Advanced Manufacturing of 4-Methyl-3-[[4-(3-Pyridyl)-2-Pyrimidinyl]Amino]Benzoic Acid for Global Pharma Supply Chains
Introduction to Advanced Nilotinib Intermediate Synthesis
The pharmaceutical industry continuously seeks robust, scalable, and safe manufacturing routes for critical active pharmaceutical ingredient (API) intermediates. A pivotal development in this domain is documented in patent CN101928277A, which details a highly efficient preparation method for 4-methyl-3-[[4-(3-pyridyl)-2-pyrimidinyl]amino]benzoic acid. This compound serves as a crucial building block in the synthesis of Nilotinib, a potent tyrosine kinase inhibitor used in cancer therapy. The structural integrity and purity of this intermediate are paramount for the efficacy and safety of the final drug product. The patented methodology represents a significant leap forward by addressing long-standing challenges associated with traditional synthetic pathways, specifically focusing on process safety, yield optimization, and operational simplicity.
![Chemical structure of 4-methyl-3-[[4-(3-pyridyl)-2-pyrimidinyl]amino]benzoic acid (Formula I)](/insights/img/nilotinib-intermediate-synthesis-pharma-supplier-20260313055908-01.png)
For R&D directors and procurement specialists, understanding the nuances of this synthesis is essential for securing a reliable pharmaceutical intermediate supplier. The innovation lies not just in the chemical transformation but in the holistic redesign of the process flow to eliminate hazardous steps and reduce the overall environmental footprint. By bypassing complex protection-deprotection sequences, the new route offers a streamlined approach that aligns perfectly with modern green chemistry principles while ensuring commercial viability. This report delves deep into the technical merits of this invention, analyzing how it transforms the manufacturing landscape for this high-value chemical entity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the standard industrial synthesis, as described in patents like US 7169791, relied on a cumbersome four-step sequence. This conventional pathway necessitated the initial esterification of 3-amino-4-methylbenzoic acid to protect the carboxylic acid functionality. Following esterification, the amine group was converted into a guanidine moiety, typically resulting in an unstable guanidine nitrate salt intermediate. This nitrate salt presented significant safety hazards due to its oxidative potential and thermal instability, posing risks during large-scale storage and processing. Furthermore, the subsequent cyclization and final hydrolysis steps to regenerate the carboxylic acid added considerable complexity, time, and cost to the operation.
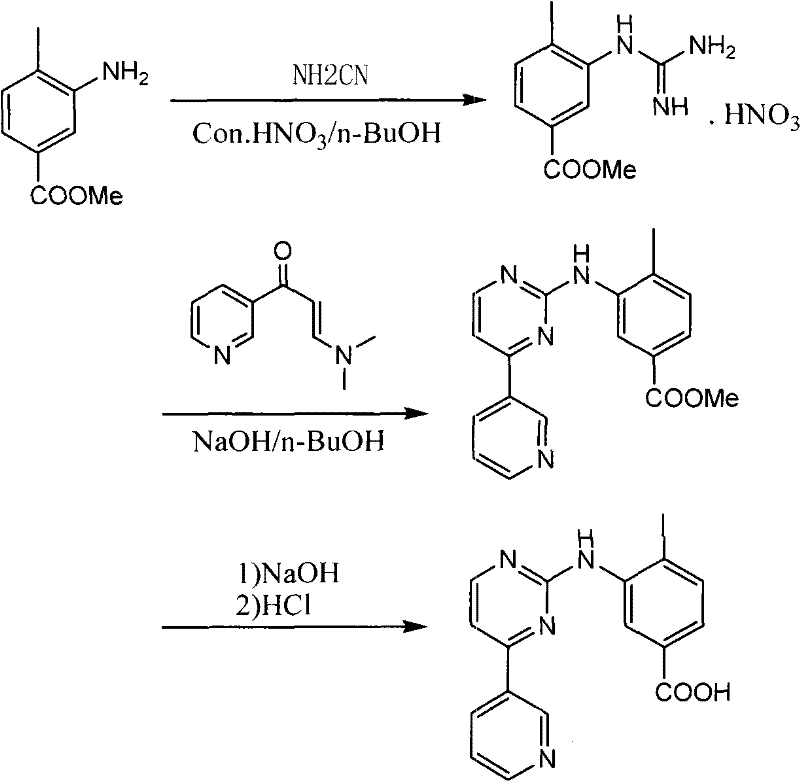
The cumulative effect of these additional steps was a substantial reduction in overall yield and an increase in waste generation. The need to handle explosive or unstable nitrate salts required specialized equipment and rigorous safety protocols, driving up capital and operational expenditures. Moreover, the multi-step nature of the process introduced more opportunities for impurity formation, complicating purification and potentially compromising the quality of the final API. For supply chain heads, these factors translated into longer lead times, higher vulnerability to production disruptions, and increased logistical burdens associated with hazardous material handling.
