Advanced Palladium-Catalyzed Direct Arylation for Scalable Pharmaceutical Intermediate Manufacturing
Advanced Palladium-Catalyzed Direct Arylation for Scalable Pharmaceutical Intermediate Manufacturing
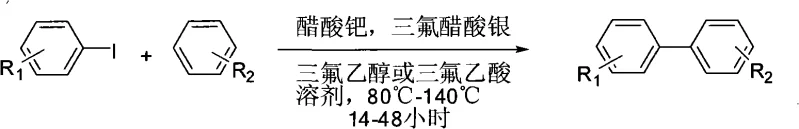
The landscape of organic synthesis for complex aromatic structures is undergoing a significant transformation, driven by the urgent need for more sustainable and cost-effective manufacturing processes. Patent CN101265147A introduces a robust methodology for the preparation of biaryl hydrocarbons, a structural motif ubiquitous in high-value active pharmaceutical ingredients (APIs) and advanced functional materials. This technology leverages a direct C-H activation strategy, bypassing the traditional requirement for pre-functionalized coupling partners. By utilizing a synergistic catalytic system composed of palladium acetate and silver trifluoroacetate in fluorinated media, this process achieves efficient carbon-carbon bond formation under relatively mild thermal conditions. For R&D directors and process chemists, this represents a pivotal shift away from multi-step sequences towards streamlined, atom-economical transformations that maintain high purity standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of biaryl scaffolds has relied heavily on classical coupling reactions such as the Ullmann reaction or the Suzuki-Miyaura coupling, both of which present distinct logistical and economic challenges in large-scale manufacturing. The Ullmann reaction, while effective, typically necessitates the use of stoichiometric amounts of copper powder, generating substantial quantities of heavy metal waste that complicate downstream purification and environmental compliance. Alternatively, the Suzuki reaction, though widely adopted, suffers from inherent inefficiencies regarding step count; it mandates the prior conversion of a simple arene into a halide, followed by metallation to form a boronic acid or ester. This pre-functionalization not only increases the consumption of reagents and solvents but also introduces additional unit operations that extend the overall production timeline and inflate the cost of goods sold (COGS) for the final intermediate.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN101265147A enables the direct coupling of iodoarenes with simple, unactivated arenes such as benzene or naphthalene in a single operational step. This ligand-free palladium-catalyzed system eliminates the need for expensive phosphine ligands and avoids the synthesis of organoboron reagents entirely. The reaction proceeds efficiently in polar solvents like 1,2-dichloroethane or trifluoroethanol, utilizing silver trifluoroacetate as a crucial additive to facilitate the catalytic cycle. By removing the prerequisite for substrate pre-activation, this approach drastically simplifies the synthetic route, offering a reliable pharmaceutical intermediates supplier pathway that reduces both material throughput and processing time while maintaining excellent regioselectivity and yield profiles suitable for industrial application.
Mechanistic Insights into Pd(II)-Catalyzed Direct C-H Arylation
The efficacy of this transformation relies on a sophisticated interplay between the divalent palladium catalyst and the silver additive, which collectively lower the activation energy barrier for C-H bond cleavage. The mechanism likely initiates with the oxidative addition of the iodoarene to the Pd(II) species, forming an aryl-palladium complex. Subsequently, the simple arene undergoes electrophilic palladation, a step significantly accelerated by the presence of trifluoroacetic acid or trifluoroethanol, which act as proton shuttles to facilitate the removal of the proton from the aromatic ring. The silver salt plays a dual role, potentially acting as a halide scavenger to maintain the cationic character of the palladium center and assisting in the re-oxidation or stabilization of the active catalytic species. This concerted action allows for the activation of relatively inert C-H bonds without the need for directing groups or harsh basic conditions often seen in other C-H functionalization protocols.
Furthermore, the tolerance of this catalytic system towards various electronic environments on the aromatic ring is a critical feature for process robustness. The patent data indicates that both electron-withdrawing groups, such as nitro substituents, and electron-donating groups, like methoxy moieties, are well-tolerated on the iodoarene component. This versatility ensures that the impurity profile remains manageable, as the reaction does not promote significant homocoupling of the iodoarene or poly-arylation of the simple arene under optimized conditions. For quality control teams, this means that the crude product typically requires only standard aqueous workup and column chromatography to achieve high purity, avoiding the complex crystallization sequences often needed to remove trace phosphine oxides or boron residues associated with alternative coupling strategies.
