Advanced Manufacturing of Livistone C: A Noble-Metal-Free Synthetic Route for Commercial Scale
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and cost-effective synthetic routes for high-value natural product derivatives. A significant breakthrough in this domain is documented in patent CN103373974A, which details a novel preparation method for 2-(4-hydroxyphenyl)-5,7-dimethoxybenzofuran, also known as Livistone C. This compound, originally isolated from Hainan palmetto, exhibits a wide spectrum of biological activities including antifungal, anti-inflammatory, and anti-tumor properties, making it a highly sought-after pharmaceutical intermediate. The patented technology addresses critical bottlenecks in traditional synthesis by eliminating the reliance on expensive noble metal catalysts and harsh reaction conditions. By leveraging a sequence of bromination, Perkin condensation, and copper-catalyzed cascade reactions, this method offers a streamlined pathway that enhances atom economy and simplifies purification. For R&D directors and procurement specialists, understanding the nuances of this route is essential for evaluating its potential in reducing manufacturing costs and securing a stable supply of high-purity intermediates for drug development and material science applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-arylbenzofuran derivatives has been plagued by significant technical and economic challenges that hinder large-scale adoption. Conventional methods often rely on oxidative cyclization of o-vinylphenols using vanadium catalysts or dehydration cyclization of α-aryloxy ketones under strong acidic conditions. These traditional approaches frequently suffer from poor selectivity, particularly when dealing with substrates containing multiple hydroxyl groups, leading to complex impurity profiles that are difficult and expensive to remove. Furthermore, many established routes require the use of precious metal catalysts such as palladium or titanium, which not only inflate raw material costs but also introduce stringent regulatory requirements for heavy metal residue control in the final API. The reaction conditions for these legacy methods are often harsh, necessitating high temperatures, long reaction times, and the use of strong acids or bases, which can compromise equipment integrity and increase safety risks in a manufacturing environment. Additionally, the starting materials for these conventional pathways are not always readily available or are prohibitively expensive, creating supply chain vulnerabilities that can disrupt production schedules and increase lead times for downstream customers.
The Novel Approach
In stark contrast to these legacy limitations, the method disclosed in patent CN103373974A introduces a fundamentally different strategy that prioritizes efficiency, cost-effectiveness, and operational simplicity. This novel approach utilizes 3,5-dimethoxyphenylacetic acid and p-hydroxybenzaldehyde as starting materials, which are commercially abundant and inexpensive compared to specialized precursors used in other methods. The core innovation lies in a tandem sequence involving bromination followed by a Perkin reaction to construct the carbon skeleton, and subsequently, a copper-catalyzed cascade reaction that achieves hydroxylation, intramolecular cyclization, and decarboxylation in a highly controlled manner. By avoiding noble metals entirely and relying on abundant copper salts, the process drastically reduces the cost of goods sold (COGS) and simplifies the regulatory compliance landscape regarding heavy metal impurities. The reaction conditions are notably milder, operating at moderate temperatures and utilizing common organic solvents, which enhances process safety and reduces energy consumption. This strategic shift not only improves the overall yield but also ensures a cleaner reaction profile, thereby minimizing the need for complex chromatographic purifications and enabling a more straightforward path to commercial scale-up.
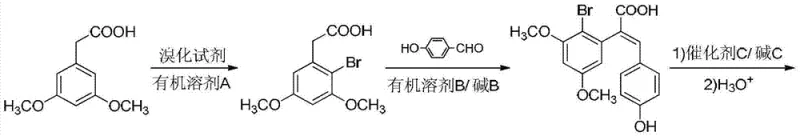
Mechanistic Insights into Copper-Catalyzed Cyclization and Decarboxylation
The heart of this synthetic innovation is the sophisticated cascade reaction that transforms the acrylic acid intermediate into the final benzofuran core. This step involves a complex interplay of hydroxylation, intramolecular ring closure, and dehydrogenation, all facilitated by a copper catalyst in an alkaline environment. Mechanistically, the copper species activates the aromatic ring towards nucleophilic attack, promoting the displacement of the bromine atom by a hydroxyl group generated in situ. This is immediately followed by an intramolecular cyclization where the phenolic oxygen attacks the activated alkene, closing the furan ring. The subsequent dehydrogenation restores aromaticity, stabilizing the benzofuran structure. This tandem process is remarkable because it accomplishes multiple bond-forming and bond-breaking events in a single operational step, significantly reducing the number of unit operations required. The use of copper sulfate as a catalyst is particularly advantageous due to its stability and low cost, while the alkaline conditions ensure the solubility of intermediates and drive the equilibrium towards the desired product. Understanding this mechanism is crucial for process chemists aiming to optimize reaction parameters such as temperature, catalyst loading, and base concentration to maximize yield and minimize byproduct formation during scale-up.
Following the formation of the benzofuran-3-carboxylic acid intermediate, the final step involves a decarboxylation reaction to yield the target Livistone C. This transformation is catalyzed by cuprous iodide in the presence of a ligand such as 1,10-phenanthroline, often under microwave irradiation or conventional heating in a high-boiling solvent like PEG-400. The decarboxylation mechanism likely involves the coordination of the carboxylate group to the copper center, facilitating the loss of carbon dioxide and the formation of the final C-H bond. The choice of solvent and ligand is critical here, as they stabilize the copper catalyst and enhance the reaction rate, allowing the process to proceed efficiently at relatively moderate temperatures compared to thermal decarboxylation methods. This step effectively removes the carboxyl group introduced during the Perkin condensation, finalizing the molecular architecture of the target compound. The ability to perform this decarboxylation under mild conditions without degrading the sensitive hydroxyl and methoxy substituents demonstrates the high chemoselectivity of the method, ensuring that the final product meets stringent purity specifications required for pharmaceutical applications.