The Novel Approach
The methodology disclosed in CN101928277A fundamentally reimagines this synthesis by collapsing the four-step sequence into a concise two-step process. The breakthrough involves the direct reaction of 3-amino-4-methylbenzoic acid with cyanamide under acidic conditions to form a stable guanidine hydrochloride intermediate. This strategic shift eliminates the need for esterification and the subsequent hydrolysis, thereby removing the requirement for carboxyl group protection entirely. The resulting hydrochloride salt is markedly more stable and safer to handle than its nitrate counterpart, mitigating the safety risks associated with the older technology.
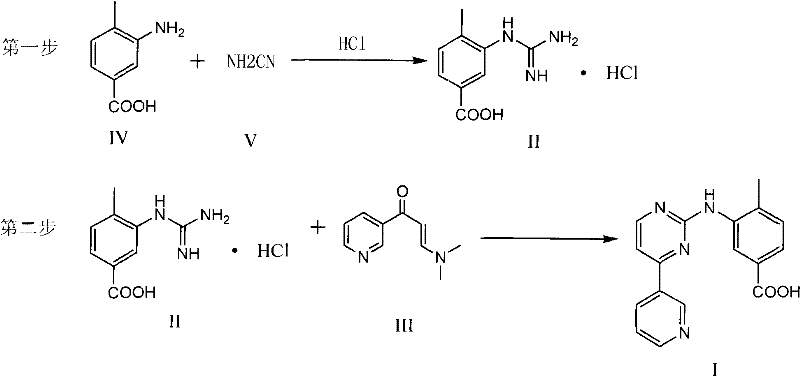
In the second step, this stable intermediate undergoes a direct cyclization reaction with 3-(dimethylamino)-1-(3-pyridyl)-2-propen-1-one. This convergence of reactants occurs under controlled pH conditions in organic solvents, leading directly to the target benzoic acid derivative. The elimination of two entire synthetic steps drastically reduces solvent consumption, energy usage, and reaction time. For a reliable pharmaceutical intermediate supplier, this simplification translates into a more robust supply chain capable of delivering high-purity materials with greater consistency and lower cost structures, effectively solving the bottlenecks inherent in the legacy manufacturing processes.
Mechanistic Insights into Direct Guanidination and Cyclization
The core chemical innovation of this process lies in the successful execution of direct guanidination on a free carboxylic acid substrate. Traditionally, the acidic proton of the carboxylic acid interferes with the formation of the guanidine group or leads to side reactions, necessitating protection. However, by optimizing the reaction medium with specific alcoholic solvents and maintaining a precise acidic pH range (typically pH 1-5 using hydrochloric acid), the reaction selectively targets the aniline nitrogen. The use of cyanamide as the guanidinating agent in the presence of HCl facilitates the formation of the guanidine hydrochloride salt directly. This salt formation acts as a driving force, precipitating the intermediate or keeping it in a stable ionic form that resists degradation.
The subsequent cyclization mechanism involves the nucleophilic attack of the guanidine nitrogen on the electrophilic carbon of the enaminone derivative (3-(dimethylamino)-1-(3-pyridyl)-2-propen-1-one). The reaction conditions, specifically the temperature range of 80-100°C and a slightly basic to neutral pH (adjusted to 7-12 depending on the specific solvent system), promote the elimination of dimethylamine and the closure of the pyrimidine ring. The presence of the free carboxylic acid during this step is tolerated and does not interfere, which is a testament to the chemoselectivity of the optimized conditions. This mechanistic efficiency ensures that the final product is formed with minimal byproduct generation, simplifying downstream purification.
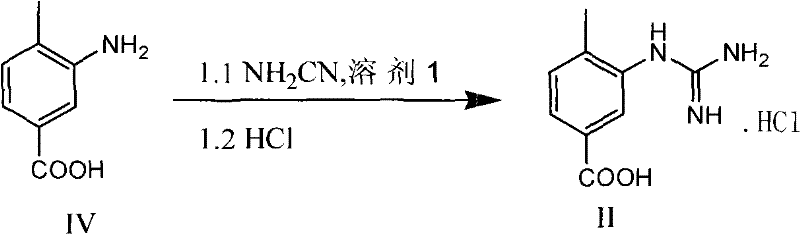
Impurity control is inherently built into this mechanism. By avoiding the nitrate salt intermediate, the process eliminates the risk of nitration byproducts or oxidative degradation products that often plague the conventional route. Furthermore, the direct crystallization of the hydrochloride intermediate allows for an effective purge of unreacted starting materials and soluble impurities before the cyclization step. This "purge point" strategy ensures that the final cyclization proceeds with high-quality inputs, resulting in a final product with exceptional purity profiles, often exceeding 99.8% as demonstrated in the patent examples. Such high purity is critical for minimizing the burden on final API purification stages.
