Scalable Synthesis of Fluorinated Quinoxalinones via Biomass-Supported Copper Catalysis
Scalable Synthesis of Fluorinated Quinoxalinones via Biomass-Supported Copper Catalysis
The introduction of trifluoromethyl groups into organic molecules is a cornerstone strategy in modern medicinal chemistry, significantly enhancing metabolic stability and lipophilicity. As illustrated in the structural diversity of known bioactive compounds, the trifluoromethyl motif is prevalent in critical therapeutics ranging from antidepressants to antiviral agents. 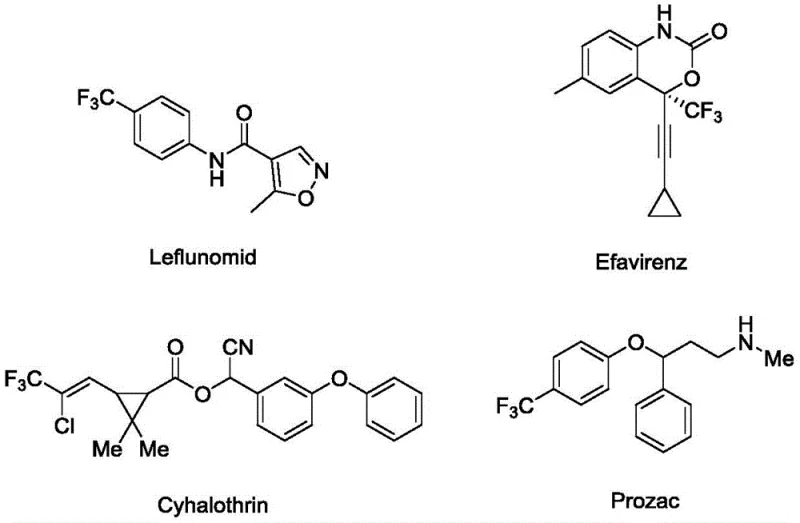 Patent CN113444076A introduces a groundbreaking application of biomass-loaded copper catalysis for the efficient preparation of these vital fluorine-containing intermediates. This technology specifically targets the synthesis of 3-(2-(trifluoromethyl)-indol-3-yl)quinoxaline-2-one derivatives through a sustainable three-component one-pot reaction. By leveraging a chitosan-supported copper catalyst, this method addresses the longstanding industry challenges associated with traditional homogeneous catalysis, such as difficult catalyst recovery and persistent metal contamination. For R&D directors and procurement specialists seeking reliable pharmaceutical intermediate suppliers, this innovation represents a significant leap towards greener, more cost-effective manufacturing processes that align with stringent global regulatory standards for impurity control.
Patent CN113444076A introduces a groundbreaking application of biomass-loaded copper catalysis for the efficient preparation of these vital fluorine-containing intermediates. This technology specifically targets the synthesis of 3-(2-(trifluoromethyl)-indol-3-yl)quinoxaline-2-one derivatives through a sustainable three-component one-pot reaction. By leveraging a chitosan-supported copper catalyst, this method addresses the longstanding industry challenges associated with traditional homogeneous catalysis, such as difficult catalyst recovery and persistent metal contamination. For R&D directors and procurement specialists seeking reliable pharmaceutical intermediate suppliers, this innovation represents a significant leap towards greener, more cost-effective manufacturing processes that align with stringent global regulatory standards for impurity control.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methodologies for constructing carbon-trifluoromethyl bonds often rely heavily on transition metal-catalyzed cross-coupling reactions involving phenylboronic acids, halocarbons, or carboxylic acids. These conventional pathways are frequently plagued by the necessity for noble and toxic transition metals, which pose severe environmental hazards and complicate the purification process due to strict limits on residual heavy metals in active pharmaceutical ingredients. Furthermore, many established trifluoromethylation protocols require harsh reaction conditions, including elevated temperatures and high catalytic loadings, which can lead to poor atom economy and increased energy consumption. The inability to recover and reuse homogeneous catalysts results in substantial operational costs and generates significant chemical waste, creating a bottleneck for the commercial scale-up of complex fluorinated scaffolds. Consequently, there is an urgent demand within the fine chemical sector for milder, more efficient, and sustainable direct C-H bond trifluoromethylation methods that can overcome these economic and ecological barriers.
