Advanced Copper-Catalyzed Synthesis of Alpha-Fluorinated Thioacrylamide Derivatives for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to incorporate fluorine atoms into organic scaffolds, driven by the unique metabolic stability and bioavailability enhancements that fluorine provides. Patent CN112341368A introduces a groundbreaking approach for the synthesis of α-fluorinated thioacrylamide derivatives, which serve as critical building blocks in the development of advanced agrochemicals, dyes, and water treatment agents. This innovation leverages a copper-catalyzed defluorinative thiolation strategy, utilizing readily available trifluoroacrylamides and disulfides to construct complex molecular architectures with high precision. By shifting away from traditional, hazardous reagents, this technology offers a pathway to high-purity intermediates that align with modern green chemistry principles while maintaining exceptional economic viability for large-scale manufacturing operations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of monofluoroalkenes and related thioacrylamide structures has relied heavily on the hydrofluorination of alkynyl sulfides or the defluorination of gem-difluoroolefins using malodorous mercaptans. These conventional pathways present significant operational challenges, including the requirement for harsh reaction conditions that can degrade sensitive functional groups and compromise overall product integrity. Furthermore, the use of volatile and foul-smelling mercaptans necessitates expensive containment systems and rigorous safety protocols to protect personnel and the environment, thereby inflating the capital expenditure required for facility setup. The limited substrate scope of these older methods often restricts their applicability, forcing process chemists to develop custom, non-scalable solutions for each new derivative, which delays time-to-market for critical active ingredients.
The Novel Approach
In stark contrast, the methodology disclosed in CN112341368A utilizes a mild, copper-catalyzed system that operates effectively at 80°C in tetrahydrofuran, significantly lowering the energy input compared to high-temperature alternatives. By employing disulfides as the sulfur source instead of thiols, the process inherently mitigates odor issues and enhances workplace safety, removing a major bottleneck in chemical manufacturing logistics. The reaction demonstrates remarkable versatility, accommodating a broad spectrum of substituents on both the amide nitrogen and the disulfide aromatic rings, which allows for the rapid generation of diverse chemical libraries. This streamlined approach not only simplifies the synthetic route by avoiding intermediate isolation but also achieves yields of up to 83 percent, establishing a new benchmark for efficiency in the production of fluorinated vinyl monomers.

Mechanistic Insights into Cu-Catalyzed Defluorinative Thiolation
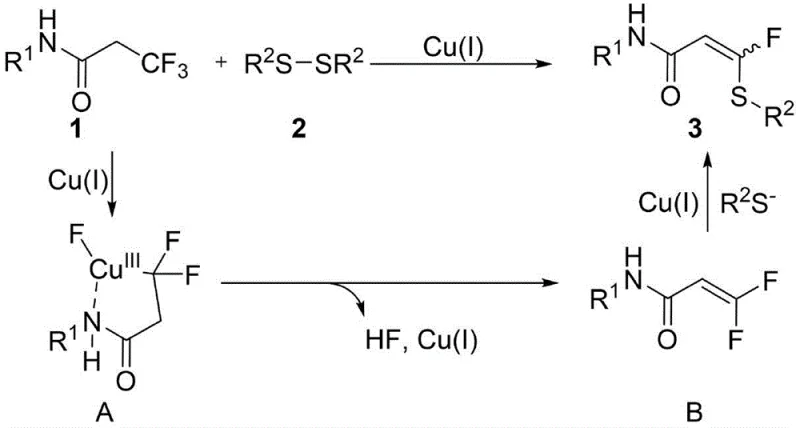
The core of this technological advancement lies in the intricate catalytic cycle mediated by copper species, specifically cuprous iodide, which facilitates the selective activation of carbon-fluorine bonds. The mechanism initiates with the generation of thiolate anions from disulfides under basic conditions, promoted by the presence of triphenylphosphine and water additives. Subsequently, the copper catalyst undergoes oxidative addition and coordinates with the nitrogen atom of the trifluoroacrylamide substrate to form a key N-chelating five-membered intermediate. This chelation is crucial as it positions the metal center for the subsequent beta-hydride elimination step, which generates a gem-difluoroacrylamide species in situ without the need for external oxidants or extreme thermal stress.
