Sustainable Synthesis of Fluorinated Quinoxalinones via Recyclable Biomass Copper Catalysis
Sustainable Synthesis of Fluorinated Quinoxalinones via Recyclable Biomass Copper Catalysis
The introduction of trifluoromethyl groups into organic molecules is a cornerstone strategy in modern medicinal chemistry, profoundly influencing the metabolic stability, lipophilicity, and bioavailability of drug candidates. As illustrated in the structural diversity of known bioactive molecules, the trifluoromethyl motif is prevalent in critical therapeutics ranging from antidepressants to antiviral agents. 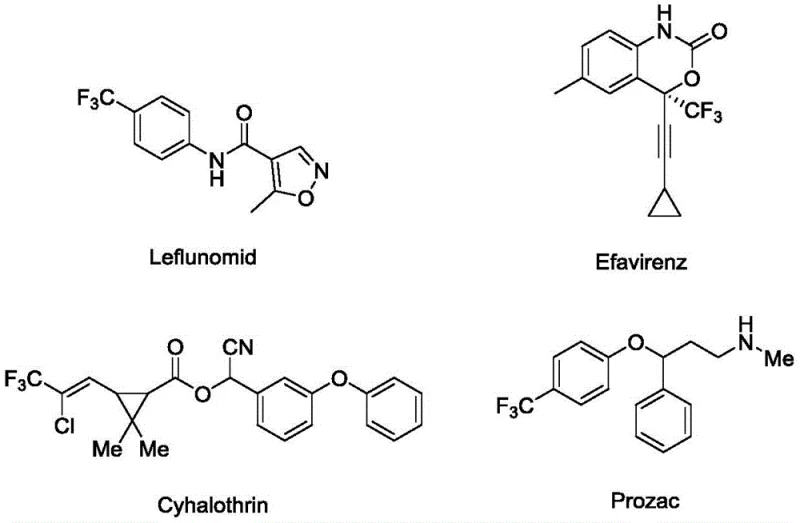 Patent CN113444076A discloses a groundbreaking application of a biomass-loaded copper-catalyzed three-component reaction specifically designed for the preparation of fluorine-containing medicaments. This technology addresses the urgent industry demand for greener, more efficient synthetic routes by utilizing a chitosan-supported copper catalyst to facilitate the coupling of quinoxalinones, indoles, and sodium trifluoromethanesulfinate. For R&D directors and procurement specialists seeking reliable pharmaceutical intermediate suppliers, this innovation represents a significant leap forward in process sustainability and cost-effectiveness, offering a robust pathway to complex fluorinated scaffolds essential for next-generation drug discovery.
Patent CN113444076A discloses a groundbreaking application of a biomass-loaded copper-catalyzed three-component reaction specifically designed for the preparation of fluorine-containing medicaments. This technology addresses the urgent industry demand for greener, more efficient synthetic routes by utilizing a chitosan-supported copper catalyst to facilitate the coupling of quinoxalinones, indoles, and sodium trifluoromethanesulfinate. For R&D directors and procurement specialists seeking reliable pharmaceutical intermediate suppliers, this innovation represents a significant leap forward in process sustainability and cost-effectiveness, offering a robust pathway to complex fluorinated scaffolds essential for next-generation drug discovery.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methodologies for introducing trifluoromethyl groups often rely heavily on homogeneous transition metal catalysis, which presents substantial challenges for industrial scale-up and regulatory compliance. These conventional processes typically require noble or toxic metals that are difficult to remove completely from the final product, posing severe risks regarding heavy metal residues in Active Pharmaceutical Ingredients (APIs). Furthermore, the harsh reaction conditions frequently associated with these methods, such as extreme temperatures or pressures, coupled with high catalytic loading requirements, drastically increase operational expenditures and safety hazards. The inability to recover and reuse homogeneous catalysts not only inflates raw material costs but also generates significant volumes of hazardous waste, complicating environmental compliance and waste treatment protocols for manufacturing facilities. Consequently, there is a persistent and critical need within the fine chemical sector to develop milder, more efficient, and environmentally benign trifluoromethylation strategies that do not compromise on yield or purity.
