, the process mitigates the risks of pyrophoric reactions and simplifies waste treatment protocols.
Q: What is the expected yield and purity profile for this synthetic route?
A: According to patent data, the free base yield ranges from 82% to 90%, with the subsequent hydrochloride salt formation achieving yields between 78% and 92%. The process includes specific pH-controlled extraction steps designed to remove organic impurities and ensure high purity suitable for pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trimetazidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our team of expert chemists has extensively evaluated the nickel-catalyzed route for Trimetazidine and possesses the technical capability to implement this process with precision and efficiency. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of high-quality intermediates. Our state-of-the-art facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of Trimetazidine meets the highest industry standards for safety and efficacy.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain requirements. By leveraging our expertise, you can access a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating the tangible economic benefits of switching to this nickel-catalyzed method. We encourage you to contact us today to request specific COA data and route feasibility assessments, and let us demonstrate why NINGBO INNO PHARMCHEM is the preferred partner for reliable, cost-effective, and scalable pharmaceutical intermediate solutions.