Following the formation of the difluoro-intermediate, the catalytic cycle proceeds with the rapid formation of a carbon-sulfur bond through the nucleophilic attack of the RS-anion, driven by the copper catalyst. This step is highly selective, ensuring that the fluorine atom is retained at the alpha position while the sulfur moiety is installed with high regiocontrol. The presence of water and phosphine ligands plays a pivotal role in stabilizing the active copper species and preventing catalyst deactivation, which is a common failure mode in transition metal catalysis. By understanding these mechanistic nuances, process engineers can fine-tune reaction parameters to minimize impurity profiles, ensuring that the final API intermediates meet the stringent purity specifications required by global regulatory bodies for pharmaceutical applications.
How to Synthesize Alpha-Fluorinated Thioacrylamide Derivatives Efficiently
Implementing this synthesis route requires careful attention to reagent stoichiometry and reaction monitoring to maximize the yield of the target fluorinated product. The process is designed to be operationally simple, avoiding the need for specialized high-pressure equipment or cryogenic conditions, which makes it accessible for standard multipurpose chemical plants. Operators should focus on maintaining the specified temperature of 80°C and ensuring adequate stirring to facilitate mass transfer between the solid base and the liquid phase. The detailed standardized synthesis steps below outline the precise protocol validated in the patent examples, providing a reliable framework for technology transfer and scale-up activities.
- Combine trifluoroacrylamide substrate, disulfide sulfur source, cuprous iodide catalyst, and potassium carbonate base in tetrahydrofuran solvent.
- Add triphenylphosphine and water as additives to the reaction mixture to facilitate the catalytic cycle and anion generation.
- Stir the reaction at 80°C for 12 hours, then filter, evaporate solvent, and purify the residue via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this copper-catalyzed methodology represents a strategic opportunity to optimize sourcing costs and enhance supply reliability. The shift from malodorous mercaptans to stable disulfides drastically simplifies the logistics of raw material handling, reducing the need for specialized storage tanks and ventilation systems that typically drive up facility overheads. Additionally, the use of commodity chemicals like potassium carbonate and tetrahydrofuran ensures that the supply chain remains resilient against market volatility, as these reagents are produced globally in massive quantities. This robustness translates directly into more predictable lead times and reduced risk of production stoppages due to raw material shortages, securing the continuity of supply for downstream customers.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous thiol reagents significantly lowers the direct material costs associated with the synthesis of these intermediates. Furthermore, the simplified workup procedure, which involves basic filtration and solvent evaporation rather than complex extraction or distillation sequences, reduces utility consumption and labor hours per batch. By avoiding the need for extensive heavy metal scavenging steps often required with other transition metal catalysts, the downstream purification costs are also substantially minimized, leading to a more favorable overall cost of goods sold.
- Enhanced Supply Chain Reliability: The broad substrate tolerance of this reaction means that manufacturers can source a variety of substituted trifluoroacetamides and disulfides from multiple vendors without compromising reaction performance. This flexibility prevents single-source dependency, a critical risk factor in global supply chains, and allows for agile switching between suppliers based on price and availability. The mild reaction conditions also reduce wear and tear on reactor vessels and ancillary equipment, extending asset life and decreasing maintenance downtime, which further stabilizes production schedules.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is straightforward due to the absence of exothermic hazards or gas evolution, allowing for safe operation in large-volume reactors. The reduction in waste solution discharge and the avoidance of noxious gas emissions align perfectly with increasingly strict environmental regulations, minimizing the liability and permitting hurdles associated with chemical manufacturing. This environmental compatibility not only protects the corporate reputation but also ensures long-term operational licenses in regions with rigorous ecological standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on process capabilities and limitations. Understanding these details is essential for R&D teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the advantages of using disulfides over mercaptans in this synthesis?
A: Using disulfides eliminates the handling of malodorous and toxic mercaptans, significantly improving operator safety and reducing the need for specialized scrubbing equipment in commercial manufacturing.
Q: What is the typical yield range for this copper-catalyzed defluorinative thiolation?
A: The patented method demonstrates robust performance with yields reaching up to 83 percent across a wide variety of substrates, ensuring high material efficiency for production.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process operates under atmospheric pressure at moderate temperatures (80°C) using common solvents like THF, making it highly scalable without requiring exotic high-pressure reactors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Fluorinated Thioacrylamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this copper-catalyzed defluorinative thiolation technology for the production of high-value fluorinated intermediates. As a seasoned CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless and efficient. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of validating the structural integrity and impurity profiles of complex fluorinated molecules, guaranteeing that every batch meets the exacting standards of the global pharmaceutical industry.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this greener, more efficient process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that will accelerate your product development timelines and enhance your competitive edge in the market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