The Novel Approach
The novel approach detailed in the patent utilizes a heterogeneous biomass-supported copper catalyst, specifically copper acetate loaded onto chitosan, to overcome the inherent drawbacks of traditional homogeneous systems. This innovative strategy enables a one-pot three-component synthesis of 3-(2-(trifluoromethyl)-indol-3-yl)quinoxalin-2-one derivatives under remarkably mild conditions, typically around 60°C in acetonitrile. By employing a solid-supported catalyst, the process allows for simple filtration to separate the catalyst from the reaction mixture, thereby eliminating the need for complex and costly metal scavenging steps. The general structure of the target fluorinated medicament is shown below, highlighting the versatile substitution patterns achievable with this method. 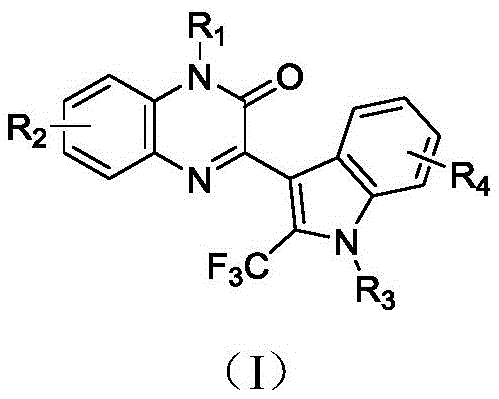 This methodology not only expands the accessible chemical space with various substituents on the indole and quinoxalinone rings but also ensures that the catalyst can be recycled multiple times while maintaining high catalytic activity, thus promoting the feasibility of industrialization for these valuable fluorinated intermediates.
This methodology not only expands the accessible chemical space with various substituents on the indole and quinoxalinone rings but also ensures that the catalyst can be recycled multiple times while maintaining high catalytic activity, thus promoting the feasibility of industrialization for these valuable fluorinated intermediates.
Mechanistic Insights into Chitosan-Supported Copper Catalyzed Trifluoromethylation
The mechanistic pathway of this transformation involves a sophisticated interplay between the heterogeneous copper species and the radical generation from the trifluoromethyl source. The chitosan support plays a dual role, acting not only as a stable matrix for the copper active sites but potentially facilitating electron transfer processes due to its amino and hydroxyl functional groups. In the presence of an oxidant such as potassium persulfate, the copper catalyst activates the sodium trifluoromethanesulfinate to generate trifluoromethyl radicals. These highly reactive species subsequently attack the electron-rich indole ring at the C3 position, followed by oxidative coupling with the quinoxalinone moiety. The precise control over the oxidation state of copper on the biomass surface is crucial for sustaining the catalytic cycle without leaching metal ions into the solution. This heterogeneous mechanism ensures that the reaction proceeds with high selectivity, minimizing side reactions and byproduct formation that often plague homogeneous radical trifluoromethylations. The overall reaction scheme demonstrates the convergence of three distinct components into a single complex molecule efficiently. 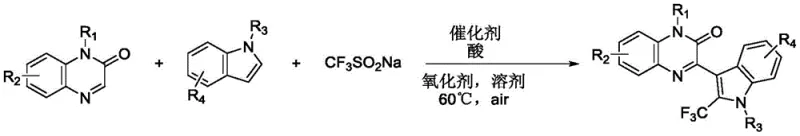 Understanding this mechanism is vital for process chemists aiming to optimize reaction parameters for maximum throughput and minimal impurity profiles in commercial manufacturing settings.
Understanding this mechanism is vital for process chemists aiming to optimize reaction parameters for maximum throughput and minimal impurity profiles in commercial manufacturing settings.
Impurity control in this system is inherently superior due to the heterogeneous nature of the catalysis, which restricts non-specific radical reactions often seen in solution-phase catalysis. The solid support limits the mobility of the active copper species, thereby reducing the likelihood of uncontrolled radical propagation that could lead to poly-fluorinated byproducts or polymerization of the indole substrate. Furthermore, the mild acidic conditions provided by additives like acetic acid help to protonate intermediates selectively, guiding the reaction towards the desired 3-substituted quinoxalinone product. The ability to tune the catalyst loading, ranging from 10 mol% to 30 mol%, allows for fine-tuning the reaction kinetics to balance rate and selectivity. For quality assurance teams, this means a cleaner crude reaction profile, which significantly simplifies downstream purification processes such as column chromatography or crystallization. The result is a high-purity final product that meets stringent pharmaceutical specifications with reduced processing time and solvent consumption, directly impacting the cost of goods sold.
How to Synthesize 3-(2-(trifluoromethyl)-indol-3-yl)quinoxalin-2-one Efficiently
The synthesis protocol outlined in the patent provides a clear and reproducible route for generating these high-value fluorinated intermediates, starting from readily available precursors. The process begins with the preparation of the specific quinoxalinone and indole derivatives, which serve as the core scaffolds for the subsequent coupling reaction. These precursors are then combined with sodium trifluoromethanesulfinate in the presence of the optimized chitosan-copper catalyst and an appropriate oxidant. The reaction is conducted in acetonitrile at a moderate temperature of 60°C, ensuring energy efficiency and operational safety. Detailed standardized synthesis steps for scaling this reaction are provided in the guide below.
- Prepare the heterogeneous catalyst by loading copper acetate onto chitosan biomass support.