The Novel Approach
The novel approach detailed in the patent utilizes a heterogeneous biomass-supported copper catalyst to drive a three-component coupling reaction between quinoxalinones, indoles, and sodium trifluoromethanesulfinate. This method fundamentally shifts the paradigm by employing chitosan, a renewable biopolymer, as a carrier for the copper species, thereby creating a robust heterogeneous system. 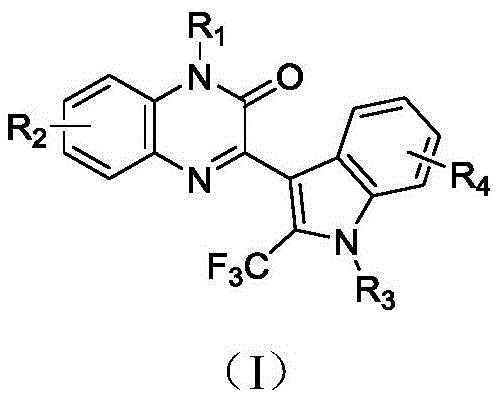 This design not only facilitates the easy separation of the catalyst from the reaction mixture via simple filtration but also enables multiple recycling cycles without compromising catalytic activity. The reaction operates under remarkably mild conditions, typically at 60°C in acetonitrile, utilizing environmentally benign oxidants like potassium persulfate. By expanding the substrate scope to include various substituted quinoxalinones and indoles, this technology offers a versatile platform for generating diverse libraries of fluorinated heterocycles. The ability to maintain high yields while eliminating metal residues makes this approach exceptionally attractive for the cost reduction in pharmaceutical intermediate manufacturing, providing a clear pathway for industrial adoption.
This design not only facilitates the easy separation of the catalyst from the reaction mixture via simple filtration but also enables multiple recycling cycles without compromising catalytic activity. The reaction operates under remarkably mild conditions, typically at 60°C in acetonitrile, utilizing environmentally benign oxidants like potassium persulfate. By expanding the substrate scope to include various substituted quinoxalinones and indoles, this technology offers a versatile platform for generating diverse libraries of fluorinated heterocycles. The ability to maintain high yields while eliminating metal residues makes this approach exceptionally attractive for the cost reduction in pharmaceutical intermediate manufacturing, providing a clear pathway for industrial adoption.
Mechanistic Insights into Biomass-Supported Copper-Catalyzed Trifluoromethylation
The core of this technological advancement lies in the unique interaction between the chitosan support and the copper active sites, which stabilizes the catalytic species and modulates its reactivity. The proposed mechanism involves the activation of sodium trifluoromethanesulfinate by the copper catalyst in the presence of an oxidant to generate trifluoromethyl radicals. These highly reactive radicals subsequently attack the electron-rich indole ring at the C3 position, followed by coupling with the quinoxalinone derivative to form the final C-C bond. 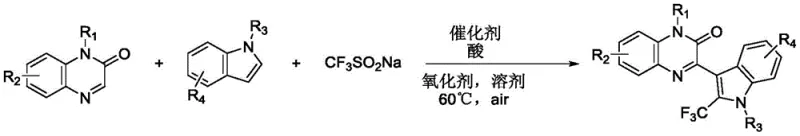 The chitosan matrix likely plays a dual role: it prevents the aggregation of copper nanoparticles, maintaining a high surface area for catalysis, and it may also participate in hydrogen bonding interactions that stabilize transition states. This synergistic effect ensures that the reaction proceeds with high selectivity and efficiency, minimizing the formation of side products. For technical teams evaluating process feasibility, understanding this mechanistic nuance is crucial, as it explains the observed tolerance for various functional groups and the consistent performance across different substrate combinations.
The chitosan matrix likely plays a dual role: it prevents the aggregation of copper nanoparticles, maintaining a high surface area for catalysis, and it may also participate in hydrogen bonding interactions that stabilize transition states. This synergistic effect ensures that the reaction proceeds with high selectivity and efficiency, minimizing the formation of side products. For technical teams evaluating process feasibility, understanding this mechanistic nuance is crucial, as it explains the observed tolerance for various functional groups and the consistent performance across different substrate combinations.
Impurity control is another critical aspect where this heterogeneous system excels compared to its homogeneous counterparts. In traditional liquid-phase catalysis, trace amounts of metal ions often remain entrapped within the product crystal lattice or solution, requiring expensive and time-consuming scavenging steps to meet pharmacopeial standards. In contrast, the solid nature of the Cu(OAc)2@CS catalyst ensures that the vast majority of copper remains bound to the chitosan support throughout the reaction cycle. Post-reaction filtration effectively removes the catalyst, resulting in a crude product with significantly lower metal content. This inherent purity advantage simplifies the downstream workup, often reducing the number of recrystallization or chromatography steps required. For supply chain managers, this translates to shorter production cycles and reduced solvent usage, directly contributing to a more sustainable and economically viable manufacturing footprint for high-purity pharmaceutical intermediates.
How to Synthesize 3-(2-(trifluoromethyl)-indol-3-yl)quinoxaline-2-one Efficiently
The synthesis protocol outlined in the patent provides a clear, step-by-step guide for implementing this green chemistry solution in a laboratory or pilot plant setting. The process begins with the preparation of the specialized catalyst, followed by the independent synthesis of the necessary quinoxalinone and indole precursors, which are then combined in the final trifluoromethylation step. The detailed standardized synthesis steps below provide the exact stoichiometric ratios, temperature controls, and workup procedures required to achieve optimal yields exceeding 80%.