How to Synthesize 2-(4-hydroxyphenyl)-5,7-dimethoxybenzofuran Efficiently
Implementing this synthesis route in a production environment requires careful attention to the specific operational parameters outlined in the patent to ensure reproducibility and safety. The process begins with the bromination of 3,5-dimethoxyphenylacetic acid, where controlling the stoichiometry of the brominating agent is vital to prevent over-bromination. Subsequent steps involve precise temperature control during the Perkin condensation and the cascade cyclization to manage exotherms and ensure complete conversion. The final decarboxylation step benefits from the use of microwave technology to accelerate reaction times, although conventional heating is also viable for larger batches. Detailed standard operating procedures (SOPs) must be established for each unit operation, including work-up and purification steps like recrystallization, to maintain consistent product quality. For a comprehensive guide on the exact reagent quantities, reaction times, and purification techniques, please refer to the standardized synthesis steps provided below.
- Bromination of 3,5-dimethoxyphenylacetic acid using DBDMH in chloroform at 40°C to form 2-bromo-3,5-dimethoxyphenylacetic acid.
- Perkin condensation with p-hydroxybenzaldehyde in acetic anhydride with triethylamine at 110°C to yield the acrylic acid intermediate.
- Cascade hydroxylation and intramolecular cyclization using copper sulfate and sodium hydroxide at 120°C, followed by copper-catalyzed decarboxylation.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented synthesis route offers substantial advantages that directly impact the bottom line and supply chain resilience for chemical manufacturers. By eliminating the need for precious metal catalysts like palladium, the process significantly reduces raw material costs, which is a major component of the overall manufacturing expense. This cost reduction is not merely theoretical; it translates into tangible savings that can be passed on to customers or reinvested into further R&D. Furthermore, the use of readily available starting materials mitigates the risk of supply disruptions that often plague specialized chemical markets. The simplicity of the post-treatment process, which relies on filtration and recrystallization rather than complex chromatography, reduces solvent consumption and waste generation, aligning with modern environmental sustainability goals. These factors combined create a robust manufacturing profile that is highly attractive for long-term supply agreements.
- Cost Reduction in Manufacturing: The elimination of noble metal catalysts is a primary driver for cost efficiency in this process. Traditional methods often require expensive palladium or platinum complexes, which not only have high upfront costs but also necessitate expensive recovery or disposal procedures to meet regulatory limits on heavy metals. By substituting these with inexpensive copper salts, the direct material cost is drastically lowered. Additionally, the high atom economy of the Perkin reaction and the tandem cyclization step means that a larger proportion of the starting material ends up in the final product, reducing waste disposal costs. The simplified purification process further contributes to cost savings by minimizing the volume of solvents and consumables required for isolation, making the overall process economically superior to conventional alternatives.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as 3,5-dimethoxyphenylacetic acid and p-hydroxybenzaldehyde ensures a stable and secure supply chain. Unlike specialized intermediates that may have limited suppliers and long lead times, these starting materials are produced by multiple manufacturers globally, reducing the risk of single-source dependency. The robustness of the reaction conditions also means that the process is less susceptible to variations in raw material quality, providing greater flexibility in sourcing. This reliability is crucial for procurement managers who need to guarantee continuous production schedules to meet downstream demand from pharmaceutical clients. The ability to scale this process from laboratory to industrial quantities without significant re-engineering further strengthens supply chain continuity.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, with reaction conditions that are manageable in large-scale reactors. The avoidance of extreme temperatures and pressures reduces the engineering complexity and capital expenditure required for plant equipment. From an environmental standpoint, the reduced use of toxic heavy metals and the potential for solvent recycling align with increasingly stringent global environmental regulations. The high yield and selectivity of the reaction minimize the generation of hazardous waste, simplifying compliance with waste disposal laws. This environmental friendliness not only reduces regulatory risk but also enhances the corporate social responsibility profile of the manufacturer, making it a preferred partner for eco-conscious multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of 2-(4-hydroxyphenyl)-5,7-dimethoxybenzofuran. These answers are derived directly from the technical specifications and beneficial effects described in the patent literature, providing clarity on the feasibility and advantages of this manufacturing route. Understanding these details is essential for stakeholders evaluating the technology for potential licensing or production partnerships.
Q: Does this synthesis route require expensive palladium catalysts?
A: No, the patented method explicitly avoids noble metals like palladium. It utilizes cost-effective copper salts (e.g., copper sulfate, cuprous iodide) and common organic bases, significantly reducing raw material costs and heavy metal residue risks.
Q: What are the critical reaction conditions for the cyclization step?
A: The cyclization involves a tandem hydroxylation/intramolecular ring closure/dehydrogenation reaction. It requires a strong base like sodium hydroxide (10% solution) and a copper catalyst at elevated temperatures around 120°C for approximately 72 hours to ensure complete conversion.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is designed for scalability. It uses readily available raw materials, avoids harsh conditions like extreme cryogenic temperatures, and employs simple post-treatment steps like filtration and recrystallization, making it highly viable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(4-hydroxyphenyl)-5,7-dimethoxybenzofuran Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the modern chemical industry. Our team of expert process chemists has extensively evaluated the technology described in patent CN103373974A and confirmed its viability for commercial production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific requirements of this synthesis, including the management of copper catalysts and the execution of precise temperature-controlled reactions. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs which employ advanced analytical techniques to verify every batch.
We invite procurement leaders and R&D directors to collaborate with us to optimize their supply chains for this valuable intermediate. By leveraging our manufacturing expertise, you can secure a reliable source of high-quality 2-(4-hydroxyphenyl)-5,7-dimethoxybenzofuran that supports your drug development or material science projects. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can enhance your project's success and reduce your overall time to market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