- Mix quinoxalinone derivative, indole derivative, sodium trifluoromethanesulfinate, oxidant, and acid in acetonitrile.
- Add the biomass-supported copper catalyst and stir at 60°C under air to complete the trifluoromethylation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this biomass-supported catalytic technology offers transformative advantages in terms of cost structure and supply reliability. The shift from homogeneous to heterogeneous catalysis fundamentally alters the economic model of producing fluorinated intermediates by removing the dependency on expensive, single-use metal catalysts. The ability to recover and reuse the chitosan-copper catalyst for multiple cycles dramatically reduces the raw material cost per kilogram of product, providing a significant competitive edge in pricing strategies. Moreover, the elimination of heavy metal residues simplifies the purification workflow, reducing the consumption of silica gel and solvents required for metal scavenging, which further drives down manufacturing expenses. This process intensification leads to a leaner production cycle with fewer unit operations, enhancing overall plant throughput and capacity utilization without requiring major capital investment in new equipment.
- Cost Reduction in Manufacturing: The implementation of a recyclable heterogeneous catalyst directly translates to substantial cost savings by minimizing catalyst consumption and waste disposal fees. Unlike traditional methods that require stoichiometric or high-loading amounts of precious metals which are lost after each batch, this system allows the catalyst to be filtered, washed, and reused effectively. This reduction in catalyst turnover frequency lowers the variable cost of production significantly. Additionally, the simplified workup procedure reduces the demand for specialized purification resins and extensive washing steps, leading to lower utility and consumable costs. The cumulative effect of these efficiencies results in a more economical manufacturing process that can withstand market fluctuations in raw material prices.
- Enhanced Supply Chain Reliability: Utilizing biomass-derived chitosan as a catalyst support enhances supply chain resilience by relying on renewable and abundant feedstocks rather than scarce geological resources. The robustness of the catalyst, demonstrated by its stability over multiple cycles, ensures consistent production schedules without frequent interruptions for catalyst replenishment. This reliability is crucial for maintaining continuous supply to downstream API manufacturers who demand strict adherence to delivery timelines. Furthermore, the mild reaction conditions reduce the risk of thermal runaways or equipment corrosion, minimizing unplanned downtime and maintenance requirements. A stable and predictable production process fosters stronger partnerships with clients who prioritize supply security and business continuity in their sourcing strategies.
- Scalability and Environmental Compliance: The green chemistry principles embedded in this process, such as the use of a biodegradable support and reduced solvent usage, align perfectly with increasingly stringent global environmental regulations. Scaling up this reaction from laboratory to commercial tonnage is facilitated by the ease of handling solid catalysts in large reactors, avoiding the complexities of mixing and separating liquid homogeneous catalysts. The reduction in hazardous waste generation simplifies effluent treatment and lowers the environmental footprint of the manufacturing facility. This compliance advantage not only mitigates regulatory risks but also enhances the corporate sustainability profile, appealing to eco-conscious stakeholders and customers in the global pharmaceutical market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel trifluoromethylation technology, based on the specific data and findings presented in the patent documentation. These insights are intended to clarify the operational benefits and technical feasibility for potential partners evaluating this synthetic route for their supply chains.
Q: What are the advantages of the chitosan-supported copper catalyst over homogeneous systems?
A: The heterogeneous nature allows for easy filtration and recovery, eliminating toxic metal residues in the final API and enabling multiple catalytic cycles without significant loss of activity.
Q: What is the substrate scope for this trifluoromethylation reaction?
A: The method tolerates various substituents on both the quinoxalinone and indole rings, including methyl, ethyl, benzyl, chloro, bromo, and aldehyde groups, maintaining high yields above 80%.
Q: Is the catalyst reusable for industrial scale-up?
A: Yes, experimental data confirms the catalyst can be recycled at least 7 times with consistent catalytic efficiency, significantly reducing raw material costs for large-scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-(2-(trifluoromethyl)-indol-3-yl)quinoxalin-2-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the immense potential of this biomass-catalyzed trifluoromethylation technology to redefine the production standards for fluorinated pharmaceutical intermediates. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from patent literature to industrial reality is seamless and efficient. We are committed to delivering high-purity intermediates that meet rigorous quality standards, leveraging our state-of-the-art rigorous QC labs and analytical capabilities to verify the absence of metal residues and confirm structural integrity. Our dedication to process optimization allows us to maximize the benefits of this green chemistry approach, offering our clients a sustainable and cost-effective sourcing solution.
We invite you to collaborate with us to explore how this advanced synthetic route can enhance your product portfolio and reduce your overall manufacturing costs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate our capability to be your trusted partner in the development and supply of complex fluorinated building blocks.