How to Synthesize Biaryl Hydrocarbons Efficiently
The implementation of this direct arylation protocol in a pilot or production setting requires careful attention to reagent stoichiometry and thermal management to maximize yield and safety. The process is notably forgiving regarding moisture and oxygen, as it does not demand rigorous anhydrous or anaerobic conditions, which simplifies reactor setup and operation. Detailed standardized synthesis steps see the guide below, which outlines the precise addition order and workup procedures validated in the patent examples to ensure reproducibility.
- Charge a reaction vessel with iodoarene, simple arene (5-50 equiv), palladium acetate (1-5 mol%), silver trifluoroacetate (0.6-1.2 equiv), and trifluoroethanol or trifluoroacetic acid.
- Heat the mixture to 80°C-140°C and stir for 14-48 hours under atmospheric conditions without strict anhydrous protection.
- Dilute with dichloromethane, wash with water to neutrality, dry the organic phase over magnesium sulfate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, adopting this direct arylation technology offers profound benefits for cost reduction in fine chemical manufacturing by fundamentally altering the bill of materials. The elimination of boronic acid derivatives, which are often subject to price volatility and supply constraints, stabilizes the raw material supply chain and reduces dependency on specialized reagent vendors. Additionally, the removal of phosphine ligands from the process recipe eliminates a significant cost driver, as these ligands are frequently expensive, air-sensitive, and require cold chain logistics for storage and transport. This simplification translates directly into lower inventory holding costs and reduced risk of material degradation prior to use.
- Cost Reduction in Manufacturing: The most significant economic advantage lies in the reduction of synthetic steps; by bypassing the preparation of organometallic coupling partners, manufacturers save on the solvents, reagents, and labor hours associated with those precursor syntheses. Furthermore, the use of simple arenes like benzene or naphthalene as coupling partners represents a shift towards commodity-grade feedstocks rather than custom-synthesized intermediates, driving down the baseline material cost per kilogram of the final product substantially.
- Enhanced Supply Chain Reliability: Utilizing broadly available starting materials mitigates the risk of supply disruptions that can occur with niche reagents. Since the reaction does not rely on exotic ligands or sensitive catalysts that require importation from limited global suppliers, procurement managers can source the majority of inputs locally or from multiple qualified vendors. This diversification enhances supply continuity and provides greater leverage in vendor negotiations, ensuring that production schedules remain uninterrupted even during market fluctuations.
- Scalability and Environmental Compliance: The simplified workup procedure, which involves standard aqueous washing and solvent removal, facilitates easier scale-up from laboratory to commercial production volumes without requiring specialized equipment for handling pyrophoric materials or toxic heavy metal wastes. The absence of stoichiometric copper waste, as seen in Ullmann-type reactions, significantly reduces the burden on wastewater treatment facilities and lowers the costs associated with hazardous waste disposal, aligning the process with increasingly stringent environmental regulations and corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this direct arylation technology, derived from the specific experimental data and claims within the patent documentation. These insights are intended to assist technical teams in evaluating the feasibility of integrating this method into existing manufacturing workflows for complex aromatic compounds.
Q: What are the primary advantages of this direct arylation method over Suzuki coupling?
A: Unlike Suzuki coupling, which requires the energy-intensive pre-synthesis of boronic acids or esters, this method utilizes simple, unfunctionalized arenes like benzene or naphthalene directly. This eliminates at least two synthetic steps, significantly reducing raw material costs and waste generation while improving overall atom economy.
Q: Does this catalyst system require expensive phosphine ligands?
A: No, a key innovation of patent CN101265147A is the use of a ligand-free palladium system stabilized by silver additives and fluorinated alcohols/acids. This removes the dependency on costly and air-sensitive phosphine ligands often required in traditional cross-coupling, simplifying storage and handling requirements.
Q: What is the typical yield range for this biaryl synthesis?
A: According to the experimental data provided in the patent, the reaction yields range from 56% to 88%, depending on the electronic nature of the substituents on the iodoarene and the specific reaction temperature employed between 80°C and 140°C.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Biaryl Hydrocarbon Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting innovative synthetic methodologies to maintain competitiveness in the global pharmaceutical and fine chemical markets. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory techniques like the Pd-catalyzed direct arylation described in CN101265147A can be successfully translated into robust industrial processes. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of biaryl intermediate meets the exacting standards required for downstream API synthesis, providing our partners with absolute confidence in material quality.
We invite you to collaborate with us to evaluate the potential of this technology for your specific project needs. Our experts are ready to provide a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this direct coupling route for your target molecules. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can optimize your supply chain and reduce your overall production costs.