- Prepare the heterogeneous biomass-supported copper catalyst by loading copper acetate onto chitosan carrier.
- Synthesize the quinoxalinone and indole precursors via standard alkylation methods.
- Perform the three-component reaction with sodium trifluoromethanesulfinate, oxidant, and acid at 60°C.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this biomass-supported copper catalysis technology offers compelling strategic advantages that extend beyond mere technical feasibility. The primary value proposition lies in the drastic simplification of the purification workflow, which directly impacts the cost of goods sold. By eliminating the need for complex metal scavenging resins and extensive chromatographic purification to remove homogeneous catalyst residues, manufacturers can achieve substantial cost savings in raw materials and processing time. Furthermore, the use of chitosan, a widely available and inexpensive biopolymer derived from shellfish waste, as a catalyst support reduces the dependency on costly synthetic ligands often required in homogeneous systems. This shift towards bio-based materials not only lowers input costs but also aligns with corporate sustainability goals, enhancing the brand value of the final pharmaceutical product in markets that prioritize green chemistry.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the recyclability of the heterogeneous catalyst and the simplified downstream processing. Unlike traditional methods where the catalyst is consumed or lost in the aqueous waste stream, the Cu(OAc)2@CS catalyst can be recovered by simple filtration and reused for multiple batches without significant loss of activity. This reusability drastically reduces the per-kilogram cost of the catalyst, which is often a major expense in transition metal-catalyzed reactions. Additionally, the mild reaction conditions (60°C) reduce energy consumption compared to high-temperature alternatives, further lowering utility costs. The elimination of expensive metal scavengers and the reduction in solvent usage for purification contribute to a leaner, more cost-efficient manufacturing model that improves overall profit margins for high-volume production.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness and simplicity of this synthetic route. The reliance on commercially available and stable starting materials, such as sodium trifluoromethanesulfinate and common heterocycles, minimizes the risk of raw material shortages. The heterogeneous nature of the catalyst ensures consistent batch-to-batch quality, reducing the variability that can lead to production delays or failed batches. Moreover, the ability to recycle the catalyst reduces the logistical burden of sourcing fresh catalyst for every production run, insulating the supply chain from fluctuations in the price and availability of copper salts and ligands. This stability is crucial for maintaining continuous supply to downstream API manufacturers, ensuring that delivery schedules are met reliably even in volatile market conditions.
- Scalability and Environmental Compliance: Scaling this process from gram to ton scale is facilitated by the straightforward engineering requirements of heterogeneous catalysis. The reaction does not require specialized high-pressure equipment or cryogenic conditions, making it compatible with standard glass-lined or stainless steel reactors found in most multipurpose chemical plants. From an environmental compliance perspective, the process generates significantly less hazardous waste. The absence of toxic homogeneous metal residues in the effluent simplifies wastewater treatment, reducing the cost and complexity of meeting environmental discharge regulations. The use of a bio-based support material further enhances the green profile of the process, potentially qualifying the manufacturing site for environmental incentives or certifications that are increasingly important in the global pharmaceutical supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this biomass-supported copper catalysis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on performance metrics and operational parameters.
Q: How does the biomass-supported copper catalyst improve product purity?
A: The heterogeneous nature of the chitosan-supported copper catalyst allows for easy filtration, effectively eliminating toxic metal residues that are common with homogeneous catalysts, thereby simplifying downstream purification.
Q: Can the catalyst be reused in industrial production?
A: Yes, the patent demonstrates that the Cu(OAc)2@CS catalyst maintains high catalytic efficiency over at least seven recycling cycles without significant loss in yield, ensuring long-term process viability.
Q: What are the reaction conditions for this trifluoromethylation?
A: The reaction proceeds under mild conditions at 60°C in acetonitrile solvent, utilizing potassium persulfate as an oxidant and acetic acid as an additive, avoiding harsh temperatures or pressures.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-(2-(trifluoromethyl)-indol-3-yl)quinoxaline-2-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the biomass-loaded copper catalytic three-component reaction in advancing the synthesis of fluorinated pharmaceutical intermediates. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering high-purity intermediates that meet stringent purity specifications, leveraging our rigorous QC labs to verify the absence of metal residues and ensure consistent quality across every batch. Our state-of-the-art facilities are equipped to handle the specific requirements of heterogeneous catalysis, including efficient filtration and catalyst recovery systems, maximizing the economic and environmental benefits of this innovative technology for our partners.
We invite forward-thinking pharmaceutical companies and research institutions to collaborate with us to unlock the full potential of this green synthesis route. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific production volumes and purity requirements. We encourage you to contact our technical procurement team today to request specific COA data for our fluorinated quinoxalinone derivatives and to discuss detailed route feasibility assessments. Together, we can drive down costs, enhance supply chain security, and accelerate the development of next-generation fluorine-containing medicines.