How to Synthesize 4-Methyl-3-[[4-(3-Pyridyl)-2-Pyrimidinyl]Amino]Benzoic Acid Efficiently
Implementing this synthesis requires precise control over reaction parameters to maximize yield and safety. The process begins with the suspension of 3-amino-4-methylbenzoic acid in an alcohol solvent, followed by the addition of aqueous cyanamide and hydrochloric acid. Temperature control is vital during the exothermic acid addition, typically maintained between 50-150°C depending on the solvent boiling point. The second stage involves reacting the isolated or in-situ generated guanidine hydrochloride with the pyridyl ketone derivative in a solvent system such as isoamyl alcohol or MIBK, with careful pH monitoring to ensure complete cyclization without degrading the sensitive pyrimidine ring.
- React 3-amino-4-methylbenzoic acid with cyanamide in an alcoholic solvent under acidic conditions (HCl) to form the stable guanidine hydrochloride intermediate.
- Perform a cyclization reaction between the guanidine hydrochloride intermediate and 3-(dimethylamino)-1-(3-pyridyl)-2-propen-1-one in a suitable organic solvent at elevated temperatures.
- Purify the crude product through recrystallization using alcohol-water mixtures and pH adjustment to achieve high purity specifications.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the transition to this patented synthesis route offers compelling economic and operational benefits. The primary advantage stems from the drastic simplification of the manufacturing workflow. By reducing the number of unit operations from four distinct chemical transformations to just two, the process inherently lowers the consumption of raw materials, solvents, and utilities. This reduction in material intensity directly correlates to a significant decrease in the cost of goods sold (COGS), allowing for more competitive pricing strategies in the global market without sacrificing margin.
- Cost Reduction in Manufacturing: The elimination of the esterification and hydrolysis steps removes the need for expensive esterifying agents and the large volumes of base required for saponification. Furthermore, the avoidance of the unstable nitrate salt intermediate reduces the costs associated with specialized safety infrastructure and hazardous waste disposal. The higher overall yield reported in the patent examples implies that less starting material is required to produce the same amount of final product, further driving down the effective cost per kilogram. These cumulative savings create a substantial cost advantage in pharmaceutical intermediate manufacturing.
- Enhanced Supply Chain Reliability: The stability of the guanidine hydrochloride intermediate is a game-changer for inventory management. Unlike the nitrate salt, which may require cold storage or immediate processing, the hydrochloride salt can be stored safely for extended periods. This stability allows manufacturers to build strategic stockpiles of the intermediate, buffering against supply chain disruptions and ensuring continuous production of the final API. Additionally, the use of common, non-hazardous solvents like alcohols and esters simplifies logistics and reduces regulatory hurdles associated with transporting dangerous goods.
- Scalability and Environmental Compliance: The shortened reaction sequence naturally leads to a smaller environmental footprint. Fewer steps mean less solvent waste, lower energy consumption for heating and cooling, and reduced wastewater load. This aligns with increasingly stringent environmental regulations and corporate sustainability goals. The process is designed for scalability, with reaction conditions that are easily transferable from laboratory to pilot and full commercial scale. The robustness of the chemistry ensures that quality remains consistent regardless of batch size, providing supply chain heads with the confidence to scale up production to meet surging demand for Nilotinib.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of this critical intermediate. These insights are derived directly from the patented technology and are intended to clarify the operational benefits for potential partners and stakeholders. Understanding these details is crucial for making informed sourcing decisions and optimizing your own downstream manufacturing processes.
Q: Why is the guanidine hydrochloride intermediate preferred over the nitrate salt in this synthesis?
A: The guanidine hydrochloride intermediate offers significantly higher stability and safety compared to the nitrate salt used in conventional methods. Nitrate salts pose oxidation risks and stability issues during storage and processing, whereas the hydrochloride salt is robust, facilitating safer large-scale handling and improving overall process reliability.
Q: How does this patented route reduce manufacturing costs compared to prior art?
A: This route eliminates the need for esterification and subsequent hydrolysis steps required to protect the carboxyl group in older methods. By reducing the synthesis from four steps to just two, it significantly lowers raw material consumption, reduces solvent usage, minimizes waste generation, and shortens production cycles, leading to substantial cost efficiencies.
Q: What purity levels can be achieved with this optimized crystallization process?
A: Through optimized recrystallization involving alcohol-water solvents and precise pH control, the process consistently achieves product purity exceeding 99.8%, with maximum single impurity levels controlled below 0.1%, meeting stringent requirements for pharmaceutical intermediate applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Methyl-3-[[4-(3-Pyridyl)-2-Pyrimidinyl]Amino]Benzoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development and commercialization of life-saving oncology drugs. Our technical team has extensively analyzed the patented route described in CN101928277A and possesses the expertise to implement this advanced synthesis at an industrial scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 4-methyl-3-[[4-(3-pyridyl)-2-pyrimidinyl]amino]benzoic acid meets the highest industry standards.
We invite you to collaborate with us to optimize your supply chain for Nilotinib production. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. By partnering with us, you gain access to a secure, cost-effective, and technologically advanced source for this essential pharmaceutical building block, ensuring the continuity and success of your drug development programs.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
